Long-term survival with CML with imatinib or transplantation as first-line treatment: Comparison of outcomes from CML Studies IIIA and IV
Summary
With introduction of the tyrosine kinase inhibitor (TKI) imatinib, the treatment strategy of CML has profoundly changed. TKIs became the fi rst-line treatment of choice for CML competing with allogeneic hematopoietic stem cell transplantation (HCT). Variables to be considered in choosing TKIs for fi rst-line therapy are as follows: conventional risk score; cytogenetic fi ndings with majorroute additional chromosomal aberrations (ACA) at diagnosis, and high-risk ACA in the course of CML; comorbidities; treatment costs. In cases of refractoriness to imatinib, the 2nd line treatment options are: clinical response milestones; adherence to therapy; resistance mutations; clonal evolution; therapy intolerance; drug safety; health care setting.
CML Study IV, a randomized treatment study concerning imatinib dose optimization and combined therapy with imatinib and cytarabine or interferon α included 1551 newly diagnosed patients in chronic phase. The key outcome was no superiority of survival of any treatment option. Imatinib 400 mg provides close to normal life expectancy in chronic-phase CML patients. Survival is independent of time to response. Outcome of CML is currently more determined by disease and patients’ factors, e.g., comorbidities and smoking, and by center eff ects than by initial treatment selection. A comparison of long-term survival aft er HCT or imatinib treatment showed that low risk patients had similar survival with both options. Attempts at improving treatment should focus on subgroups of refractory disease e.g. by HCT, and on non-CML determinants of survival. After progression to blast crisis, HCT did not provide a signifi cant survival advantage, although a special study showed that most long-term survivors (72%) were patients who received a transplant. The 10-year deep molecular remission rates of 70%-80% indicate that the majority of imatinib-treated patients are candidates for treatment discontinuation.
Keywords
Chronic myeloid leukemia, tyrosine kinase inhibitors, imatinib, treatment strategy, hematopoietic stem cell transplantation, survival.
Introduction
The only curative treatment for chronic myeloid leukemia (CML) was previously allogenous hemopoietic cell transplantation (HCT) [1]. With the introduction of the tyrosine kinase inhibitor (TKI) imatinib into CML management 15 years ago and the stunning response and survival results, treatment strategy of CML has profoundly changed. TKI became the first line treatment of choice for CML.
Long term survival
Meanwhile several long-term observational and randomized studies have matured and 10-year survival outcomes are available. An overview is shown in Table 1. 5-year survival ranges around 90%, 10-year survival around 83% and 10-year relative survival compared to the general population is more than 90% [2, 3]. Similar results have been observed in population based registries [4, 5, 6]. More patients died of comorbidities than of CML [7].
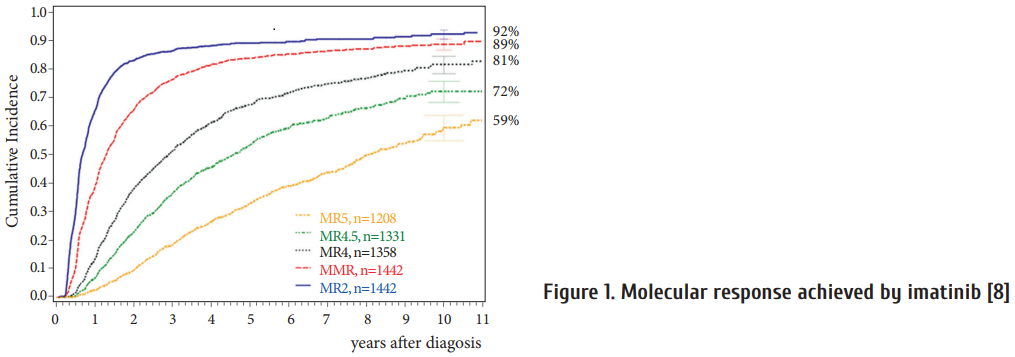
Deep molecular responses are achieved in up to 80% aft er 5 to 10 years (Figure 1) suggesting that treatment discontinuation should be possible in these patients [8].

First-line treatment
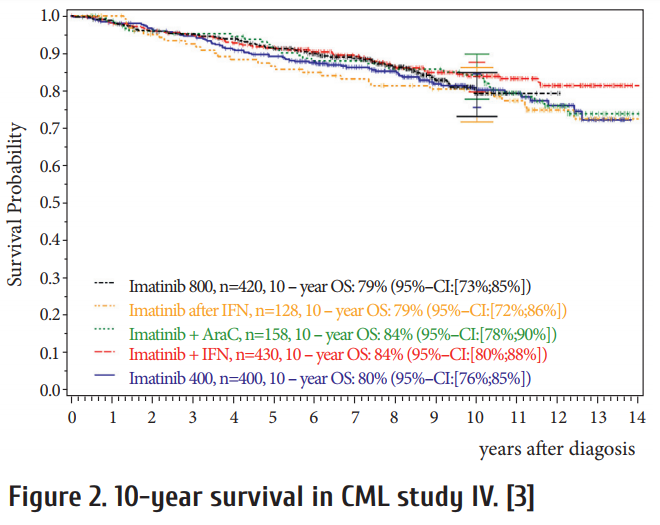
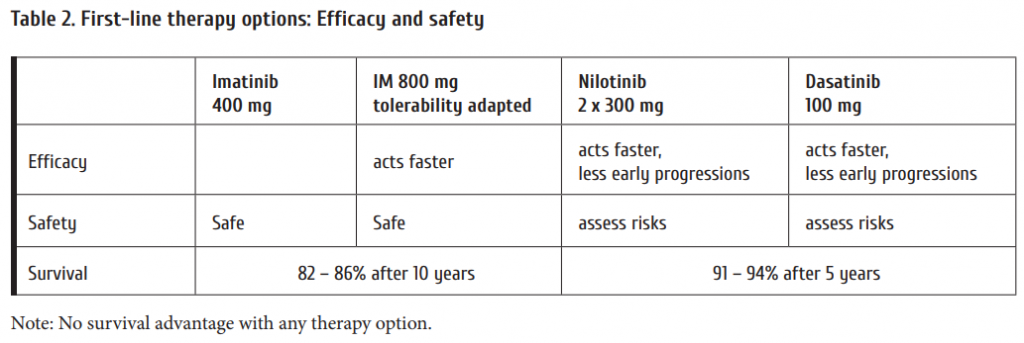
Current fi rst-line options with TKI are shown in Table 2. Whereas imatinib has been proven to be safe both at 400 and the faster acting 800 mg daily even aft er prolonged periods of time, the also faster acting 2nd generation (2G-)TKI require risk assessment due to rare but serious, potentially life threatening adverse drug reactions. As seen from Fig. 2, no survival advantage has been observed with any treatment option [3, 16, 17]. The lower progression rate to blast crisis observed with 2G-TKI is off set by more deaths due to adverse drug reactions. Variables to be considered in choosing first-line therapy are:
– Risk score;
– Cytogenetics (major-route ACA at diagnosis, high-risk
ACA in the course of CML);
– Comorbidities;
– Costs.
The impact of karyotype at diagnosis was demonstrated by Fabarius et al. [18]. Patients with major route ACA which occur in 1-2% of cases at diagnosis have a much poorer prognosis. Comorbidities do not infl uence progression of CML, but impact survival more than CML. Generic imatinib has become available recently. It decreases treatment costs at equal effi cacy and adds to the advantages of matinib over 2G-TKI.
Second-line therapy
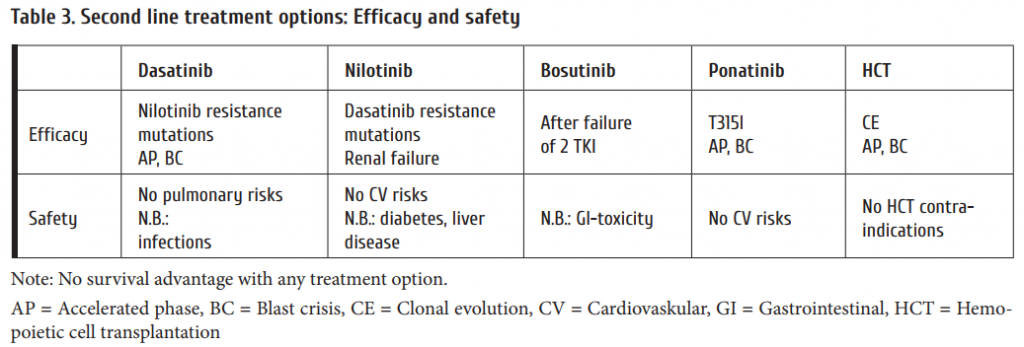
2nd-line therapy is needed in cases of refractoriness to imatinib. Table 3 summarizes comparative efficacy and safety of 2nd-line treatment options. The variables to be considered for second line therapy are:
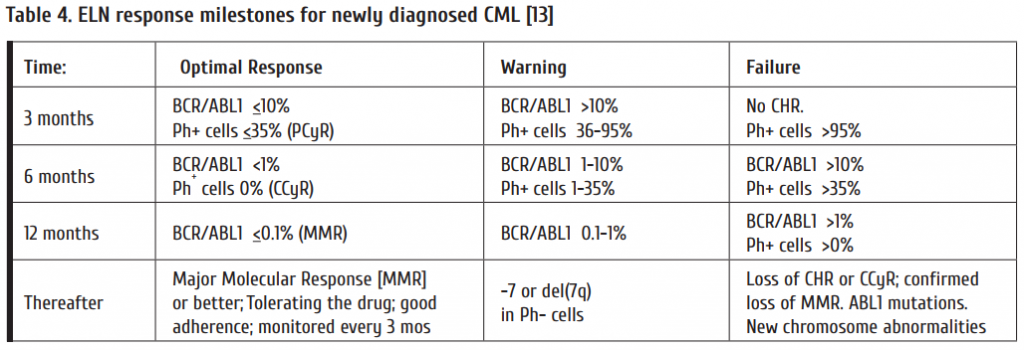
– Response milestones (Table 4);
– Adherence to therapy;
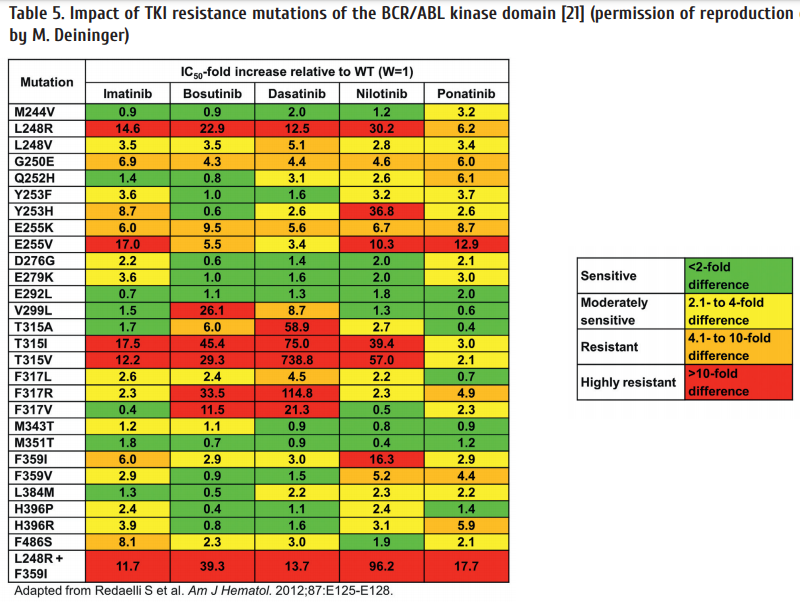
– Resistance mutations (Table 5);
– Clonal evolution;
– Intolerance;
– Drug safety;
– Health care setting.
The criteria for assessing TKI-response were proposed by the European LeukemiaNet (ELN) for newly diagnosed CML [13] and are depicted in Table 4. Before changing treatment due to resistance, non-adherence to drug-treatment has to be excluded. Non-adherence has been reported as the most frequent reason for treatment failure [19].
When changing treatment due to confi rmed resistance a mutation analysis should be initiated. This can be done simultaneously with changing to the new drug. If the new drug still does not work, the mutation analysis will give a rational basis for selecting the right drug. Table 5 lists the most important mutations and there sensitivity to the currently available TKI.
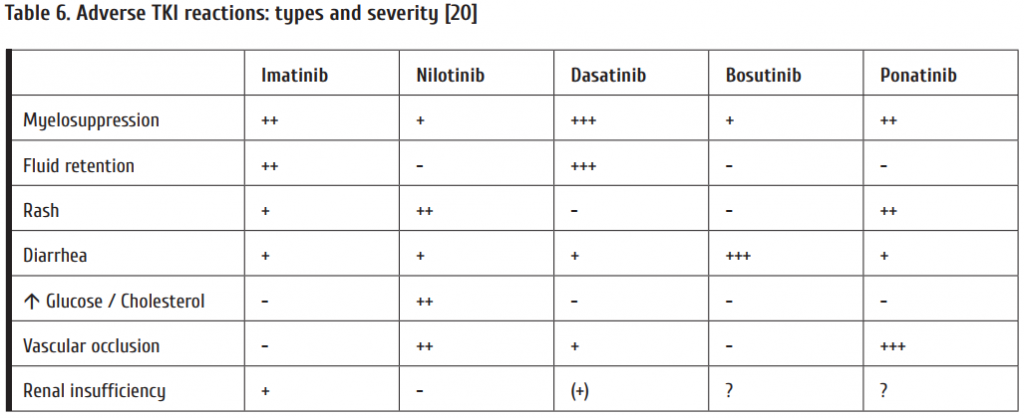
Adverse TKI reactions have recently been reviewed on behalf of ELN by [20]. Table 6 gives an overview over the most frequently observed adverse TKI reactions.
CML Studies IIIA and IV
CML study IIIA is a geneticly randomized study comparing allogenous HCT with best available drug treatment. It recruited 662 patients, randomized 427 eligible patients (family donor available vs not available) and was published after a median observation time of 12.1 years [15]. Th e key result was equivalence of outcome for low risk patients after transplantion, if performed within one year of diagnosis, and imatinib.
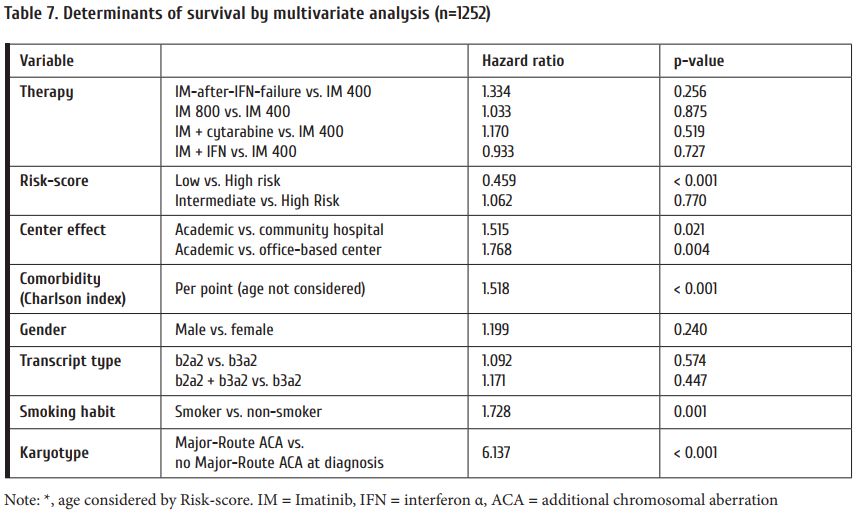
CML study IV is a randomized 5-arm treatment optimization study to explore whether treatment with imatinib 400mg can be improved by doubling the dose, combining imatinib with cytarabine or interferon α (IFN) or applying imatinib aft er IFN failure. 1551 newly diagnosed patients in chronic phase where recruited and the study published after a median observation time of 9.5 years [3]. The key outcome was no superiority of survival of any treatment option (Fig. 2) in spite of signifi cantly faster responses with imatinib 800 mg and the recognition of determinants of survival independent of treatment by multivariate analysis (Table 7).
A comparison of long-term survival aft er HCT or drug treatment showed that low risk patients had similar survival with both options (Fig. 3) [22, 23].
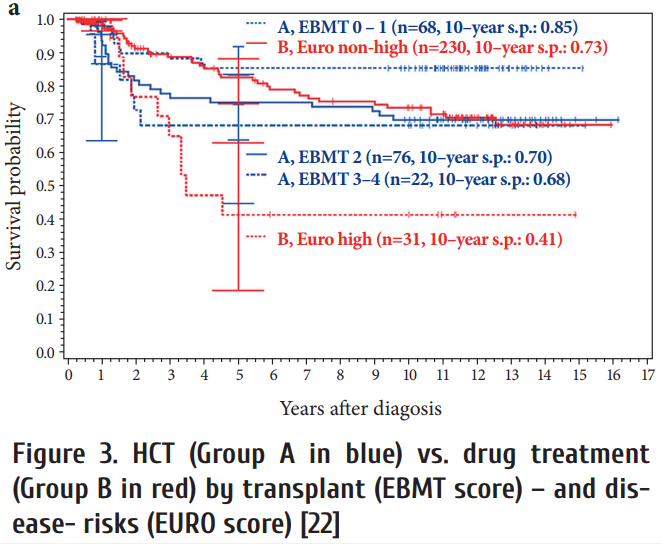
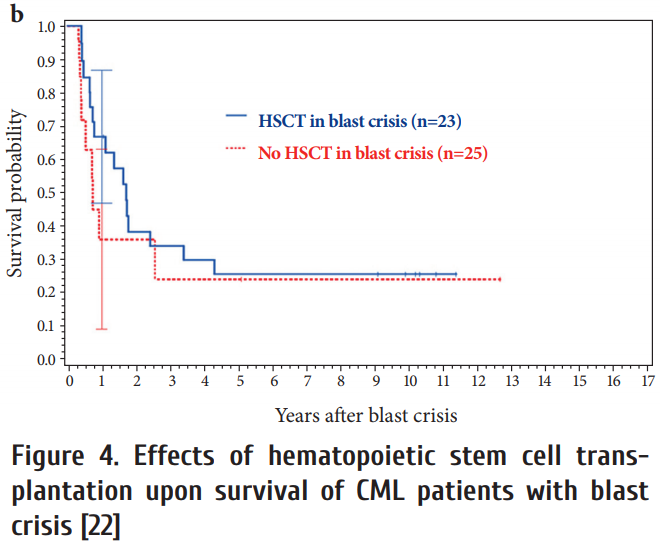
After progression to blast crisis HCT did not provide a significant survival advantage (Fig. 4), although long-term observations of 699 blast crises from the German CML studies showed that most long-term survivors (72%) were patients who received a transplant [24].
Conclusion
• Imatinib 400 mg provides close to normal life expectancy in chronic-phase CML patients.
• Survival is independent of time to response.
• Outcome of CML is currently more determined by disease and patients’ factors e.g. comorbidities and smoking, and by center effects than by initial treatment selection.
• In low risk patients survival aft er imatinib and transplantation may be similar.
• Attempts at improving treatment should focus on subgroups of refractory disease e.g. by HCT, and on non-CML determinants of survival.
• The 10-year deep molecular remission rates of 70%–80% indicate that the majority of imatinib treated patients are candidates for treatment discontinuation.
Conflict of interest
The author has no conflicts of interest to declare.
References
1. Goldman JM. Chronic myeloid leukemia: reversing the chronic phase. J Clin Oncol. 2010;28(3):363-365.
2. Pfirrmann M, Baccarani M, Saussele S, Guilhot J, Cervantes F, Ossenkoppele G, Hoff mann VS, Castagnetti F, Hasford J, Hehlmann R, Simonsson B. Prognosis of long-term survival considering disease-specifi c death in patients with chronic myeloid leukemia. Leukemia. 2016;30(1):48-56.
3. Hehlmann R, Lauseker M, Saußele S, Pfi rrmann M, Krause S, Kolb HJ, Neubauer A, Hossfeld DK, Nerl C, Gratwohl A, Baerlocher GM, Heim D, Brümmendorf TH, Fabarius A, Haferlach C, Schlegelberger B, Müller MC, Jeromin S, Proetel U, Kohlbrenner K, Voskanyan A, Rinaldetti S, Seifarth W, Spieß B, Balleisen L, Goebeler MC, Hänel M, Ho A, Dengler J, Falge C, Kanz L, Kremers S, Burchert A, Kneba M, Stegelmann F, Köhne CA, Lindemann HW, Waller CF, Pfreundschuh M, Spiekermann K, Berdel WE, Müller L, Edinger M, Mayer J, Beelen DW, Bentz M, Link H, Hertenstein B, Fuchs R, Wernli M, Schlegel F, Schlag R, de Wit M, Trümper L, Hebart H, Hahn M, Thomalla J, Scheid C, Schafhausen P, Verbeek W, Eckart MJ, Gassmann W, Pezzutto A, Schenk M, Brossart P, Geer T, Bildat S, Schäfer E, Hochhaus A, Hasford J. Assessment of imatinib as first-line treatment of chronic myeloid leukemia: 10-year survival results of the randomized CML study IV and impact of non-CML determinants. Leukemia. 2017;31(11):2398-2406.
4. Höglund M, Sandin F, Hellström K, Björeman M, Björkholm M, Brune M, Dreimane A, Ekblom M, Lehmann S, Ljungman P, Malm C, Markevärn B, Myhr-Eriksson K, Ohm L, Olsson-Strömberg U, Själander A, Wadenvik H, Simonsson B, Stenke L, Richter J. Tyrosine kinase inhibitor usage, treatment outcome, and prognostic scores in CML: report from the population-based Swedish CML registry. Blood. 2013;122(7):1284-1292.
5. Thielen N, Visser O, Ossenkoppele G, Janssen J. Chronic myeloid leukemia in the Netherlands: a population-based study on incidence, treatment, and survival in 3585 patients from 1989 to 2012. Eur J Haematol. 2016;97(2):145-154. 6. Bower H, Björkholm M, Dickmann P, Höglund M, Lambert PC, Andersson TM. Life expectancy of patients with CML approaches the life expectancy of the general population. J Clin Oncol 2016;34(24): 2851-2858.
7. Saussele S, Krauss MP, Hehlmann R, Lauseker M, Proetel U, Kalmanti L, Hanfstein B, Fabarius A, Kraemer D, Berdel WE, Bentz M, Staib P, de Wit M, Wernli M, Zettl F, Hebart HF, Hahn M, Heymanns J, Schmidt-Wolf I, Schmitz N, Eckart MJ, Gassmann W, Bartholomäus A, Pezzutto A, Leibundgut EO, Heim D, Krause SW, Burchert A, Hofmann WK, Hasford J, Hochhaus A, Pfirrmann M, Müller MC; Schweizerische Arbeitsgemeinschaft für Klinische Krebsforschung and the German CML Study Group. Impact of comorbidities on overall survival in patients with chronic myeloid leukemia: results of the randomized CML study IV. Blood 2015;126(1):42-49.
8. Kalmanti L, Saussele S, Lauseker M, Müller MC, Dietz CT, Heinrich L, Hanfstein B, Proetel U, Fabarius A, Krause SW, Rinaldetti S, Dengler J, Falge C, Oppliger-Leibundgut E, Burchert A, Neubauer A, Kanz L, Stegelmann F, Pfreundschuh M, Spiekermann K, Scheid C, Pfi rrmann M, Hochhaus A, Hasford J, Hehlmann R. Safety and efficacy of imatinib in CML over a period of 10 years: data from the randomized CML-study IV.Leukemia. 2015; 29(5):1123-1132.
9. Hochhaus A, Larson RA, Guilhot F, Radich JP, Branford S, Hughes TP, Baccarani M, Deininger MW, Cervantes F, Fujihara S, Ortmann CE, Menssen HD, Kantarjian H, O’Brien SG, Druker BJ; IRIS Investigators. Long-Term Outcomes of Imatinib Treatment for Chronic Myeloid Leukemia. N Engl J Med. 2017;376(10):917-927.
10. Palandri F, Castagnetti F, Alimena G, Testoni N, Breccia M, Luatti S, Rege-Cambrin G, Stagno F, Specchia G, Martino B, Levato L, Merante S, Liberati AM, Pane F, Saglio G, Alberti D, Martinelli G, Baccarani M, Rosti G. Th e long-term durability of cytogenetic responses in patients with accelerated phase chronic myeloid leukemia treated with imatinib 600 mg: the GIMEMA CML Working Party experience after a 7-year follow-up. Haematologica. 2009;94(2):205-12.
11. de Lavallade H, Apperley JF, Khorashad JS, Milojkovic D, Reid AG, Bua M, Szydlo R, Olavarria E, Kaeda J, Goldman JM, Marin D. Imatinib for newly diagnosed patients with chronic myeloid leukemia: incidence of sustained responses in an intention-to-treat analysis. J Clin Oncol. 2008;26(20):3358-3363.
12. Cervantes F, López-Garrido P, Montero MI, Jonte F, Martínez J, Hernández-Boluda JC, Calbacho M, Sureda A, Pérez-Rus G, Nieto JB, Pérez-López C, Román-Gómez J, González M, Pereira A, Colomer D. Early intervention during imatinib therapy in patients with newly diagnosed chronic- phase chronic myeloid leukemia: a study of the Spanish PETHEMA group. Haematologica. 2010 Aug;95(8):1317- 1324.
13. Baccarani M, Druker BJ, Branford S, Kim DW, Pane F, Mongay L, Mone M, Ortmann CE, Kantarjian HM, Radich JP, Hughes TP, Cortes JE, Guilhot F. Long-term response to imatinib is not aff ected by the initial dose in patients with Philadelphia chromosome-positive chronic myeloid leukemia in chronic phase: fi nal update from the Tyrosine Kinase Inhibitor Optimization and Selectivity (TOPS) study. Int J Hematol. 2014;99(5):616-624.
14. Jain P, Kantarjian H, Alattar ML, Jabbour E, Sasaki K, Nogueras Gonzalez G, Dellasala S, Pierce S, Verstovsek S, Wierda W, Borthakur G, Ravandi F, O’Brien S, Cortes J. Long-term molecular and cytogenetic response and survival outcomes with imatinib 400 mg, imatinib 800 mg, dasatinib, and nilotinib in patients with chronic-phase chronic myeloid leukaemia: retrospective analysis of patient data from fi ve clinical trials. Lancet Haematol. 2015;2(3):e118-28.
15. Gambacorti-Passerini C, Antolini L, Mahon FX, Guilhot F, Deininger M, Fava C, Nagler A, Della Casa CM, Morra E, Abruzzese E, D’Emilio A, Stagno F, le Coutre P, Hurtado- Monroy R, Santini V, Martino B, Pane F, Piccin A, Giraldo P, Assouline S, Durosinmi MA, Leeksma O, Pogliani EM, Puttini M, Jang E, Reiff ers J, Piazza, Valsecchi MG, Kim DW. Multicenter independent assessment of outcomes in chronic myeloid leukemia patients treated with imatinib. J Natl Cancer Inst. 2011;103(7):553-561.
16. Hochhaus A, Saglio G, Hughes TP, Larson RA, Kim DW, Issaragrisil S, le Coutre PD, Etienne G, Dorlhiac-Llacer PE, Clark RE, Flinn IW, Nakamae H, Donohue B, Deng W, Dalal D, Menssen HD, Kantarjian HM. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia. 2016 May;30(5):1044-1054.
17. Cortes JE, Saglio G, Kantarjian HM, Baccarani M, Mayer J, Boqué C, Shah NP, Chuah C, Casanova L, Bradley-Garelik B, Manos G, Hochhaus A.Final 5-Year Study Results of DASISION: Th e Dasatinib Versus Imatinib Study in Treatment-Naïve Chronic Myeloid Leukemia Patients Trial. J Clin Oncol. 2016;34(20):2333-2340.
18. Fabarius A, Kalmanti L, Dietz CT, Lauseker M, Rinaldetti S, Haferlach C, Göhring G, Schlegelberger B, Jotterand M, Hanfstein B, Seifarth W, Hänel M, Köhne CH, Lindemann HW, Berdel WE, Staib P, Müller MC, Proetel U, Balleisen L, Goebeler ME, Dengler J, Falge C, Kanz L, Burchert A, Kneba M, Stegelmann F, Pfreundschuh M, Waller CF, Spiekermann K, Brümmendorf TH, Edinger M, Hofmann WK, Pfi rrmann M, Hasford J, Krause S, Hochhaus A, Saußele S, Hehlmann R; SAKK and the German CML Study Group. Impact of unbalanced minor route versus major route karyotypes at diagnosis on prognosis of CML. Ann Hematol. 2015;94(12):2015-2024.
19. Noens L, Hensen M, Kucmin-Bemelmans I, Lofgren C, Gilloteau I, Vrijens B. Measurement of adherence to BCRABL inhibitor therapy in chronic myeloid leukemia: current situation and future challenges. Haematologica. 2014; 99(3):437-447.
20. Steegmann JL, Baccarani M, Breccia M, Casado LF, García-Gutiérrez V, Hochhaus A, Kim DW, Kim TD, Khoury HJ, Le Coutre P, Mayer J, Milojkovic D, Porkka K, Rea D, Rosti G, Saussele S, Hehlmann R, Clark RE. European LeukemiaNet recommendations for the management and avoidance of adverse events of treatment in chronic myeloid leukaemia.Leukemia. 2016;30(8):1648-1671.
21. Eiring AM, Deininger MW. Individualizing kinase-targeted cancer therapy: the paradigm of chronic myeloid leukemia. Genome Biol. 2014 Sep 17;15(9):461. doi: 10.1186/ s13059-014-0461-8.
22. Gratwohl A, Pfi rrmann M, Zander A, Kröger N, Beelen D, Novotny J, Nerl C, Scheid C, Spiekermann K, Mayer J, Sayer HG, Falge C, Bunjes D, Döhner H, Ganser A, Schmidt-Wolf I, Schwerdtfeger R, Baurmann H, Kuse R, Schmitz N, Wehmeier A, Fischer JT, Ho AD, Wilhelm M, Goebeler ME, Lindemann HW, Bormann M, Hertenstein B, Schlimok G, Baerlocher GM, Aul C, Pfreundschuh M, Fabian M, Staib P, Edinger M, Schatz M, Fauser A, Arnold R, Kindler T, Wulf G, Rosselet A, Hellmann A, Schäfer E, Prümmer O, Schenk M, Hasford J, Heimpel H, Hossfeld DK, Kolb HJ, Büsche G, Haferlach C, Schnittger S, Müller MC, Reiter A, Berger U, Saußele S, Hochhaus A, Hehlmann R; SAKK; German CML Study Group. Long-term outcome of patients with newly diagnosed chronic myeloid leukemia: a randomized comparison of stem cell transplantation with drug treatment. Leukemia. 2016 Mar;30(3):562-569.
23. Saussele S, Lauseker M, Müller MC, Gratwohl A, Beelen D, Bunjes DW et al. Allogeneic hematopoietic stem cell transplantation (HSCT) in the imatinib-era: update on the survival outcome following allogeneic HSCT after imatinib failure; results of the German CML study IV. Blood. 2014;124 (ASH Meeting abstract No. 2567).
24. Hehlmann R, Saussele S. Treatment of chronic myeloid leukemia in blast crisis. Haematologica 2008; 93:1765-1769.


