Study of immunomodulating effect of lenalidomide in up front therapy of follicular lymphoma
Darima S. Badmazhapova, Irina V. Galtseva, Yulia O. Davydova, Nikolay M. Kapranov, Evgeni E. Zvonkov, Elena N. Parovichnikova
National Research Center for Hematology, Moscow, Russia
Summary
Introduction
According to modern concepts of lymphoma pathogenesis tumor cells could evade the immune surveillance and cause T-cell anergy. This is the basis for using immunomodulatory drugs in therapy, such as lenalidomide. It enhances the immune synapse formation by increasing the expression of costimulatory molecules on tumor B-cells, cytotoxic activity of T-cells and differentiation of T-helpers. Our aim was to study the changes in PD-1, PD-L1, FAS, CD80, CD86 proportion in tumor cells and subpopulations of T-cells during therapy with lenalidomide in a patient with follicular lymphoma.Clinical case and methods
The study was performed in one patient (m, 32 y.o.) with follicular lymphoma (t(14;18), cytological type 2, stage IVB, high risk FLIPI-2), who underwent an upfront therapeutic protocol: iv rituximab 375 mg/m2 once, lenalidomide with an initial dose 5 mg/day with gradual escalation up to 20 mg/day for 27 days. Lenalidomide was intererrupted due to the remaining large tumor mass, and R-CHOP was started. Blood samples were tested prior to and after rituximab, on day 1, day 2, day 5, day 7 and day 21 of lenalidomide and in day 3 after its interruption. PD-1-, PD-L1-, FAS-, CD80-, CD86-positive cells were evaluated from all the tumor cells. PD-1- and PD-L1-positive cells were determined from CD4+ and CD8+ T cells anda subset of naïve (NV, CD28+CD95-), effector (EF, CD28-CD95+), memory cells (ME, CD28+CD95+) by flow cytometry (BD FACS CantoII). Twenty-nine donor blood samples were studied as a control.
Results
Before treatment, there were 35.6% CD4+PD-1+ cells (in donors, 2.0%-20.7%); CD8+PD-1+ 23.6% (in donors, 1.8%-23.5%). The following results were seen after rituximab: CD4+PD-1+ decreased by 3.5 times (10.8%); CD8+PD-1+, by 2.5 times (9.4%). During lenalidomide therapy: CD4+PD-1+ gradually increased to 25.0%; CD8+PD-1+ shifted up to 18.8%. After lenalidomide interruption, the CD4+PD-1+ population increased to 44.9%. On day 2 of lenalidomide therapy, the CD4+PD-L1+ increased by 6.8 times (from 0.5% to 3.4%), CD8+PD-L1+ increased 4 times (from 0.3 to 1.2%). On day 21: CD4+PD-L1+, and CD8+PD-L1+ showed a 4.4% and 2.5% increase, respectively. After lenalidomide interruption, CD4+PD-L1+ and CD8+PD-L1+ decreased to 1.1% and 0.5%, respectively. CD4+ memory effectors (MEs) increased from 42.9% to 70.6%, and CD4+ naïve (NVs) decreased from 56.4% to 27.9% during lenalidomide therapy. After its interruption, there was a growth of NV cells to 32.8% and decrease of ME to 66.2%. On day 5: CD8+EFs increased from 19.3% to 59.9%. CD8+EFs decreased and reached baseline on the 3rd day after lenalidomide interruption.Stimulatory and inhibitory
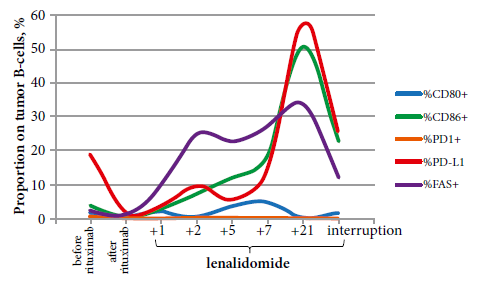
Figure 1.
Co-expression rates of stimulatory and inhibitory molecules on tumor cells were low: CD80+, 1,9% (in donor B cells, 0.0%-33.6%); CD86+, 3,9% (in donors, 0.0%-32.0%); FAS+, 23% (in donors 5.4%-38.0%); PD-1 0.7% (in donors 0.0%-3.8%); PD-L1+, 19.1% (in donors, 5.6%-69.4%) before lenalidomide. During lenalidomide therapy, amount of tumor cells expressing CD86, FAS, PD-L1 increased up to 50.8%, 33.9%, 57.6%, and decreased after therapy interruption to 23.3% 12.5%, 26.1%, respectively. Tumor B- cells CD19+CD80+, CD19+PD-1+ did not change during all the period of therapy (Figure 1).


