Introduction
Implementation of modern treatment strategies in leukemia patients, e.g., extensive usage of novel targeted drugs have resulted into sufficient increase of complete remission rates and higher survival rates. However, introduction of the new-generation treatment is associated with additional adverse effects including granulocytopenia and injuries of tissue barriers (skin and mucosae). These side effects cause significant risks for infectious complications, up to bloodstream infections and generalized septic conditions. Spectrum of detectable microbial pathogens in the hematooncological patients was sufficiently changed over last decades. I.e., Gram-positive microflora predominated in the 90’s, making up to 70% of the microbial findings [13]. More recently, however, an increasing proportion of Gram-negative infections was registered in the immunocompromised patients [4]. Potential role of viral pathogens, especially, herpesviruses, in development of bloodstream infections is also poorly understood, despite their common reactivation during mixed infections in hemato-oncological patients. [5].
At present time, a septic state is determined as a generalized inflammatory response to infectious pathogens of different origin, either bacterial, fungal, parasitic, or viral etiology. Clinical criteria of the sepsis diagnostics first approved in 1992, are well known and applicable worldwide [6]. In accordance with these criteria, sepsis is considered a systemic inflammation syndrome in response to infectious factor(s).
Viral infections are widely known to play a significant role in genesis of infectious complications in patients with hemato-oncological disorders. In most cases, the virus-related conditions are caused by reactivation of latent virus(es), whereas reinfections seems to be more rare. Viral reactivation is most commonly detected for herperviruses, e.g., cytomegalovirus (CMV), Herpes Simplex types 1 and 2 (HSV 1/2), Varicella Zoster virus (VZV). CMV is considered a most common risk factor for life-threatening infections in hemato-oncology patients However, some other viruses (adenovirus, hepatitis B virus) may also undergo reactivation in immunocompromised patients. In particular, respiratory viral infections are common to this cohort.
Before implementation of pre-emptive antiviral therapies in oncohematology, CMV infection was associated with severe organ affection and high mortality rates among recipients of hematopoietic stem cells (HSCs). In our previous experience [11], mortality of the patients with clinical CMV disease was 32% (7/22). Most cases of CMV disease were registered following allogeneic HSCT. Noteworthy, the CMV disease was accompanied by different aggravating conditions (bacterial sepsis, graft rejection, multi-organ failure, GvHD (graft versus host diseases), thus presuming severity of the condition and worse prognosis. CMV disease following auto-HSCT, was associated with retarded engraftment. Meanwhile, only 5% (3/66) of leukemia patients who received intensive chemotherapy have developed CMV disease (hemorrhagic cystitis, cytopenic fever, interstitial pneumonia). Anyway, clinically sound CMV reactivation may prevent a full-scale chemotherapy, thus suggesting worse prognosis for primary malignancy.
Modern approaches to management of CMV infection allowed to decrease risk of fatalities in this cohort. There exist, however, some open questions concerning pathogenesis of CMV infections and their role as an underlying factor for development of other viral and microbial infections, e.g., respiratory conditions.
The aim of our study was to assess some features of emerging infectious complications in immunocompromised oncohematological patients and possible role of herpesviruses in their genesis.
Patients and methods
The first case series included eighty-five randomly chosen adult patients with oncohematological disorders (Table 1). We assessed general frequency of infectious complications and their etiological agents focusing, mainly, on mixed infections of bacterial, fungal and viral origin. Throat smears, venous blood, as well as urine and sputum specimens were taken for bacteriology and virological testing under standard examination protocol at the Hematology Clinics.
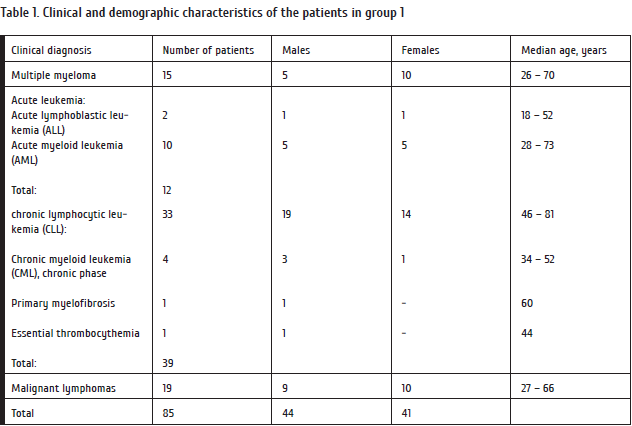
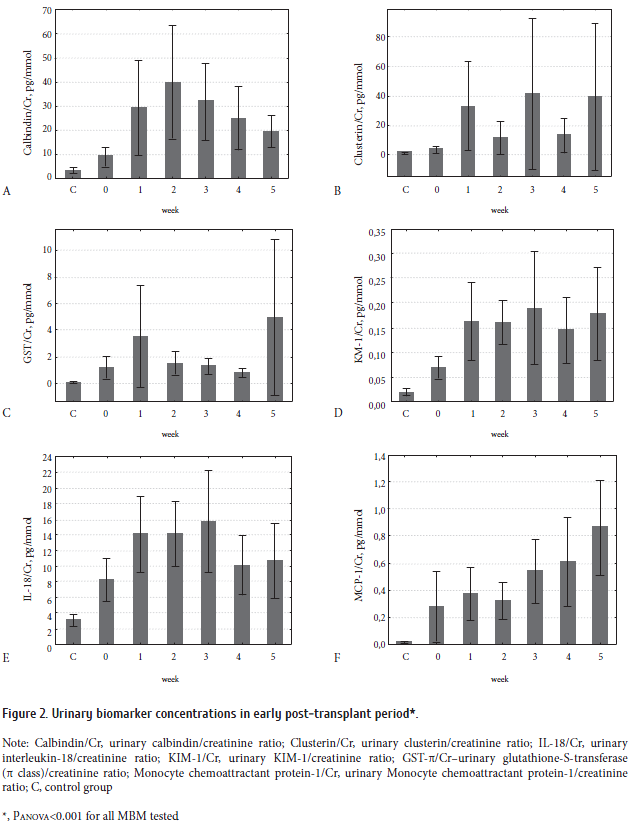
As seen from Table 1, 80% of the patients were admitted with CLL (n=33), malignant lymphomas (n=19), multiple myeloma (n=15). Acute leukemias made only 12% of the group.
All the patients received specific cytostatic therapy according to their primary diagnosis, i.e., cytostatic drugs, monoclonal antibodies, proteasome inhibitors, immunomodulatory drugs, tyrosine kinase inhibitors, hypomethylating drugs. Appropriate treatment protocols were as follows: 7+3 for AML; 7+3+ATRA for acute promyelocytic leukemia; VRP-prolong for ALL; R-CHOP for non-Hodgkin’s lymphoma etc.
Among these patients, infectious complications during chemotherapy or after its completion have been revealed in 50 cases (63% of total). Thirty-five patients who did not show clinical signs of infection comprised a comparison group. The patients with clinical infections were administered empirical therapy with broad-spectrum antibiotics. The drug therapy was corrected according to in vitro antibiotic sensitivity testing.
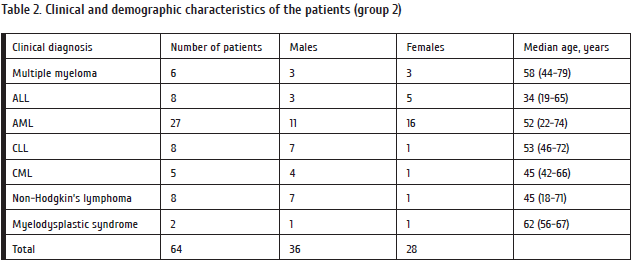
To assess rates of clinical conditions and outcomes of the bloodstream infections, as well as to specify possible complicating role of herpesviruses, we carried out an additional retrospective analysis of 64 clinical cases of different oncohematological disorders treated at the Department of Hematology of our Institute within 2005-2013 (Series 2, Table 2). The patients’ data were collected from clinical charts. Primary microbiological information was retrieved from the laboratory worksheets. All positive hemocultures and other microbiological findings were documented in worksheets.
We have analyzed individual data on blood bacteriological testing in patients admitted to our Department of Hematology and treated for different myelo- and lymphoproliferative disorders (acute and chronic leukemias, multiple myeloma, non-Hodgkin’s lymphomas, myelodysplastic syndrome).
Primary diagnosis in the patients was based on routine hematological examinations, i.e., blood counts, bone marrow punctures, trephine biopsies, cytogenetic, molecular biology, biochemical and immunological parameters. Multiple myeloma (MM) was confirmed by monoclonal protein revealed in blood serum or urine, along with lytic skeletal lesions detected upon X ray radiography. Diagnostics of lymphomas and chronic lymphatic leukemia included abdominal and retroabdominal ultrasonography, CT scanning, NMR and PET (if indicated), as well as immunohistochemical examination of affected lymph nodes, blood and marrow. CML diagnosis was based on detection of Ph'-chromosome and chymeric BCR/ABL. Phenotyping in acute leukemia was performed using cytochemical and immunological tests.
Antibacterial therapy was started immediately upon diagnosis of infection. First-line therapy was usually performed at empirical basis, in accordance with conventional approaches and at average therapeutic doses [3].
Due to severe clinical state of the patients, antimicrobial therapy usually included two broad-spectrum antibiotics (third- and fourth-generation cephalosporins, aminoglycosides, fluoroquinolones), sometimes combined with metronidazole. Two or three days later, upon getting data on antibiotics resistance, the antimicrobial therapy could be modified, carbapenems were added in the most severe cases. Immune correction included immunoglobulin-based drugs (Pentaglobin, Octagam), being an obligate therapeutic component. When detecting herpesviruses (EBV, HSC-1/2, HHV6, or CMV) the therapy was accomplished by antiviral drugs, i.e., Acyclovir or Valacyclovir (for CMV treatment).
Diagnostics of septic conditions was based on clinical data (evidence of primary site and port of infection, septic syndrome, secondary sites of infection), and confirmed by the pathogen isolation in culture. During Grade IV neutropenia (< 0.5×109/L), clinical diagnosis of infectious conditions was suggested upon single increase of the body temperature (>38.0 оС), or repeated hyperthermia over 38.5 оС within 24 hours which could not be explained by clinical course of the primary disorder, or by the treatment performed. Systemic inflammatory reaction syndrome was based upon registration of, at least, 2 of 4 following clinical symptoms: [6].
– body temperature > 38 °С, or < 36 °С;
– heart rate > 90 per minute;
– breathing rate > 20 per minute, or hyperventilation (pСО2 < 32 mm Hg)
– peripheral leukocyte counts > 12×109/L, or < 4×109/L, or at > 10% immature forms.
In most cases, the blood specimens for bacteriological culture were taken 2, 3 or more times. The primary bacterial strain isolated from hemoculture was regarded as the etiological agent. Bacteriological analyses and identification of micromycetes were performed by uniform technique over the entire study period, according to the valid guidelines [15]. Micromycetes were morphologically identified in culture, by evaluating their growth patterns in agar cultures and fluid media. Enzyme activities and filamentation were used for detection of yeast-like microflora. Standard disc diffusion methods were applied to assess antibiotic sensitivity of isolated strains. E test was applied to determine minimal inhibiting concentration (MIC). The test kits were purchased from AB Biodisc (Sweden) [2].
DNA was extracted from peripheral blood leukocytes. For DNA-diagnostics, we used gene-specific PCR with real- time registration at ANK32 thermocycler. Commercial kits for multiplex PCR were from InterLabService (AmpliSens ®, Moscow, Russia) allowing to detect respiratory viruses (RSV, influenza А/В и А/H1N1sw types) parainfluenza virus (PIV), rhinovirus, adenovirus, coronavirus, metapneumovirus). The herpesvirus panel included Herpes Simplex type 1 and 2 (HSV); Cytomegalovirus (CMV); Epstein-Barr virus (EBV), and Human Herpesvirus type 6 (HHV6), as well as Clamydophila pneumoniae and Mycoplasma pneumoniae. PCR techniques were performed according to manufacturer instructions. The studies were performed at an. The declared analytical sensitivity for the test systems was 500…1000 copies/ mL for HSV1/2, and 5×105 per 105 leukocytes for EBV, CMV, and HHV type 6.
Statistical evaluation of the data was performed by means of STATISTICA 6.0 software, using χ2 and Student criteria. Correlation quotients and their significance were determined by Spearman criterion. The differences were considered significant at Р< 0.05.
Results
Our primary task was to evaluate frequencies of viral infections in the patients treated by intensive cytostatic therapy. Results of the virological study for Group 1 (herpesviruse and respiratory infections) are shown in Table 3.
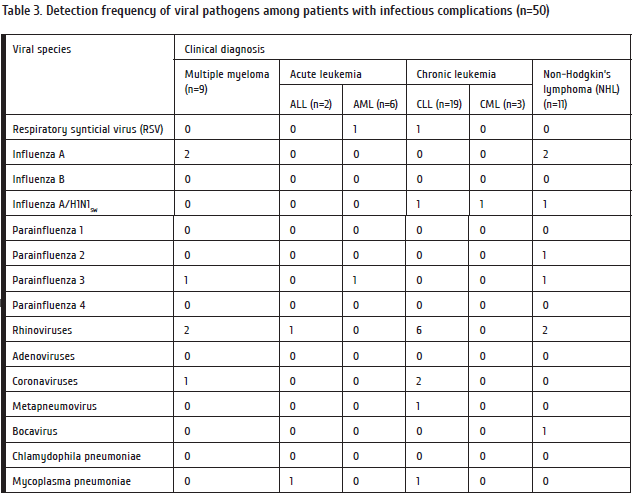
RSV infection manifesting with fever was found in two patients (association with М.pneumonia in 1 case). Influenza А virus was detected in four patients exhibiting high fever (up to 39.0оС). Three cases of А/H1N1sw influenza were observed in 2009. A single patient with CLL treated with Fludarabine+Cyclophosphamide developed a mixed А/H1N1/rhinovirus infection. All the patients with influenza exhibited prolonged fever (< 38.6 оС) followed by pneumonia in one case of diffuse large-cell B lymphoma after R-CHOP. PIV 2 and 3 viruses were detectable in four patients accompanied by high fever (up to 38.5 ºС). In a patient with follicular lymphoma treated by R-CHOP regimen, an association of PIV 3 and bocavirus was found. PIV 3 infection was also registered in AML patient after the 7+3 chemotherapy and grade severe granulocytopenia followed by acute focal pneumonia. A patient with multiple myeloma after a course of Bortezomib/Dexamethasone developed positivity for PIV 3, with herpes labialis.
Rhinovirus DNA was most common in the studied group, with only mild clinical manifestations, i.e., subfebrile temperature. However, an acute unilateral pneumonia was registered in one ALL patient. An association of rhinovirus with 229Е coronavirus was found in one CLL patient. Coronaviruses were detected in 3 cases. The patients exhibited respiratory symptoms (cough, chills), subfebrile state. Metapneumovirus was shown in only 1 case of CLL, manifesting with subfebrility and respiratory symptoms.
The infections were accompanied by herpesvirus reactivation in 42% of the cases. HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. HHV6-specific DNA was not found in any sample from the patients. Antiviral therapy was initiated in patients with herpesvirus- positive blood samples.
Bacteriological study of the throat smears showed prevalence of normal Gram-positive microflora typical to oral microbiota. In 1 case, however, a patient with CLL had E.Coli bacteraemia, in absence of other evident local or systemic pathogens.
In comparison group (infection-free patients), infectious agents were found in 7/35 (20%) of the cases. I.e., rhinoviruses was revealed in 3 patients, coronaviruses, in 2 cases, and M. рneumoniae, in 2 patients. Anti-infectious treatment was not provided in these cases.
Blood stream infections group: etiological structure
Our second group included cases with proven sepsis primarily diagnosed by clinical criteria. Table 3 presents data on Gram-positivity and species composition of blood-borne microorganisms in patients with hemoblastoses observed from 1991 to 2013. In general, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, relative incidence of these microbial classes showed a clear predominance of Gram-negative bacteria over Gram-positives in 1993, 2004, and 2005. The percentage of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p < 0.05). Coagulase-negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria. Noteworthy, incidence of Pseudomonas spp. and Klebsiella spp. decreased over last decade.
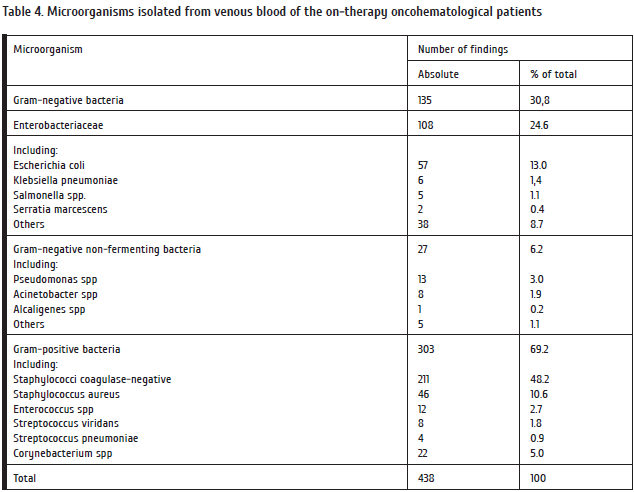
A trend towards a higher incidence of Micromycetae among total microflora was also observed. E.g., their proportion in blood cultures was rather low in 2009, followed by increased detection of Micromycetae in 2010, 2011 and 2012 (respectively, 3.3, 9.7, and 5.3%).
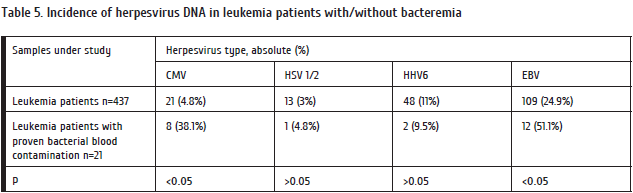
Another task of our study was to specify a potential role of herpesviruses in bacteriaemia observed in leukemia patients. To test this hypothesis, we performed a comparative study of herpesvirus frequency in cases of proven bacterial infections in bloodstream. A comparison group consisted of leukemia patients free of detectable bacteremia. (Table 5). We have revealed a significant increased EBV and CMV incidence in blood of the patients who developed bacteremia, as compared with bacteriemia-free cases.
Possible combined effects of blood stream infections and viral reactivation upon clinical outcomes
We have analyzed distinct laboratory features of sepsis in the patients with resistant/relapsing hemoblastoses and lymphomas pre-treated with intensive chemotherapy (Tables 6 and 7).
As seen from Table 6, E.coli was the most common Gram-negative pathogen found in bloodstream (9 episodes). In one patient, E.сoli bacteremia was combined with EBV, while the second had CMV reactivation. Other bacteria were more rare (Pseudomonas aeruginosa – 1 case with EBV, HSV 1/2 and HHV6. In 2 cases, bacteremia was caused by Moraxella spp., all the cases were associated with herpesviruses (1, with CMV, and one, with a combination of CMV, HSV 1/2, HHV6, and EBV). Despite massive treatment with the most effective drugs, four patients with Gram-negative sepsis developed septic shock with multi-organ failure. Overall mortality in Gram-negative sepsis was 57% (8 cases).
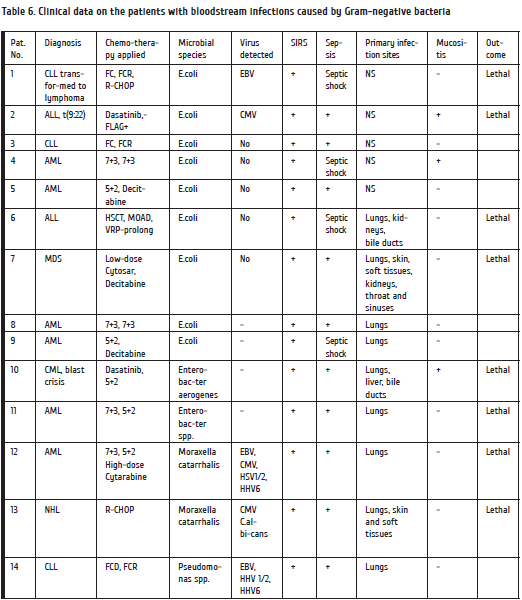
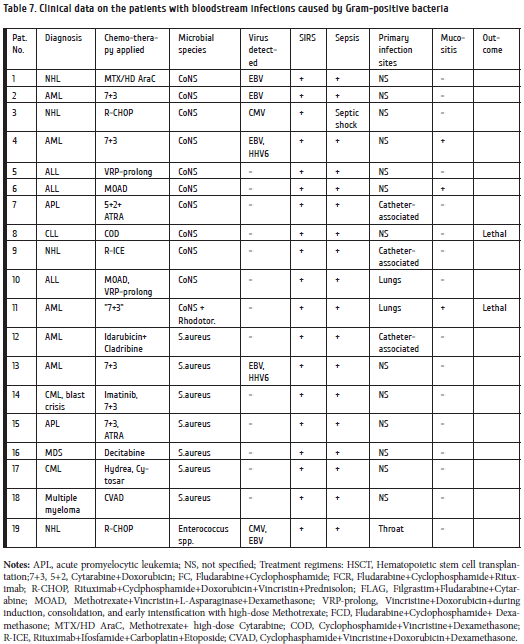
Etiological role of Gram-positive microbes is more discutable. As seen from Table 7, CoNS were most common Gram-positive microbes isolated. A universal recommendation for detection of the microbes in blood specimens is to administer multiple blood sampling. In most cases, we used a more practical and quite efficient recommendation based on the terms of in vitro bacterial outgrowth, e.g., the test is likely to be positive if it is detected at short terms of culture (< 3…5 days). Late outgrowth suggests an artifactual microbial contamination [1]. Based on these criteria and clinical data, we have suggested a sufficient role of CoNS in systemic inflammatory response for eleven patients with different hematological tumors following intensive cytostatic chemotherapy.
Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: two cases were associated with EBV, and one, with CMV positivity. A combination of HHV6/EBV was found in one patient, thus potentially proving the immunocompromised condition in these cases. In four patients, we have found a constellation of three herpesvirus types, i.e., EBV, CMV, and HHV6. Therefore, a high ratio of viral and bacterial co-infections may be typical to a number of patients with proven sepsis after intensive cytostatic chemotherapy.
Discussion
Modern methods of leukemia treatment allow to sufficiently increase complete remission rates and to increase survival of the patients. The role of infectious complications following intensive leukemia therapy becomes increasingly high, due to improved diagnostics of infectious pathogens over last decade. Urgent diagnostics of these infections permits timely usage of specific therapy. Two decades ago, most pneumonia cases in such patients were classified as ‘unknown origin’ since full-scale PCR assays for viruses were not used, like as express diagnostics of bacterial cultures. Introduction of molecular biology testing allowed to extend opportunities of viral diagnostics in immunocompromised patients with leukemias and lymphoma [5].
We used real-time PCR approach covering the main set of respiratory pathogens. Application of this diagnostic set combined with bacteriological methods allowed to assess etiology of infectious processes in 62% of cases. In comparison group (leukemia patients without clinical signs of infections), the infectious agents were revealed in 7/35 (20%) of the patients. In general, our data are in accordance with results by G.Gerna et al.[7], who performed their studies by similar scenario in the patients receiving lung transplants.
Noteworthy, development of respiratory infections in the patients under study was accompanied by rather common incidence (42%) of herpesvirus detection, especially, CMV and EBV which are associated with immunosuppressive conditions. The data presented are confirming a significant role of viral pathogens, especially, herpesviruses, which are potential factors of immunosuppression and concomitant bacterial infections in leukemia patients. The data from second group of patients seem to confirm this finding. E.coli was commonly revealed in patients’ blood.
Moraxella catarrhalis was associated with septicemia in two cases, both accompanied by herpesvirus activation. Previously, M.catarrhalis was considered a causal factor for otitis media, sinusitis, and conjunctivitis in children. Later on, a role of this microorganism was suggested for septicemias in acute leukemia patients [8, 12]. Therefore, M.catarrhalis seems to play a distinct role in development of infectious complications in immunocompromised patients with oncohematological disorders [10, 14].
What concerns etiological significance of Gram-positive microorganisms in bacteremia, one should mind a high probability of skin contamination, e.g., with coagulase-negative staphylococci, thus increasing chances for false-positive results of hemocultures, due to the CoNS transfer during vein puncture. A possible role of CoNS in systemic inflammatory syndrome was assessed in 57.8% of hematological patients. Of them, 36% exhibited EBV, CMV, or HHV6 in blood leukocytes. Hence, the observed viral co-infection may be a biological marker of a generalized immune suppression. Nevertheless, one should bear in mind a probable independent role of herpesviruses in pathogenesis of infectious complications.
In the present study, antibacterial therapy started with β-lactame antibiotics combined with fluoroquinolones, aminoglycosides, metronidazole. If required, the antimicrobial strategy was revised 48 to 72 hours later as based on clinical and microbiological data, applying carbapenems also combined with other anti-infectious drugs. Nevertheless, mortality rates in Gram-negative and Gram-positive sepsis comprised, respectively, 57% and 11%. These patients suffered with severe primary malignancies, being mostly resistant to cytostatic treatment, with bulky disease and expressed immunosuppression due to drug-induced cytopenia.
One should mind a role of fungi in evolving infectious complications which may occur at any step of anticancer therapy, due to decreased innate immunity. Appropriate risk factor include neutropenia, damage to skin, intestinal mucosa, early administration of broad-spectrum antibiotics, glucocorticosteroids, immunosuppressive drugs that favor colonization with Candida spp. [9]. When treating these patients, we used Fluconazole, Voriconazole, or Caspofungin.
Conclusion
In summary, our data support a general viewpoint on regular monitoring of infectious pathogens upon intensive chemotherapy of oncohematological patients prone to both bacterial and viral infections. Severe infectious complications (pneumonia, sepsis) are often associated with fungal invasions, and herpesvirus reactivation.
In particular, our results suggest that different viruses, e.g., herpesviruses, may cause immunosuppression, or may serve as additional immunodeficiency markers predictive for bacterial infections at later terms. Most patients who developed severe infections (e.g., AML and NHL cases) are potential candidates for hematopoietic stem cell transplantation. Therefore, one should take into account their predisposition for infectious complications when planning HSCT for these patients.
Conflict of interests
No conflict of interests is declared.
References
- Souvenir D, Anderson DE Jr, Palpant S et al. Blood Cultures Positive for Coagulase-Negative Staphylococci: Antisepsis, Pseudobacteremia, and Therapy of Patients. J Clin Microbiol 1998; 36 (7): 1923–1926
- Baker CN, Stocker SA, Culver DH et al. Comparison of E-test to agar dilution broth microdilution and agar diffusion susceptibility testing techniques by using a special challenge set of bacteria. J Clin Microbiol 1991; 29: 533-538.
- Bessmeltsev SS, Abdulkadyrov KM. Multiple myeloma: The Physicians’ Guide. Moscow: MK Publishers, 2016, 504 p. (In Russian)
- Chebotkevich V, Kiseleva E, Stizhak N et al. Epidemiology and clinical characteristics of bloodstream infections in hematological cancer patients. Haematologica 2016; 101 (s1): 765.
- Chebotkevitch V, Volkov A. Community respiratory virus infections in patients with haematological malignancies // 17th ECCMID, March 31 – April 3, 2007. Munich, Germany. Ab. 589
- Dellinger RP, Levy MM, Rhodes A et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock. 2012 Intensive Care Med 2013; 39 (2): 165-228.
- Gerna G, Vitulo P, Rovida F, Lilleri D, Pellegrini C, Oggionni T, Campanini G, Baldanti F, Revello MG. Impact of human metapneumovirus and human cytomegalovirus versus other respiratory viruses on the lower respiratory tract infections of lung transplant recipients. J Med Virol 2006; 78 (3): 408-416.
- Henny FC, Mulder CJ, Lampe AS, van der Meer JW et al. Branhamella catarrhalis septicaemia in a granulocytopenic patient. Infection 1984; 12(3):208–209.
- Bhatt VR, Viola GM, Ferrajoli A. Invasive Fungal Infections in Acute Leukemia. Ther Adv Hematol. 2011; 2(4): 231–247.
- Leszczyсska K, Jakoniuk P, Sacha PT et al. Susceptibility of Branhamella catarrhalis to antibiotics. Med Dosw Mikrobiol 2004; 56: 231–237.
- Moiseev SI, Nuia ML, Chebotkevich VN, Gonchar VA, Abdulkadyrov KM. Cytomegalovirus infection in practice of bone marrow transplantation. Terapevticheskyi Archiv 2002; 74 (7): 44-48. (In Russian)
- Saito H, Anaissie EJ, Khardori N et al. Branhamella catarrhalis septicemia in patients with leukemia. Cancer 1988; 61 (11): 2315–2317.
- Girmenia C, Menichetti F. Current epidemiology and prevention of infectious complications in cancer patients. European Oncology and Haemotology 2011; 7 (4): 270-277.
- Sirwar SB, Indupalli AS, Pal R et al. Moraxella catarrhalis: an emerging pathogen in bronchopulmonary infections. Ann Trop Med Publ Health 2013; 6(1): 76–79.
- Versalovic J, Carroll KC, Funke G, Jorgensen JH, Landry ML, Warnock DW. (Eds.). Manual of Clinical Microbiology, 10th Edition, Vol. 1, 2011, ASM Press.
" ["~DETAIL_TEXT"]=> string(27613) "
Introduction
Implementation of modern treatment strategies in leukemia patients, e.g., extensive usage of novel targeted drugs have resulted into sufficient increase of complete remission rates and higher survival rates. However, introduction of the new-generation treatment is associated with additional adverse effects including granulocytopenia and injuries of tissue barriers (skin and mucosae). These side effects cause significant risks for infectious complications, up to bloodstream infections and generalized septic conditions. Spectrum of detectable microbial pathogens in the hematooncological patients was sufficiently changed over last decades. I.e., Gram-positive microflora predominated in the 90’s, making up to 70% of the microbial findings [13]. More recently, however, an increasing proportion of Gram-negative infections was registered in the immunocompromised patients [4]. Potential role of viral pathogens, especially, herpesviruses, in development of bloodstream infections is also poorly understood, despite their common reactivation during mixed infections in hemato-oncological patients. [5].
At present time, a septic state is determined as a generalized inflammatory response to infectious pathogens of different origin, either bacterial, fungal, parasitic, or viral etiology. Clinical criteria of the sepsis diagnostics first approved in 1992, are well known and applicable worldwide [6]. In accordance with these criteria, sepsis is considered a systemic inflammation syndrome in response to infectious factor(s).
Viral infections are widely known to play a significant role in genesis of infectious complications in patients with hemato-oncological disorders. In most cases, the virus-related conditions are caused by reactivation of latent virus(es), whereas reinfections seems to be more rare. Viral reactivation is most commonly detected for herperviruses, e.g., cytomegalovirus (CMV), Herpes Simplex types 1 and 2 (HSV 1/2), Varicella Zoster virus (VZV). CMV is considered a most common risk factor for life-threatening infections in hemato-oncology patients However, some other viruses (adenovirus, hepatitis B virus) may also undergo reactivation in immunocompromised patients. In particular, respiratory viral infections are common to this cohort.
Before implementation of pre-emptive antiviral therapies in oncohematology, CMV infection was associated with severe organ affection and high mortality rates among recipients of hematopoietic stem cells (HSCs). In our previous experience [11], mortality of the patients with clinical CMV disease was 32% (7/22). Most cases of CMV disease were registered following allogeneic HSCT. Noteworthy, the CMV disease was accompanied by different aggravating conditions (bacterial sepsis, graft rejection, multi-organ failure, GvHD (graft versus host diseases), thus presuming severity of the condition and worse prognosis. CMV disease following auto-HSCT, was associated with retarded engraftment. Meanwhile, only 5% (3/66) of leukemia patients who received intensive chemotherapy have developed CMV disease (hemorrhagic cystitis, cytopenic fever, interstitial pneumonia). Anyway, clinically sound CMV reactivation may prevent a full-scale chemotherapy, thus suggesting worse prognosis for primary malignancy.
Modern approaches to management of CMV infection allowed to decrease risk of fatalities in this cohort. There exist, however, some open questions concerning pathogenesis of CMV infections and their role as an underlying factor for development of other viral and microbial infections, e.g., respiratory conditions.
The aim of our study was to assess some features of emerging infectious complications in immunocompromised oncohematological patients and possible role of herpesviruses in their genesis.
Patients and methods
The first case series included eighty-five randomly chosen adult patients with oncohematological disorders (Table 1). We assessed general frequency of infectious complications and their etiological agents focusing, mainly, on mixed infections of bacterial, fungal and viral origin. Throat smears, venous blood, as well as urine and sputum specimens were taken for bacteriology and virological testing under standard examination protocol at the Hematology Clinics.

As seen from Table 1, 80% of the patients were admitted with CLL (n=33), malignant lymphomas (n=19), multiple myeloma (n=15). Acute leukemias made only 12% of the group.
All the patients received specific cytostatic therapy according to their primary diagnosis, i.e., cytostatic drugs, monoclonal antibodies, proteasome inhibitors, immunomodulatory drugs, tyrosine kinase inhibitors, hypomethylating drugs. Appropriate treatment protocols were as follows: 7+3 for AML; 7+3+ATRA for acute promyelocytic leukemia; VRP-prolong for ALL; R-CHOP for non-Hodgkin’s lymphoma etc.
Among these patients, infectious complications during chemotherapy or after its completion have been revealed in 50 cases (63% of total). Thirty-five patients who did not show clinical signs of infection comprised a comparison group. The patients with clinical infections were administered empirical therapy with broad-spectrum antibiotics. The drug therapy was corrected according to in vitro antibiotic sensitivity testing.
To assess rates of clinical conditions and outcomes of the bloodstream infections, as well as to specify possible complicating role of herpesviruses, we carried out an additional retrospective analysis of 64 clinical cases of different oncohematological disorders treated at the Department of Hematology of our Institute within 2005-2013 (Series 2, Table 2). The patients’ data were collected from clinical charts. Primary microbiological information was retrieved from the laboratory worksheets. All positive hemocultures and other microbiological findings were documented in worksheets.
We have analyzed individual data on blood bacteriological testing in patients admitted to our Department of Hematology and treated for different myelo- and lymphoproliferative disorders (acute and chronic leukemias, multiple myeloma, non-Hodgkin’s lymphomas, myelodysplastic syndrome).

Primary diagnosis in the patients was based on routine hematological examinations, i.e., blood counts, bone marrow punctures, trephine biopsies, cytogenetic, molecular biology, biochemical and immunological parameters. Multiple myeloma (MM) was confirmed by monoclonal protein revealed in blood serum or urine, along with lytic skeletal lesions detected upon X ray radiography. Diagnostics of lymphomas and chronic lymphatic leukemia included abdominal and retroabdominal ultrasonography, CT scanning, NMR and PET (if indicated), as well as immunohistochemical examination of affected lymph nodes, blood and marrow. CML diagnosis was based on detection of Ph'-chromosome and chymeric BCR/ABL. Phenotyping in acute leukemia was performed using cytochemical and immunological tests.
Antibacterial therapy was started immediately upon diagnosis of infection. First-line therapy was usually performed at empirical basis, in accordance with conventional approaches and at average therapeutic doses [3].
Due to severe clinical state of the patients, antimicrobial therapy usually included two broad-spectrum antibiotics (third- and fourth-generation cephalosporins, aminoglycosides, fluoroquinolones), sometimes combined with metronidazole. Two or three days later, upon getting data on antibiotics resistance, the antimicrobial therapy could be modified, carbapenems were added in the most severe cases. Immune correction included immunoglobulin-based drugs (Pentaglobin, Octagam), being an obligate therapeutic component. When detecting herpesviruses (EBV, HSC-1/2, HHV6, or CMV) the therapy was accomplished by antiviral drugs, i.e., Acyclovir or Valacyclovir (for CMV treatment).
Diagnostics of septic conditions was based on clinical data (evidence of primary site and port of infection, septic syndrome, secondary sites of infection), and confirmed by the pathogen isolation in culture. During Grade IV neutropenia (< 0.5×109/L), clinical diagnosis of infectious conditions was suggested upon single increase of the body temperature (>38.0 оС), or repeated hyperthermia over 38.5 оС within 24 hours which could not be explained by clinical course of the primary disorder, or by the treatment performed. Systemic inflammatory reaction syndrome was based upon registration of, at least, 2 of 4 following clinical symptoms: [6].
– body temperature > 38 °С, or < 36 °С;
– heart rate > 90 per minute;
– breathing rate > 20 per minute, or hyperventilation (pСО2 < 32 mm Hg)
– peripheral leukocyte counts > 12×109/L, or < 4×109/L, or at > 10% immature forms.
In most cases, the blood specimens for bacteriological culture were taken 2, 3 or more times. The primary bacterial strain isolated from hemoculture was regarded as the etiological agent. Bacteriological analyses and identification of micromycetes were performed by uniform technique over the entire study period, according to the valid guidelines [15]. Micromycetes were morphologically identified in culture, by evaluating their growth patterns in agar cultures and fluid media. Enzyme activities and filamentation were used for detection of yeast-like microflora. Standard disc diffusion methods were applied to assess antibiotic sensitivity of isolated strains. E test was applied to determine minimal inhibiting concentration (MIC). The test kits were purchased from AB Biodisc (Sweden) [2].
DNA was extracted from peripheral blood leukocytes. For DNA-diagnostics, we used gene-specific PCR with real- time registration at ANK32 thermocycler. Commercial kits for multiplex PCR were from InterLabService (AmpliSens ®, Moscow, Russia) allowing to detect respiratory viruses (RSV, influenza А/В и А/H1N1sw types) parainfluenza virus (PIV), rhinovirus, adenovirus, coronavirus, metapneumovirus). The herpesvirus panel included Herpes Simplex type 1 and 2 (HSV); Cytomegalovirus (CMV); Epstein-Barr virus (EBV), and Human Herpesvirus type 6 (HHV6), as well as Clamydophila pneumoniae and Mycoplasma pneumoniae. PCR techniques were performed according to manufacturer instructions. The studies were performed at an. The declared analytical sensitivity for the test systems was 500…1000 copies/ mL for HSV1/2, and 5×105 per 105 leukocytes for EBV, CMV, and HHV type 6.
Statistical evaluation of the data was performed by means of STATISTICA 6.0 software, using χ2 and Student criteria. Correlation quotients and their significance were determined by Spearman criterion. The differences were considered significant at Р< 0.05.
Results
Our primary task was to evaluate frequencies of viral infections in the patients treated by intensive cytostatic therapy. Results of the virological study for Group 1 (herpesviruse and respiratory infections) are shown in Table 3.

RSV infection manifesting with fever was found in two patients (association with М.pneumonia in 1 case). Influenza А virus was detected in four patients exhibiting high fever (up to 39.0оС). Three cases of А/H1N1sw influenza were observed in 2009. A single patient with CLL treated with Fludarabine+Cyclophosphamide developed a mixed А/H1N1/rhinovirus infection. All the patients with influenza exhibited prolonged fever (< 38.6 оС) followed by pneumonia in one case of diffuse large-cell B lymphoma after R-CHOP. PIV 2 and 3 viruses were detectable in four patients accompanied by high fever (up to 38.5 ºС). In a patient with follicular lymphoma treated by R-CHOP regimen, an association of PIV 3 and bocavirus was found. PIV 3 infection was also registered in AML patient after the 7+3 chemotherapy and grade severe granulocytopenia followed by acute focal pneumonia. A patient with multiple myeloma after a course of Bortezomib/Dexamethasone developed positivity for PIV 3, with herpes labialis.
Rhinovirus DNA was most common in the studied group, with only mild clinical manifestations, i.e., subfebrile temperature. However, an acute unilateral pneumonia was registered in one ALL patient. An association of rhinovirus with 229Е coronavirus was found in one CLL patient. Coronaviruses were detected in 3 cases. The patients exhibited respiratory symptoms (cough, chills), subfebrile state. Metapneumovirus was shown in only 1 case of CLL, manifesting with subfebrility and respiratory symptoms.
The infections were accompanied by herpesvirus reactivation in 42% of the cases. HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. HHV6-specific DNA was not found in any sample from the patients. Antiviral therapy was initiated in patients with herpesvirus- positive blood samples.
Bacteriological study of the throat smears showed prevalence of normal Gram-positive microflora typical to oral microbiota. In 1 case, however, a patient with CLL had E.Coli bacteraemia, in absence of other evident local or systemic pathogens.
In comparison group (infection-free patients), infectious agents were found in 7/35 (20%) of the cases. I.e., rhinoviruses was revealed in 3 patients, coronaviruses, in 2 cases, and M. рneumoniae, in 2 patients. Anti-infectious treatment was not provided in these cases.
Blood stream infections group: etiological structure
Our second group included cases with proven sepsis primarily diagnosed by clinical criteria. Table 3 presents data on Gram-positivity and species composition of blood-borne microorganisms in patients with hemoblastoses observed from 1991 to 2013. In general, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, relative incidence of these microbial classes showed a clear predominance of Gram-negative bacteria over Gram-positives in 1993, 2004, and 2005. The percentage of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p < 0.05). Coagulase-negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria. Noteworthy, incidence of Pseudomonas spp. and Klebsiella spp. decreased over last decade.

A trend towards a higher incidence of Micromycetae among total microflora was also observed. E.g., their proportion in blood cultures was rather low in 2009, followed by increased detection of Micromycetae in 2010, 2011 and 2012 (respectively, 3.3, 9.7, and 5.3%).
Another task of our study was to specify a potential role of herpesviruses in bacteriaemia observed in leukemia patients. To test this hypothesis, we performed a comparative study of herpesvirus frequency in cases of proven bacterial infections in bloodstream. A comparison group consisted of leukemia patients free of detectable bacteremia. (Table 5). We have revealed a significant increased EBV and CMV incidence in blood of the patients who developed bacteremia, as compared with bacteriemia-free cases.

Possible combined effects of blood stream infections and viral reactivation upon clinical outcomes
We have analyzed distinct laboratory features of sepsis in the patients with resistant/relapsing hemoblastoses and lymphomas pre-treated with intensive chemotherapy (Tables 6 and 7).
As seen from Table 6, E.coli was the most common Gram-negative pathogen found in bloodstream (9 episodes). In one patient, E.сoli bacteremia was combined with EBV, while the second had CMV reactivation. Other bacteria were more rare (Pseudomonas aeruginosa – 1 case with EBV, HSV 1/2 and HHV6. In 2 cases, bacteremia was caused by Moraxella spp., all the cases were associated with herpesviruses (1, with CMV, and one, with a combination of CMV, HSV 1/2, HHV6, and EBV). Despite massive treatment with the most effective drugs, four patients with Gram-negative sepsis developed septic shock with multi-organ failure. Overall mortality in Gram-negative sepsis was 57% (8 cases).


Etiological role of Gram-positive microbes is more discutable. As seen from Table 7, CoNS were most common Gram-positive microbes isolated. A universal recommendation for detection of the microbes in blood specimens is to administer multiple blood sampling. In most cases, we used a more practical and quite efficient recommendation based on the terms of in vitro bacterial outgrowth, e.g., the test is likely to be positive if it is detected at short terms of culture (< 3…5 days). Late outgrowth suggests an artifactual microbial contamination [1]. Based on these criteria and clinical data, we have suggested a sufficient role of CoNS in systemic inflammatory response for eleven patients with different hematological tumors following intensive cytostatic chemotherapy.
Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: two cases were associated with EBV, and one, with CMV positivity. A combination of HHV6/EBV was found in one patient, thus potentially proving the immunocompromised condition in these cases. In four patients, we have found a constellation of three herpesvirus types, i.e., EBV, CMV, and HHV6. Therefore, a high ratio of viral and bacterial co-infections may be typical to a number of patients with proven sepsis after intensive cytostatic chemotherapy.
Discussion
Modern methods of leukemia treatment allow to sufficiently increase complete remission rates and to increase survival of the patients. The role of infectious complications following intensive leukemia therapy becomes increasingly high, due to improved diagnostics of infectious pathogens over last decade. Urgent diagnostics of these infections permits timely usage of specific therapy. Two decades ago, most pneumonia cases in such patients were classified as ‘unknown origin’ since full-scale PCR assays for viruses were not used, like as express diagnostics of bacterial cultures. Introduction of molecular biology testing allowed to extend opportunities of viral diagnostics in immunocompromised patients with leukemias and lymphoma [5].
We used real-time PCR approach covering the main set of respiratory pathogens. Application of this diagnostic set combined with bacteriological methods allowed to assess etiology of infectious processes in 62% of cases. In comparison group (leukemia patients without clinical signs of infections), the infectious agents were revealed in 7/35 (20%) of the patients. In general, our data are in accordance with results by G.Gerna et al.[7], who performed their studies by similar scenario in the patients receiving lung transplants.
Noteworthy, development of respiratory infections in the patients under study was accompanied by rather common incidence (42%) of herpesvirus detection, especially, CMV and EBV which are associated with immunosuppressive conditions. The data presented are confirming a significant role of viral pathogens, especially, herpesviruses, which are potential factors of immunosuppression and concomitant bacterial infections in leukemia patients. The data from second group of patients seem to confirm this finding. E.coli was commonly revealed in patients’ blood.
Moraxella catarrhalis was associated with septicemia in two cases, both accompanied by herpesvirus activation. Previously, M.catarrhalis was considered a causal factor for otitis media, sinusitis, and conjunctivitis in children. Later on, a role of this microorganism was suggested for septicemias in acute leukemia patients [8, 12]. Therefore, M.catarrhalis seems to play a distinct role in development of infectious complications in immunocompromised patients with oncohematological disorders [10, 14].
What concerns etiological significance of Gram-positive microorganisms in bacteremia, one should mind a high probability of skin contamination, e.g., with coagulase-negative staphylococci, thus increasing chances for false-positive results of hemocultures, due to the CoNS transfer during vein puncture. A possible role of CoNS in systemic inflammatory syndrome was assessed in 57.8% of hematological patients. Of them, 36% exhibited EBV, CMV, or HHV6 in blood leukocytes. Hence, the observed viral co-infection may be a biological marker of a generalized immune suppression. Nevertheless, one should bear in mind a probable independent role of herpesviruses in pathogenesis of infectious complications.
In the present study, antibacterial therapy started with β-lactame antibiotics combined with fluoroquinolones, aminoglycosides, metronidazole. If required, the antimicrobial strategy was revised 48 to 72 hours later as based on clinical and microbiological data, applying carbapenems also combined with other anti-infectious drugs. Nevertheless, mortality rates in Gram-negative and Gram-positive sepsis comprised, respectively, 57% and 11%. These patients suffered with severe primary malignancies, being mostly resistant to cytostatic treatment, with bulky disease and expressed immunosuppression due to drug-induced cytopenia.
One should mind a role of fungi in evolving infectious complications which may occur at any step of anticancer therapy, due to decreased innate immunity. Appropriate risk factor include neutropenia, damage to skin, intestinal mucosa, early administration of broad-spectrum antibiotics, glucocorticosteroids, immunosuppressive drugs that favor colonization with Candida spp. [9]. When treating these patients, we used Fluconazole, Voriconazole, or Caspofungin.
Conclusion
In summary, our data support a general viewpoint on regular monitoring of infectious pathogens upon intensive chemotherapy of oncohematological patients prone to both bacterial and viral infections. Severe infectious complications (pneumonia, sepsis) are often associated with fungal invasions, and herpesvirus reactivation.
In particular, our results suggest that different viruses, e.g., herpesviruses, may cause immunosuppression, or may serve as additional immunodeficiency markers predictive for bacterial infections at later terms. Most patients who developed severe infections (e.g., AML and NHL cases) are potential candidates for hematopoietic stem cell transplantation. Therefore, one should take into account their predisposition for infectious complications when planning HSCT for these patients.
Conflict of interests
No conflict of interests is declared.
References
- Souvenir D, Anderson DE Jr, Palpant S et al. Blood Cultures Positive for Coagulase-Negative Staphylococci: Antisepsis, Pseudobacteremia, and Therapy of Patients. J Clin Microbiol 1998; 36 (7): 1923–1926
- Baker CN, Stocker SA, Culver DH et al. Comparison of E-test to agar dilution broth microdilution and agar diffusion susceptibility testing techniques by using a special challenge set of bacteria. J Clin Microbiol 1991; 29: 533-538.
- Bessmeltsev SS, Abdulkadyrov KM. Multiple myeloma: The Physicians’ Guide. Moscow: MK Publishers, 2016, 504 p. (In Russian)
- Chebotkevich V, Kiseleva E, Stizhak N et al. Epidemiology and clinical characteristics of bloodstream infections in hematological cancer patients. Haematologica 2016; 101 (s1): 765.
- Chebotkevitch V, Volkov A. Community respiratory virus infections in patients with haematological malignancies // 17th ECCMID, March 31 – April 3, 2007. Munich, Germany. Ab. 589
- Dellinger RP, Levy MM, Rhodes A et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock. 2012 Intensive Care Med 2013; 39 (2): 165-228.
- Gerna G, Vitulo P, Rovida F, Lilleri D, Pellegrini C, Oggionni T, Campanini G, Baldanti F, Revello MG. Impact of human metapneumovirus and human cytomegalovirus versus other respiratory viruses on the lower respiratory tract infections of lung transplant recipients. J Med Virol 2006; 78 (3): 408-416.
- Henny FC, Mulder CJ, Lampe AS, van der Meer JW et al. Branhamella catarrhalis septicaemia in a granulocytopenic patient. Infection 1984; 12(3):208–209.
- Bhatt VR, Viola GM, Ferrajoli A. Invasive Fungal Infections in Acute Leukemia. Ther Adv Hematol. 2011; 2(4): 231–247.
- Leszczyсska K, Jakoniuk P, Sacha PT et al. Susceptibility of Branhamella catarrhalis to antibiotics. Med Dosw Mikrobiol 2004; 56: 231–237.
- Moiseev SI, Nuia ML, Chebotkevich VN, Gonchar VA, Abdulkadyrov KM. Cytomegalovirus infection in practice of bone marrow transplantation. Terapevticheskyi Archiv 2002; 74 (7): 44-48. (In Russian)
- Saito H, Anaissie EJ, Khardori N et al. Branhamella catarrhalis septicemia in patients with leukemia. Cancer 1988; 61 (11): 2315–2317.
- Girmenia C, Menichetti F. Current epidemiology and prevention of infectious complications in cancer patients. European Oncology and Haemotology 2011; 7 (4): 270-277.
- Sirwar SB, Indupalli AS, Pal R et al. Moraxella catarrhalis: an emerging pathogen in bronchopulmonary infections. Ann Trop Med Publ Health 2013; 6(1): 76–79.
- Versalovic J, Carroll KC, Funke G, Jorgensen JH, Landry ML, Warnock DW. (Eds.). Manual of Clinical Microbiology, 10th Edition, Vol. 1, 2011, ASM Press.
" ["DETAIL_TEXT_TYPE"]=> string(4) "html" ["~DETAIL_TEXT_TYPE"]=> string(4) "html" ["PREVIEW_TEXT"]=> string(0) "" ["~PREVIEW_TEXT"]=> string(0) "" ["PREVIEW_TEXT_TYPE"]=> string(4) "text" ["~PREVIEW_TEXT_TYPE"]=> string(4) "text" ["PREVIEW_PICTURE"]=> NULL ["~PREVIEW_PICTURE"]=> NULL ["LANG_DIR"]=> string(4) "/ru/" ["~LANG_DIR"]=> string(4) "/ru/" ["SORT"]=> string(3) "500" ["~SORT"]=> string(3) "500" ["CODE"]=> string(100) "infektsii-krovotoka-i-reaktivatsiya-virusov-posle-intensivnoy-khimioterapii-vzroslykh-bolnykh-onkoge" ["~CODE"]=> string(100) "infektsii-krovotoka-i-reaktivatsiya-virusov-posle-intensivnoy-khimioterapii-vzroslykh-bolnykh-onkoge" ["EXTERNAL_ID"]=> string(3) "673" ["~EXTERNAL_ID"]=> string(3) "673" ["IBLOCK_TYPE_ID"]=> string(7) "journal" ["~IBLOCK_TYPE_ID"]=> string(7) "journal" ["IBLOCK_CODE"]=> string(7) "volumes" ["~IBLOCK_CODE"]=> string(7) "volumes" ["IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["~IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["LID"]=> string(2) "s2" ["~LID"]=> string(2) "s2" ["EDIT_LINK"]=> NULL ["DELETE_LINK"]=> NULL ["DISPLAY_ACTIVE_FROM"]=> string(0) "" ["IPROPERTY_VALUES"]=> array(18) { ["ELEMENT_META_TITLE"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["ELEMENT_META_KEYWORDS"]=> string(143) "лейкозы лимфомы интенсивная химиотерапия сепсис виремия сочетанные инфекции" ["ELEMENT_META_DESCRIPTION"]=> string(343) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиляBloodstream infections and herpesvirus activation following intensive chemotherapy of adult oncohematological patients" ["ELEMENT_PREVIEW_PICTURE_FILE_ALT"]=> string(5167) "<p> Интенсивная цитостатическая химиотерапия является стандартной стратегией лечения лейкозов и лимфом. В то же время такое лечение вызывает негативные эффекты, в том числе лимфопению, гранулоцитопению и повреждение тканевых барьеров, что ассоциировано с существенными рисками инфекционных осложнений, особенно – бактериальным сепсисом и виремией. Целью нашей работы было выявление бактериемии и фунгемии у онкогематологических больных после интенсивной химиотерапии и оценка потенциальной модифицирующей роли герпесвирусных инфекций. </p> <p> Мы определяли частоту развития инфекционных осложнений и соответствующих этиологических факторов в 2 группах пациентов онкогематологического профиля, проходивших лечение в Российском институте гематологии и трансфузиологии, учитывая, в частности, случаи смешанных инфекций. Мазки из зева, венозная кровь, а также образцы мочи и мокроты отбирали для рутинных бактериологических и вирусологических исследований. Лейкоциты цельной крови тестировали на вирусы посредством ПЦР с применением стандартных протоколов. </p> <p> В первой группе пациентов (85 случаев) проводили исследования на респираторные вирусные инфекции. Вирусы гриппа и парагриппа, респираторно-синцитиальный вирус (РСВ), риновирус, аденовирус определялись в крови у единичных пациентов. В то же время герпесвирусы выявлялись, в целом, в 42% случаев. В частности, ДНК вирусов простого герпеса, Эпштейна-Барр (ВЭБ) и цитомегаловируса (ЦМВ) в лейкоцитах крови обнаружены в 5.2%, 26.3%, и 10.5%, соответственно. Вирусные инфекции в этой группе пациентов не были достоверно ассоциированы с положительными бактериологическими пробами. </p> <p> Отдельно анализировали результаты в группе из 33 случаев доказанного сепсиса, которые выявили среди 64 пациентов. В целом, частота выявления Грам-позитивных микроорганизмов преобладала над Грам-негативными культурами (69,2% и 30,8%, соответственно). Однако соотношение выявленной Грам-негативной флоры повысилось с 23% до 40% в период с 2002 по 2013 гг. (p < 0,05). Коагулазо-негативные стафилококки (КНС) преобладали среди Грам-позитивных микроорганизмов, в частности, S. epidermidis и S. aureus, тогда как Enterobacteriaceae, в особенности, E.coli, были наиболее частыми Грам-негативными бактериями. </p> <p> Представляет интерес, что у 4 из 11 пациентов с КНС-сепсисом отмечены сопутствующие герпесвирусные инфекции, а именно 2 случая были ассоциированы с ВЭБ; 1 – с ЦМВ и 1 – с герпесвирусом человека 6 типа и ВЭБ. Частая реактивация герпесвирусов может вызвать иммуносупрессию или быть дополнительным прогностическим признаком иммунодефицита в плане риска бактериальных инфекций в более поздние сроки. Поэтому в таких случаях следует учитывать предрасположенность к тяжелым инфекционным осложнениям при планировании трансплантацию гемопоэтических клеток этим больным. </p>" ["ELEMENT_PREVIEW_PICTURE_FILE_TITLE"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["ELEMENT_DETAIL_PICTURE_FILE_ALT"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["ELEMENT_DETAIL_PICTURE_FILE_TITLE"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["SECTION_META_TITLE"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["SECTION_META_KEYWORDS"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["SECTION_META_DESCRIPTION"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["SECTION_PICTURE_FILE_ALT"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["SECTION_PICTURE_FILE_TITLE"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["SECTION_PICTURE_FILE_NAME"]=> string(100) "infektsii-krovotoka-i-reaktivatsiya-virusov-posle-intensivnoy-khimioterapii-vzroslykh-bolnykh-onkoge" ["SECTION_DETAIL_PICTURE_FILE_ALT"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["SECTION_DETAIL_PICTURE_FILE_TITLE"]=> string(225) "Инфекции кровотока и реактивация вирусов после интенсивной химиотерапии взрослых больных онкогематологического профиля" ["SECTION_DETAIL_PICTURE_FILE_NAME"]=> string(100) "infektsii-krovotoka-i-reaktivatsiya-virusov-posle-intensivnoy-khimioterapii-vzroslykh-bolnykh-onkoge" ["ELEMENT_PREVIEW_PICTURE_FILE_NAME"]=> string(100) "infektsii-krovotoka-i-reaktivatsiya-virusov-posle-intensivnoy-khimioterapii-vzroslykh-bolnykh-onkoge" ["ELEMENT_DETAIL_PICTURE_FILE_NAME"]=> string(100) "infektsii-krovotoka-i-reaktivatsiya-virusov-posle-intensivnoy-khimioterapii-vzroslykh-bolnykh-onkoge" } ["FIELDS"]=> array(1) { ["IBLOCK_SECTION_ID"]=> string(2) "20" } ["PROPERTIES"]=> array(18) { ["KEYWORDS"]=> array(36) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(6) { [0]=> string(4) "8260" [1]=> string(4) "8261" [2]=> string(4) "8262" [3]=> string(4) "8263" [4]=> string(4) "8264" [5]=> string(4) "8265" } ["VALUE"]=> array(6) { [0]=> string(2) "94" [1]=> string(3) "344" [2]=> string(3) "669" [3]=> string(3) "670" [4]=> string(3) "671" [5]=> string(3) "672" } ["DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(6) { [0]=> string(2) "94" [1]=> string(3) "344" [2]=> string(3) "669" [3]=> string(3) "670" [4]=> string(3) "671" [5]=> string(3) "672" } ["~DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" } ["SUBMITTED"]=> array(36) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8205" ["VALUE"]=> string(22) "06/17/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "06/17/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL } ["ACCEPTED"]=> array(36) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8206" ["VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL } ["PUBLISHED"]=> array(36) { ["ID"]=> string(2) "22" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Дата публикации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "PUBLISHED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "22" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Дата публикации" ["~DEFAULT_VALUE"]=> NULL } ["CONTACT"]=> array(36) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8225" ["VALUE"]=> string(3) "663" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "663" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHORS"]=> array(36) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(6) { [0]=> string(4) "8266" [1]=> string(4) "8267" [2]=> string(4) "8268" [3]=> string(4) "8269" [4]=> string(4) "8270" [5]=> string(4) "8271" } ["VALUE"]=> array(6) { [0]=> string(3) "663" [1]=> string(3) "664" [2]=> string(3) "665" [3]=> string(3) "666" [4]=> string(3) "667" [5]=> string(3) "668" } ["DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(6) { [0]=> string(3) "663" [1]=> string(3) "664" [2]=> string(3) "665" [3]=> string(3) "666" [4]=> string(3) "667" [5]=> string(3) "668" } ["~DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_RU"]=> array(36) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8213" ["VALUE"]=> array(2) { ["TEXT"]=> string(233) "Виталий Н. Чеботкевич, Станислав С. Бессмельцев, Екатерина Е. Киселева, Наталья П. Стижак, Елена И. Кайтанджан, Виталий В. Бурылев" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(233) "Виталий Н. Чеботкевич, Станислав С. Бессмельцев, Екатерина Е. Киселева, Наталья П. Стижак, Елена И. Кайтанджан, Виталий В. Бурылев" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_RU"]=> array(36) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8214" ["VALUE"]=> array(2) { ["TEXT"]=> string(187) "Российский научно-исследовательский институт гематологии и трансфузиологии, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(187) "Российский научно-исследовательский институт гематологии и трансфузиологии, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_RU"]=> array(36) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8258" ["VALUE"]=> array(2) { ["TEXT"]=> string(5167) "<p> Интенсивная цитостатическая химиотерапия является стандартной стратегией лечения лейкозов и лимфом. В то же время такое лечение вызывает негативные эффекты, в том числе лимфопению, гранулоцитопению и повреждение тканевых барьеров, что ассоциировано с существенными рисками инфекционных осложнений, особенно – бактериальным сепсисом и виремией. Целью нашей работы было выявление бактериемии и фунгемии у онкогематологических больных после интенсивной химиотерапии и оценка потенциальной модифицирующей роли герпесвирусных инфекций. </p> <p> Мы определяли частоту развития инфекционных осложнений и соответствующих этиологических факторов в 2 группах пациентов онкогематологического профиля, проходивших лечение в Российском институте гематологии и трансфузиологии, учитывая, в частности, случаи смешанных инфекций. Мазки из зева, венозная кровь, а также образцы мочи и мокроты отбирали для рутинных бактериологических и вирусологических исследований. Лейкоциты цельной крови тестировали на вирусы посредством ПЦР с применением стандартных протоколов. </p> <p> В первой группе пациентов (85 случаев) проводили исследования на респираторные вирусные инфекции. Вирусы гриппа и парагриппа, респираторно-синцитиальный вирус (РСВ), риновирус, аденовирус определялись в крови у единичных пациентов. В то же время герпесвирусы выявлялись, в целом, в 42% случаев. В частности, ДНК вирусов простого герпеса, Эпштейна-Барр (ВЭБ) и цитомегаловируса (ЦМВ) в лейкоцитах крови обнаружены в 5.2%, 26.3%, и 10.5%, соответственно. Вирусные инфекции в этой группе пациентов не были достоверно ассоциированы с положительными бактериологическими пробами. </p> <p> Отдельно анализировали результаты в группе из 33 случаев доказанного сепсиса, которые выявили среди 64 пациентов. В целом, частота выявления Грам-позитивных микроорганизмов преобладала над Грам-негативными культурами (69,2% и 30,8%, соответственно). Однако соотношение выявленной Грам-негативной флоры повысилось с 23% до 40% в период с 2002 по 2013 гг. (p < 0,05). Коагулазо-негативные стафилококки (КНС) преобладали среди Грам-позитивных микроорганизмов, в частности, S. epidermidis и S. aureus, тогда как Enterobacteriaceae, в особенности, E.coli, были наиболее частыми Грам-негативными бактериями. </p> <p> Представляет интерес, что у 4 из 11 пациентов с КНС-сепсисом отмечены сопутствующие герпесвирусные инфекции, а именно 2 случая были ассоциированы с ВЭБ; 1 – с ЦМВ и 1 – с герпесвирусом человека 6 типа и ВЭБ. Частая реактивация герпесвирусов может вызвать иммуносупрессию или быть дополнительным прогностическим признаком иммунодефицита в плане риска бактериальных инфекций в более поздние сроки. Поэтому в таких случаях следует учитывать предрасположенность к тяжелым инфекционным осложнениям при планировании трансплантацию гемопоэтических клеток этим больным. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(5103) "
Интенсивная цитостатическая химиотерапия является стандартной стратегией лечения лейкозов и лимфом. В то же время такое лечение вызывает негативные эффекты, в том числе лимфопению, гранулоцитопению и повреждение тканевых барьеров, что ассоциировано с существенными рисками инфекционных осложнений, особенно – бактериальным сепсисом и виремией. Целью нашей работы было выявление бактериемии и фунгемии у онкогематологических больных после интенсивной химиотерапии и оценка потенциальной модифицирующей роли герпесвирусных инфекций.
Мы определяли частоту развития инфекционных осложнений и соответствующих этиологических факторов в 2 группах пациентов онкогематологического профиля, проходивших лечение в Российском институте гематологии и трансфузиологии, учитывая, в частности, случаи смешанных инфекций. Мазки из зева, венозная кровь, а также образцы мочи и мокроты отбирали для рутинных бактериологических и вирусологических исследований. Лейкоциты цельной крови тестировали на вирусы посредством ПЦР с применением стандартных протоколов.
В первой группе пациентов (85 случаев) проводили исследования на респираторные вирусные инфекции. Вирусы гриппа и парагриппа, респираторно-синцитиальный вирус (РСВ), риновирус, аденовирус определялись в крови у единичных пациентов. В то же время герпесвирусы выявлялись, в целом, в 42% случаев. В частности, ДНК вирусов простого герпеса, Эпштейна-Барр (ВЭБ) и цитомегаловируса (ЦМВ) в лейкоцитах крови обнаружены в 5.2%, 26.3%, и 10.5%, соответственно. Вирусные инфекции в этой группе пациентов не были достоверно ассоциированы с положительными бактериологическими пробами.
Отдельно анализировали результаты в группе из 33 случаев доказанного сепсиса, которые выявили среди 64 пациентов. В целом, частота выявления Грам-позитивных микроорганизмов преобладала над Грам-негативными культурами (69,2% и 30,8%, соответственно). Однако соотношение выявленной Грам-негативной флоры повысилось с 23% до 40% в период с 2002 по 2013 гг. (p < 0,05). Коагулазо-негативные стафилококки (КНС) преобладали среди Грам-позитивных микроорганизмов, в частности, S. epidermidis и S. aureus, тогда как Enterobacteriaceae, в особенности, E.coli, были наиболее частыми Грам-негативными бактериями.
Представляет интерес, что у 4 из 11 пациентов с КНС-сепсисом отмечены сопутствующие герпесвирусные инфекции, а именно 2 случая были ассоциированы с ВЭБ; 1 – с ЦМВ и 1 – с герпесвирусом человека 6 типа и ВЭБ. Частая реактивация герпесвирусов может вызвать иммуносупрессию или быть дополнительным прогностическим признаком иммунодефицита в плане риска бактериальных инфекций в более поздние сроки. Поэтому в таких случаях следует учитывать предрасположенность к тяжелым инфекционным осложнениям при планировании трансплантацию гемопоэтических клеток этим больным.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["DOI"]=> array(36) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8215" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-21-31" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-21-31" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_EN"]=> array(36) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8216" ["VALUE"]=> array(2) { ["TEXT"]=> string(132) "Vitaly N. Chebotkevich, Stanislav S. Bessmeltsev, Ekaterina E. Kiseleva, Natalya P. Stizhak, Elena I. Kaytandzhan, Vitaly V. Burylev" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(132) "Vitaly N. Chebotkevich, Stanislav S. Bessmeltsev, Ekaterina E. Kiseleva, Natalya P. Stizhak, Elena I. Kaytandzhan, Vitaly V. Burylev" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_EN"]=> array(36) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8217" ["VALUE"]=> array(2) { ["TEXT"]=> string(507) "Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St.16, 191024 St. Petersburg, Russia.<br> <br> Professor Vitaly N. Chebotkevich, Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St. 16, 191024 St. Petersburg, Russia.<br> <br> Phone: +7 (812) 717-29-58<br> Fax: +7 (812) 717-25-50<br> E-mail: <a href="mailto:vitnikcheb@mail.ru">vitnikcheb@mail.ru</a>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(449) "Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St.16, 191024 St. Petersburg, Russia.Professor Vitaly N. Chebotkevich, Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St. 16, 191024 St. Petersburg, Russia.
Phone: +7 (812) 717-29-58
Fax: +7 (812) 717-25-50
E-mail: vitnikcheb@mail.ru" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_EN"]=> array(36) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8259" ["VALUE"]=> array(2) { ["TEXT"]=> string(2746) "<p> Intensive cytostatic chemotherapy is a standard strategy for leukemia treatment. Meanwhile, such treatment causes negative effects, i.e., including lymphopenia, granulocytopenia and damage to tissue barriers associated with significant risks of infectious complications, especially, bacterial sepsis and viremia. Our study was aimed for identification of bacteremia and fungemia in oncohematological patients following intensive chemotherapy, and assessment of potential modifying role of herpesvirus infections. </p> <p> We assessed frequency of infectious complications and their etiological agents in two groups of oncohematological patients treated at the Russian Institute of Hematology and Transfusion, especially focusing on mixed infections. Throat smears, venous blood, as well as urine and sputum specimens were taken for routine bacteriological cultures. Whole blood leukocytes were virologically tested by PCR, using standard examination protocol. </p> <p> Our first virological study was performed for respiratory infections and included 85 randomly chosen patients. Influenza and parainfluenza viruses, respiratory syncytial virus, rhinovirus, adenovirus were detected in blood of single patients. Meanwhile, herpesviruses were detectable in 42% proportion of cases, i.e., HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. Viral infections were not associated with positive bacteriological findings in this group. Our group of 33 clinical cases with proven sepsis observed among 64 patients. Generally, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, the ratio of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p<0.05). Coagulase- negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria. </p> <p> Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: 2 cases were associated with EBV; 1, with CMV, and 1, with HHV6/EBV coinfection. Therefore, a high ratio of viral and bacterial co-infections may be revealed in a number of patients with proven sepsis. Moreover, common reactivation of herpesviruses may cause immunosuppression, or represent additional immunodeficiency markers predictive for bacterial infections at later terms. Therefore, one should take into account their predisposal for severe infectious complications when planning hematopoietic stem cell transplantation (HSCT) for these patients. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2694) "
Intensive cytostatic chemotherapy is a standard strategy for leukemia treatment. Meanwhile, such treatment causes negative effects, i.e., including lymphopenia, granulocytopenia and damage to tissue barriers associated with significant risks of infectious complications, especially, bacterial sepsis and viremia. Our study was aimed for identification of bacteremia and fungemia in oncohematological patients following intensive chemotherapy, and assessment of potential modifying role of herpesvirus infections.
We assessed frequency of infectious complications and their etiological agents in two groups of oncohematological patients treated at the Russian Institute of Hematology and Transfusion, especially focusing on mixed infections. Throat smears, venous blood, as well as urine and sputum specimens were taken for routine bacteriological cultures. Whole blood leukocytes were virologically tested by PCR, using standard examination protocol.
Our first virological study was performed for respiratory infections and included 85 randomly chosen patients. Influenza and parainfluenza viruses, respiratory syncytial virus, rhinovirus, adenovirus were detected in blood of single patients. Meanwhile, herpesviruses were detectable in 42% proportion of cases, i.e., HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. Viral infections were not associated with positive bacteriological findings in this group. Our group of 33 clinical cases with proven sepsis observed among 64 patients. Generally, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, the ratio of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p<0.05). Coagulase- negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria.
Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: 2 cases were associated with EBV; 1, with CMV, and 1, with HHV6/EBV coinfection. Therefore, a high ratio of viral and bacterial co-infections may be revealed in a number of patients with proven sepsis. Moreover, common reactivation of herpesviruses may cause immunosuppression, or represent additional immunodeficiency markers predictive for bacterial infections at later terms. Therefore, one should take into account their predisposal for severe infectious complications when planning hematopoietic stem cell transplantation (HSCT) for these patients.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["NAME_EN"]=> array(36) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8218" ["VALUE"]=> string(118) "Bloodstream infections and herpesvirus activation following intensive chemotherapy of adult oncohematological patients" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(118) "Bloodstream infections and herpesvirus activation following intensive chemotherapy of adult oncohematological patients" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" } ["FULL_TEXT_RU"]=> array(36) { ["ID"]=> string(2) "42" ["TIMESTAMP_X"]=> string(19) "2015-09-07 20:29:18" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(23) "Полный текст" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(12) "FULL_TEXT_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "42" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(23) "Полный текст" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["PDF_RU"]=> array(36) { ["ID"]=> string(2) "43" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF RUS" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_RU" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "43" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8232" ["VALUE"]=> string(3) "248" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "248" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF RUS" ["~DEFAULT_VALUE"]=> string(0) "" } ["PDF_EN"]=> array(36) { ["ID"]=> string(2) "44" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF ENG" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "44" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8233" ["VALUE"]=> string(3) "249" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "249" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF ENG" ["~DEFAULT_VALUE"]=> string(0) "" } ["NAME_LONG"]=> array(36) { ["ID"]=> string(2) "45" ["TIMESTAMP_X"]=> string(19) "2023-04-13 00:55:00" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(72) "Название (для очень длинных заголовков)" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "NAME_LONG" ["DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "45" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(80) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(72) "Название (для очень длинных заголовков)" ["~DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } } } ["DISPLAY_PROPERTIES"]=> array(13) { ["AUTHOR_EN"]=> array(37) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8216" ["VALUE"]=> array(2) { ["TEXT"]=> string(132) "Vitaly N. Chebotkevich, Stanislav S. Bessmeltsev, Ekaterina E. Kiseleva, Natalya P. Stizhak, Elena I. Kaytandzhan, Vitaly V. Burylev" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(132) "Vitaly N. Chebotkevich, Stanislav S. Bessmeltsev, Ekaterina E. Kiseleva, Natalya P. Stizhak, Elena I. Kaytandzhan, Vitaly V. Burylev" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(132) "Vitaly N. Chebotkevich, Stanislav S. Bessmeltsev, Ekaterina E. Kiseleva, Natalya P. Stizhak, Elena I. Kaytandzhan, Vitaly V. Burylev" } ["SUMMARY_EN"]=> array(37) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8259" ["VALUE"]=> array(2) { ["TEXT"]=> string(2746) "<p> Intensive cytostatic chemotherapy is a standard strategy for leukemia treatment. Meanwhile, such treatment causes negative effects, i.e., including lymphopenia, granulocytopenia and damage to tissue barriers associated with significant risks of infectious complications, especially, bacterial sepsis and viremia. Our study was aimed for identification of bacteremia and fungemia in oncohematological patients following intensive chemotherapy, and assessment of potential modifying role of herpesvirus infections. </p> <p> We assessed frequency of infectious complications and their etiological agents in two groups of oncohematological patients treated at the Russian Institute of Hematology and Transfusion, especially focusing on mixed infections. Throat smears, venous blood, as well as urine and sputum specimens were taken for routine bacteriological cultures. Whole blood leukocytes were virologically tested by PCR, using standard examination protocol. </p> <p> Our first virological study was performed for respiratory infections and included 85 randomly chosen patients. Influenza and parainfluenza viruses, respiratory syncytial virus, rhinovirus, adenovirus were detected in blood of single patients. Meanwhile, herpesviruses were detectable in 42% proportion of cases, i.e., HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. Viral infections were not associated with positive bacteriological findings in this group. Our group of 33 clinical cases with proven sepsis observed among 64 patients. Generally, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, the ratio of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p<0.05). Coagulase- negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria. </p> <p> Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: 2 cases were associated with EBV; 1, with CMV, and 1, with HHV6/EBV coinfection. Therefore, a high ratio of viral and bacterial co-infections may be revealed in a number of patients with proven sepsis. Moreover, common reactivation of herpesviruses may cause immunosuppression, or represent additional immunodeficiency markers predictive for bacterial infections at later terms. Therefore, one should take into account their predisposal for severe infectious complications when planning hematopoietic stem cell transplantation (HSCT) for these patients. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2694) "Intensive cytostatic chemotherapy is a standard strategy for leukemia treatment. Meanwhile, such treatment causes negative effects, i.e., including lymphopenia, granulocytopenia and damage to tissue barriers associated with significant risks of infectious complications, especially, bacterial sepsis and viremia. Our study was aimed for identification of bacteremia and fungemia in oncohematological patients following intensive chemotherapy, and assessment of potential modifying role of herpesvirus infections.
We assessed frequency of infectious complications and their etiological agents in two groups of oncohematological patients treated at the Russian Institute of Hematology and Transfusion, especially focusing on mixed infections. Throat smears, venous blood, as well as urine and sputum specimens were taken for routine bacteriological cultures. Whole blood leukocytes were virologically tested by PCR, using standard examination protocol.
Our first virological study was performed for respiratory infections and included 85 randomly chosen patients. Influenza and parainfluenza viruses, respiratory syncytial virus, rhinovirus, adenovirus were detected in blood of single patients. Meanwhile, herpesviruses were detectable in 42% proportion of cases, i.e., HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. Viral infections were not associated with positive bacteriological findings in this group. Our group of 33 clinical cases with proven sepsis observed among 64 patients. Generally, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, the ratio of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p<0.05). Coagulase- negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria.
Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: 2 cases were associated with EBV; 1, with CMV, and 1, with HHV6/EBV coinfection. Therefore, a high ratio of viral and bacterial co-infections may be revealed in a number of patients with proven sepsis. Moreover, common reactivation of herpesviruses may cause immunosuppression, or represent additional immunodeficiency markers predictive for bacterial infections at later terms. Therefore, one should take into account their predisposal for severe infectious complications when planning hematopoietic stem cell transplantation (HSCT) for these patients.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(2694) "Intensive cytostatic chemotherapy is a standard strategy for leukemia treatment. Meanwhile, such treatment causes negative effects, i.e., including lymphopenia, granulocytopenia and damage to tissue barriers associated with significant risks of infectious complications, especially, bacterial sepsis and viremia. Our study was aimed for identification of bacteremia and fungemia in oncohematological patients following intensive chemotherapy, and assessment of potential modifying role of herpesvirus infections.
We assessed frequency of infectious complications and their etiological agents in two groups of oncohematological patients treated at the Russian Institute of Hematology and Transfusion, especially focusing on mixed infections. Throat smears, venous blood, as well as urine and sputum specimens were taken for routine bacteriological cultures. Whole blood leukocytes were virologically tested by PCR, using standard examination protocol.
Our first virological study was performed for respiratory infections and included 85 randomly chosen patients. Influenza and parainfluenza viruses, respiratory syncytial virus, rhinovirus, adenovirus were detected in blood of single patients. Meanwhile, herpesviruses were detectable in 42% proportion of cases, i.e., HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. Viral infections were not associated with positive bacteriological findings in this group. Our group of 33 clinical cases with proven sepsis observed among 64 patients. Generally, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, the ratio of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p<0.05). Coagulase- negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria.
Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: 2 cases were associated with EBV; 1, with CMV, and 1, with HHV6/EBV coinfection. Therefore, a high ratio of viral and bacterial co-infections may be revealed in a number of patients with proven sepsis. Moreover, common reactivation of herpesviruses may cause immunosuppression, or represent additional immunodeficiency markers predictive for bacterial infections at later terms. Therefore, one should take into account their predisposal for severe infectious complications when planning hematopoietic stem cell transplantation (HSCT) for these patients.
" } ["DOI"]=> array(37) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8215" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-21-31" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-21-31" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-21-31" } ["NAME_EN"]=> array(37) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8218" ["VALUE"]=> string(118) "Bloodstream infections and herpesvirus activation following intensive chemotherapy of adult oncohematological patients" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(118) "Bloodstream infections and herpesvirus activation following intensive chemotherapy of adult oncohematological patients" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(118) "Bloodstream infections and herpesvirus activation following intensive chemotherapy of adult oncohematological patients" } ["ORGANIZATION_EN"]=> array(37) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8217" ["VALUE"]=> array(2) { ["TEXT"]=> string(507) "Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St.16, 191024 St. Petersburg, Russia.<br> <br> Professor Vitaly N. Chebotkevich, Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St. 16, 191024 St. Petersburg, Russia.<br> <br> Phone: +7 (812) 717-29-58<br> Fax: +7 (812) 717-25-50<br> E-mail: <a href="mailto:vitnikcheb@mail.ru">vitnikcheb@mail.ru</a>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(449) "Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St.16, 191024 St. Petersburg, Russia.Professor Vitaly N. Chebotkevich, Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St. 16, 191024 St. Petersburg, Russia.
Phone: +7 (812) 717-29-58
Fax: +7 (812) 717-25-50
E-mail: vitnikcheb@mail.ru" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(449) "Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St.16, 191024 St. Petersburg, Russia.
Professor Vitaly N. Chebotkevich, Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St. 16, 191024 St. Petersburg, Russia.
Phone: +7 (812) 717-29-58
Fax: +7 (812) 717-25-50
E-mail: vitnikcheb@mail.ru" } ["AUTHORS"]=> array(38) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(6) { [0]=> string(4) "8266" [1]=> string(4) "8267" [2]=> string(4) "8268" [3]=> string(4) "8269" [4]=> string(4) "8270" [5]=> string(4) "8271" } ["VALUE"]=> array(6) { [0]=> string(3) "663" [1]=> string(3) "664" [2]=> string(3) "665" [3]=> string(3) "666" [4]=> string(3) "667" [5]=> string(3) "668" } ["DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(6) { [0]=> string(3) "663" [1]=> string(3) "664" [2]=> string(3) "665" [3]=> string(3) "666" [4]=> string(3) "667" [5]=> string(3) "668" } ["~DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(6) { [0]=> string(65) "Vitaly N. Chebotkevich" [1]=> string(67) "Stanislav S. Bessmeltsev" [2]=> string(64) "Ekaterina E. Kiseleva" [3]=> string(61) "Natalya P. Stizhak" [4]=> string(63) "Elena I. Kaytandzhan" [5]=> string(60) "Vitaly V. Burylev" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["AUTHOR_RU"]=> array(37) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8213" ["VALUE"]=> array(2) { ["TEXT"]=> string(233) "Виталий Н. Чеботкевич, Станислав С. Бессмельцев, Екатерина Е. Киселева, Наталья П. Стижак, Елена И. Кайтанджан, Виталий В. Бурылев" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(233) "Виталий Н. Чеботкевич, Станислав С. Бессмельцев, Екатерина Е. Киселева, Наталья П. Стижак, Елена И. Кайтанджан, Виталий В. Бурылев" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(233) "Виталий Н. Чеботкевич, Станислав С. Бессмельцев, Екатерина Е. Киселева, Наталья П. Стижак, Елена И. Кайтанджан, Виталий В. Бурылев" } ["SUBMITTED"]=> array(37) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8205" ["VALUE"]=> string(22) "06/17/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "06/17/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "06/17/2016 12:00:00 am" } ["ACCEPTED"]=> array(37) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8206" ["VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "11/25/2016 12:00:00 am" } ["KEYWORDS"]=> array(38) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(6) { [0]=> string(4) "8260" [1]=> string(4) "8261" [2]=> string(4) "8262" [3]=> string(4) "8263" [4]=> string(4) "8264" [5]=> string(4) "8265" } ["VALUE"]=> array(6) { [0]=> string(2) "94" [1]=> string(3) "344" [2]=> string(3) "669" [3]=> string(3) "670" [4]=> string(3) "671" [5]=> string(3) "672" } ["DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(6) { [0]=> string(2) "94" [1]=> string(3) "344" [2]=> string(3) "669" [3]=> string(3) "670" [4]=> string(3) "671" [5]=> string(3) "672" } ["~DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(6) { [0]=> string(57) "лейкозы" [1]=> string(58) "лимфомы" [2]=> string(91) "интенсивная химиотерапия" [3]=> string(56) "сепсис" [4]=> string(58) "виремия" [5]=> string(81) "сочетанные инфекции" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["CONTACT"]=> array(38) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8225" ["VALUE"]=> string(3) "663" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "663" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(65) "Vitaly N. Chebotkevich" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["SUMMARY_RU"]=> array(37) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8258" ["VALUE"]=> array(2) { ["TEXT"]=> string(5167) "<p> Интенсивная цитостатическая химиотерапия является стандартной стратегией лечения лейкозов и лимфом. В то же время такое лечение вызывает негативные эффекты, в том числе лимфопению, гранулоцитопению и повреждение тканевых барьеров, что ассоциировано с существенными рисками инфекционных осложнений, особенно – бактериальным сепсисом и виремией. Целью нашей работы было выявление бактериемии и фунгемии у онкогематологических больных после интенсивной химиотерапии и оценка потенциальной модифицирующей роли герпесвирусных инфекций. </p> <p> Мы определяли частоту развития инфекционных осложнений и соответствующих этиологических факторов в 2 группах пациентов онкогематологического профиля, проходивших лечение в Российском институте гематологии и трансфузиологии, учитывая, в частности, случаи смешанных инфекций. Мазки из зева, венозная кровь, а также образцы мочи и мокроты отбирали для рутинных бактериологических и вирусологических исследований. Лейкоциты цельной крови тестировали на вирусы посредством ПЦР с применением стандартных протоколов. </p> <p> В первой группе пациентов (85 случаев) проводили исследования на респираторные вирусные инфекции. Вирусы гриппа и парагриппа, респираторно-синцитиальный вирус (РСВ), риновирус, аденовирус определялись в крови у единичных пациентов. В то же время герпесвирусы выявлялись, в целом, в 42% случаев. В частности, ДНК вирусов простого герпеса, Эпштейна-Барр (ВЭБ) и цитомегаловируса (ЦМВ) в лейкоцитах крови обнаружены в 5.2%, 26.3%, и 10.5%, соответственно. Вирусные инфекции в этой группе пациентов не были достоверно ассоциированы с положительными бактериологическими пробами. </p> <p> Отдельно анализировали результаты в группе из 33 случаев доказанного сепсиса, которые выявили среди 64 пациентов. В целом, частота выявления Грам-позитивных микроорганизмов преобладала над Грам-негативными культурами (69,2% и 30,8%, соответственно). Однако соотношение выявленной Грам-негативной флоры повысилось с 23% до 40% в период с 2002 по 2013 гг. (p < 0,05). Коагулазо-негативные стафилококки (КНС) преобладали среди Грам-позитивных микроорганизмов, в частности, S. epidermidis и S. aureus, тогда как Enterobacteriaceae, в особенности, E.coli, были наиболее частыми Грам-негативными бактериями. </p> <p> Представляет интерес, что у 4 из 11 пациентов с КНС-сепсисом отмечены сопутствующие герпесвирусные инфекции, а именно 2 случая были ассоциированы с ВЭБ; 1 – с ЦМВ и 1 – с герпесвирусом человека 6 типа и ВЭБ. Частая реактивация герпесвирусов может вызвать иммуносупрессию или быть дополнительным прогностическим признаком иммунодефицита в плане риска бактериальных инфекций в более поздние сроки. Поэтому в таких случаях следует учитывать предрасположенность к тяжелым инфекционным осложнениям при планировании трансплантацию гемопоэтических клеток этим больным. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(5103) "
Интенсивная цитостатическая химиотерапия является стандартной стратегией лечения лейкозов и лимфом. В то же время такое лечение вызывает негативные эффекты, в том числе лимфопению, гранулоцитопению и повреждение тканевых барьеров, что ассоциировано с существенными рисками инфекционных осложнений, особенно – бактериальным сепсисом и виремией. Целью нашей работы было выявление бактериемии и фунгемии у онкогематологических больных после интенсивной химиотерапии и оценка потенциальной модифицирующей роли герпесвирусных инфекций.
Мы определяли частоту развития инфекционных осложнений и соответствующих этиологических факторов в 2 группах пациентов онкогематологического профиля, проходивших лечение в Российском институте гематологии и трансфузиологии, учитывая, в частности, случаи смешанных инфекций. Мазки из зева, венозная кровь, а также образцы мочи и мокроты отбирали для рутинных бактериологических и вирусологических исследований. Лейкоциты цельной крови тестировали на вирусы посредством ПЦР с применением стандартных протоколов.
В первой группе пациентов (85 случаев) проводили исследования на респираторные вирусные инфекции. Вирусы гриппа и парагриппа, респираторно-синцитиальный вирус (РСВ), риновирус, аденовирус определялись в крови у единичных пациентов. В то же время герпесвирусы выявлялись, в целом, в 42% случаев. В частности, ДНК вирусов простого герпеса, Эпштейна-Барр (ВЭБ) и цитомегаловируса (ЦМВ) в лейкоцитах крови обнаружены в 5.2%, 26.3%, и 10.5%, соответственно. Вирусные инфекции в этой группе пациентов не были достоверно ассоциированы с положительными бактериологическими пробами.
Отдельно анализировали результаты в группе из 33 случаев доказанного сепсиса, которые выявили среди 64 пациентов. В целом, частота выявления Грам-позитивных микроорганизмов преобладала над Грам-негативными культурами (69,2% и 30,8%, соответственно). Однако соотношение выявленной Грам-негативной флоры повысилось с 23% до 40% в период с 2002 по 2013 гг. (p < 0,05). Коагулазо-негативные стафилококки (КНС) преобладали среди Грам-позитивных микроорганизмов, в частности, S. epidermidis и S. aureus, тогда как Enterobacteriaceae, в особенности, E.coli, были наиболее частыми Грам-негативными бактериями.
Представляет интерес, что у 4 из 11 пациентов с КНС-сепсисом отмечены сопутствующие герпесвирусные инфекции, а именно 2 случая были ассоциированы с ВЭБ; 1 – с ЦМВ и 1 – с герпесвирусом человека 6 типа и ВЭБ. Частая реактивация герпесвирусов может вызвать иммуносупрессию или быть дополнительным прогностическим признаком иммунодефицита в плане риска бактериальных инфекций в более поздние сроки. Поэтому в таких случаях следует учитывать предрасположенность к тяжелым инфекционным осложнениям при планировании трансплантацию гемопоэтических клеток этим больным.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(5103) "Интенсивная цитостатическая химиотерапия является стандартной стратегией лечения лейкозов и лимфом. В то же время такое лечение вызывает негативные эффекты, в том числе лимфопению, гранулоцитопению и повреждение тканевых барьеров, что ассоциировано с существенными рисками инфекционных осложнений, особенно – бактериальным сепсисом и виремией. Целью нашей работы было выявление бактериемии и фунгемии у онкогематологических больных после интенсивной химиотерапии и оценка потенциальной модифицирующей роли герпесвирусных инфекций.
Мы определяли частоту развития инфекционных осложнений и соответствующих этиологических факторов в 2 группах пациентов онкогематологического профиля, проходивших лечение в Российском институте гематологии и трансфузиологии, учитывая, в частности, случаи смешанных инфекций. Мазки из зева, венозная кровь, а также образцы мочи и мокроты отбирали для рутинных бактериологических и вирусологических исследований. Лейкоциты цельной крови тестировали на вирусы посредством ПЦР с применением стандартных протоколов.
В первой группе пациентов (85 случаев) проводили исследования на респираторные вирусные инфекции. Вирусы гриппа и парагриппа, респираторно-синцитиальный вирус (РСВ), риновирус, аденовирус определялись в крови у единичных пациентов. В то же время герпесвирусы выявлялись, в целом, в 42% случаев. В частности, ДНК вирусов простого герпеса, Эпштейна-Барр (ВЭБ) и цитомегаловируса (ЦМВ) в лейкоцитах крови обнаружены в 5.2%, 26.3%, и 10.5%, соответственно. Вирусные инфекции в этой группе пациентов не были достоверно ассоциированы с положительными бактериологическими пробами.
Отдельно анализировали результаты в группе из 33 случаев доказанного сепсиса, которые выявили среди 64 пациентов. В целом, частота выявления Грам-позитивных микроорганизмов преобладала над Грам-негативными культурами (69,2% и 30,8%, соответственно). Однако соотношение выявленной Грам-негативной флоры повысилось с 23% до 40% в период с 2002 по 2013 гг. (p < 0,05). Коагулазо-негативные стафилококки (КНС) преобладали среди Грам-позитивных микроорганизмов, в частности, S. epidermidis и S. aureus, тогда как Enterobacteriaceae, в особенности, E.coli, были наиболее частыми Грам-негативными бактериями.
Представляет интерес, что у 4 из 11 пациентов с КНС-сепсисом отмечены сопутствующие герпесвирусные инфекции, а именно 2 случая были ассоциированы с ВЭБ; 1 – с ЦМВ и 1 – с герпесвирусом человека 6 типа и ВЭБ. Частая реактивация герпесвирусов может вызвать иммуносупрессию или быть дополнительным прогностическим признаком иммунодефицита в плане риска бактериальных инфекций в более поздние сроки. Поэтому в таких случаях следует учитывать предрасположенность к тяжелым инфекционным осложнениям при планировании трансплантацию гемопоэтических клеток этим больным.
" } ["ORGANIZATION_RU"]=> array(37) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8214" ["VALUE"]=> array(2) { ["TEXT"]=> string(187) "Российский научно-исследовательский институт гематологии и трансфузиологии, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(187) "Российский научно-исследовательский институт гематологии и трансфузиологии, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(187) "Российский научно-исследовательский институт гематологии и трансфузиологии, Санкт-Петербург, Россия" } } } [1]=> array(49) { ["IBLOCK_SECTION_ID"]=> string(2) "20" ["~IBLOCK_SECTION_ID"]=> string(2) "20" ["ID"]=> string(3) "662" ["~ID"]=> string(3) "662" ["IBLOCK_ID"]=> string(1) "2" ["~IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(216) "Современные вопросы аллогенной трансплантации гемопоэтических стволовых клеток и солидных органов тому же пациенту" ["~NAME"]=> string(216) "Современные вопросы аллогенной трансплантации гемопоэтических стволовых клеток и солидных органов тому же пациенту" ["ACTIVE_FROM"]=> NULL ["~ACTIVE_FROM"]=> NULL ["TIMESTAMP_X"]=> string(22) "03/29/2017 04:31:28 pm" ["~TIMESTAMP_X"]=> string(22) "03/29/2017 04:31:28 pm" ["DETAIL_PAGE_URL"]=> string(150) "/en/archive/vypusk-5-nomer-4/klinicheskie-raboty/sovremennye-voprosy-allogennoy-transplantatsii-gemopoeticheskikh-stvolovykh-kletok-i-solidnykh-organ/" ["~DETAIL_PAGE_URL"]=> string(150) "/en/archive/vypusk-5-nomer-4/klinicheskie-raboty/sovremennye-voprosy-allogennoy-transplantatsii-gemopoeticheskikh-stvolovykh-kletok-i-solidnykh-organ/" ["LIST_PAGE_URL"]=> string(12) "/en/archive/" ["~LIST_PAGE_URL"]=> string(12) "/en/archive/" ["DETAIL_TEXT"]=> string(25310) "Introduction
Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. About 32000 SOTs were performed in the European Union in 2014, comprising approximately 20000 kidney, 7400 liver, 2100 heart, 1800 lung and 900 other transplants [6]. Allogeneic hematopoietic stem cell transplantation (HSCT) is the only cure or the most effective treatment for a large number of malignant and non-malignant diseases of the lympho-hematopoietic system with an otherwise poor prognosis. At present, the number of allogeneic HSCTs in Europe is more than 15000 per year [17]. With these large numbers of patients, the likelihood of coincidental occurrence of disorders potentially indicating SOT and HSCT in the same patient is increasing. Such transplantation combinations can be problematic for many reasons.
Allogeneic HSCT is an intensive treatment which frequently causes organ injury and failure, resulting in a potential indication for a SOT. The mechanisms of organ injury include cytotoxic drugs and radiotherapy in the pre-transplant conditioning, organ-toxic agents in supportive care, and immunological mechanisms. The organs most often affected are kidney, liver, lung and heart. Factors that may complicate a SOT in this setting are the fragility of the patient as a consequence of the toxicity of HSCT, immunosuppressive treatment for the HSCT which may not be optimal for the SOT, potential immunological problems caused by tissue type differences between the recipient, the hematopoietic graft and the solid organ graft, and infections. In SOT recipients, the long-lasting immunosuppressive medication may affect the function of organs, and increase the risk of infections. SOT can be associated with the development of hematological disorders comprising aplastic anemia, post-transplant lymphoproliferative disease, acute myeloid leukemia and myelodysplastic syndromes which can form a potential indication for allogeneic HSCT.
As the clinical situation is often found to be complicated and the risk of treatment failure regarded as high, the numbers of SOTs to HSCT recipients and those of HSCTs to SOT recipients have been low as compared to the number of patients who would have a potential indication for such treatment. Until recent years the literature of such transplantations consisted mainly of case reports as reviewed elsewhere [1,3,13], with the obvious risk of publication bias. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients.
Experience with organ transplantation in hematopoietic stem cell transplant recipients
There is a rather limited published experience available for the assessment of the indications for SOT in HSCT recipients. Koenecke et al [13] carried out a survey among 107 EBMT (European Society for Blood and Marrow Transplantation) centres, with 67000 allogeneic transplantations performed during the study period of 1984-2007. In 28 centres a total of 45 SOTs had been carried out in 40 patients who had previously undergone allogeneic HSCT, of them 15 renal, 15 liver, 13 lung, 1 heart and 1 skin transplantation. Hence, the approximate frequency of these interventions was 67 SOTs per 100 000 HSCTs. Moreover, seven additional SOTs were identified from case reports or from centres that decided not to participate, altogether bringing the total number to 52 SOTs. The median age of the patients was between 20 and 35 years, depending of the organ graft type. The liver transplant patients were the youngest ones. Most of the patients had leukaemia, but about 15% had aplastic anaemia and another 15% an inherited disorder. A large majority of the patients received myeloablative conditioning. The graft had been bone marrow in 60%, and the donor was a matched related donor in 54% of the cases. Approximately half of the patients developed acute and/or chronic graft-versus-host disease. The median time from the HSCT to the organ transplantation ranged from 34 (liver transplants) to 84 (kidney transplants) months.
In kidney transplantations, the most frequent cause of renal failure was drug treatment or radiotherapy. All patients were on dialysis at the time of the SOT. The donor was unrelated in four cases and related in nine cases; of the latter four were the same as in the blood stem cell donation.
Patients receiving a liver transplantation could be divided into two groups, early and late transplantations after HSCT. Early liver transplantation (before six months post-HSCT) was performed because of acute liver failure due to veno-occlusive disease (VOD) or acute GVHD, late transplantation (usually 2-5 years post-HSCT) mostly because of chronic liver-GVHD or cirrhosis. In 75% of the transplantations an unrelated deceased donor was used.
In all lung transplantations the cause of the respiratory failure was bronchiolitis obliterans. In ten of the twelve cases the donor was a deceased unrelated donor. One of the two related donors was the HSCT donor.
The overall 5-year survival after SOT was 78%, in renal transplantations 100%, in liver transplantations 71% and in lung transplantations 63%. None of the renal transplantation patients was on dialysis at the end of the follow-up. The 2-year incidence of SOT failure was overall 16%, 20% in patients with GVHD preceding the SOT and 7% in those with no GVHD. Graft rejection occurred in approximately 30% of the patients with each type of transplantation, but it could usually be treated. Two patients with renal transplantation, 2 with liver transplantation and 4 with lung transplantation experienced graft failure. The relapse incidence of the underlying malignant disease was 4% at 5 years after the SOT. Of the six patients who received the SOT from the original HSCT donor (4 renal, 1 liver, 1 lung transplantation), all but the lung transplantation patient were alive at the time of the analysis.
Bronchiolitis obliterans has been the main indication for lung transplantation after allogeneic HSCT. Holm et al [10] reported on 13 such patients from the Nordic countries. Their median age was 34 (range 16-55) years. The indication for the HSCT had been a malignant hematological disease in all but two patients. All suffered from chronic GvHD. The median time from the HSCT to the lung transplantation was 8.2 (range 0.7-16) years. All had a bilateral lung transplantation. With a median follow-up time of 4.2 years from the lung transplantation, the survival of the patients was 90% at one year and 75% at 5 years. Of the two patients who died, one died of relapse of the underlying disease and one of infection. These results were compared to data obtained from the Scandiatransplant registry and found not to be differ ent from the general population of lung transplant patients. Four patients developed BOS in the transplanted lung; in one case, a second transplantation was carried out. Vogl and coworkers [27] reported a cohort of seven patients who received lung transplantation for bronchiolitis obliterans after allogeneic HSCT. In this cohort there was a case fatality rate of 57% and the median survival was 24 months after the lung transplantation. In this group of patients the interval between the HSCT and the lung transplantation was much shorter than in the study of Holm et al, median 18 (6-120) months. Cheng et al [2] reported on the outcome of nine patients with lung transplantation after allogeneic HSCT with a survival of 89 and 37% at 1 and 5 years, respectively. Jung ad coworkers [11] published recently their experience with nine patients treated with lung transplantation for BOS. After a relatively short median follow-up of 17 months, six of the nine patients were in good health. Soubani et al [21] reviewed recently the literature and identified 79 recipients of lung transplantation after allogeneic HSCT, including the patients of most of the reports referred to above. The median time from the HSCT to the lung transplantation was 52 months, and at three years the survival was 79%. Thirty per cent of the patients had developed BOS after the transplantation. The risk of relapse of the underlying hematological disease did not seem to be increased.
Upadhyay and Fine [26] have presented a review on solid organ transplantation following end-organ failure in recipients of hematopoietic stem cell transplantation in children. The results have been dependent on various risk factors, but especially the kidney transplant patients have fared well, and the authors recommend transplantation of the failed organs to be considered as potential treatment in selected patients.
Experience with allogeneic HSCT in solid organ transplant recipients
Basak et al [1] reported a survey of allogeneic HSCT carried out after SOT at EBMT centres. Thirty-one HSCTs had been performed in 28 SOT recipients. Thirteen patients had a preceding liver transplantation, 12 kidney transplantation, and 3 heart transplantation. The indication for the HSCT was a malignant hematological disorder in 22/28 patients, mostly acute leukemia. The diagnosis leading to HSCT was known before the SOT in 8/28 patients. The median time from SOT to HSCT was 37 (range 1-315 months). Two thirds of the hematopoietic grafts were from peripheral blood. Approximately one half of the donors were HLA-identical siblings, the rest unrelated or haploidentical. Half of the patients received myeloablative, the other half reduced intensity conditioning. All evaluable patients except one showed engraftment, one graft was lost later. The incidence of acute and chronic GVHD was not clearly different from what could be expected in a general HSCT population. Despite the often advanced state of the underlying disease at the time of HSCT, the relapse rate was low, 22%. With a 5-year follow-up, solid organ graft failure occurred in 9 of 31 patients, in 5/13 (38%) of the renal transplantations, 3/15 (20%) of the liver transplantations, and in one of the three heart transplantations. Five of the nine failures were defined as graft rejection. The median time to graft failure was 1.8 months with a range of 0 to 131 months. The TRM at three months was 25%. Infection was the most common cause of death. The overall survival at five years after the allogeneic HSCT was 40% for all patients, 51% for liver transplant recipients and 42% for renal transplant recipients. There were no significant differences in the outcomes between different types of HSCT donors or grafts, or the conditioning intensity.
Doney et al [5] reported on eight patients who had received an allogeneic HSCT after SOT at their center. Four of the eight patients were alive after a median follow-up of 8.7 years post-transplantation. Schechter et al [20] published four pediatric patients, two of whom had been treated with HSCT for PTLD after heart transplantation, and two patients transplanted for aplastic anemia after liver transplantation. All four fared poorly and died within one year.
Discussion
Although a considerable number of reports describing SOT following HSCT have been published, the number of such transplantations has been small in relation to the frequent problems of organ injury and failure seen in clinical practice. There are apparently many reasons to this, including the often complicated clinical situation, poor general condition of the patient, and possibly a certain resistance of clinicians to consider such a treatment approach, but also insufficient information of the results of such transplantations. The described recent publications show that the outcomes can be quite good. In a previous survey [13], the 5-year survival of patients receiving a renal transplantation was 100 %, with no patient being on dialysis. The 5-year survival for liver transplantation was 71% and that for lung transplantation 63%. These figures are not markedly different from the outcomes in general SOT populations. It is clear that the patient populations in dual transplantations have been selected, with young patient age, benign diseases somewhat overrepresented, and underlying malignant disease under good control, but the results show that SOT carried out for organ failure after HSCT is a feasible treatment option in carefully selected patients. There is no indication that prolonged immunosuppressive treatment due to the SOT would significantly increase the risk of disease relapse. However, it has to be noted that in a large proportion of the patients in the survey [13], the SOTs following HSCT were carried out late, when the risk of relapse is already relatively low.
Bronchiolitis obliterans is a serious and problematic complication of allogeneic HSCT, for which there is no effective treatment. Therefore the good results of lung transplantations for this disorder, reported in some studies, are encouraging. In the report of Holm et al [10], the 5-year OS was 75%, in the report by Koenecke [13], 63%. In contrast, in the report of Vogl et al [27], the median survival in a group of seven patients was only 24 months and the case fatality rate 57%. As discussed by Holm et al [10], a major difference between the patient materials between their study and that of Vogl et al was the timing of the lung transplantation, the median time from the HSCT being 8.2 years in the Holm study vs. 18 months in the study by Vogl [27]. This proba bly reflects, at least in part, the general importance of patient selection; patients undergoing SOT early after HSCT being more likely to be at a high risk of failure because of a rapidly progressive complication and a more fragile general condition due to the recent HSCT.
The experience with dual transplantations including a cardiac transplantation published is small, consisting mainly of case reports [1,3,13,15,16,20]. Therefore, no general conclusions of heart transplantations in combination with HSCT can be drawn.
In a HSCT recipient treated with a SOT, the immunological conditions may be complicated. The immunosuppressive strategy applied in the two types of transplantations is somewhat different. These factors might affect the risk of solid organ graft rejection. In the survey [13], graft rejection was seen in renal transplantations in 4/13 cases, leading to kidney failure in two patients. These were treated with a second renal transplantation. In 2/14 liver transplantations rejection led to graft failure. In 2/10 lung transplantations rejection resulted in respiratory failure, in one case this was treated with retransplantation. Among the patients treated with lung transplantation in the study [10], no rejection leading to organ failure took place. Therefore, also given the small numbers of patients, the incidences of graft rejection were not markedly different from those that can be seen in non-HSCT SOT patients. The occurrence or absence of GVHD did not significantly affect the outcome.
The number of reports of HSCT in SOT recipients in the literature is rather limited. In such HSCTs the function and fate of the solid organ graft would be a concern. As shown in the survey [1], graft failure is a concrete risk; this was seen in 9/31 patients. There was a renal graft failure in 38%, liver graft failure in 20% and heart graft failure in 1/3 of the patients. In approximately half of the cases this was reported as being the result of rejection. There were no major problems with hematopoietic engraftment; with one exception, engraftment took place in all evaluable patients.
There are many possible mechanism of graft rejection in this transplant setting. An immune response can be directed against mismatched HLA molecules, because the hematopoietic graft is HLA-matched with the recipient rather than the solid organ. Conditioning prior to allogeneic HSCT and infection may increase the immunogenicity of the organ graft by enhancing antigen presentation, increasing costimulatory signals, changing the properties of the vascular endothelium, and suppressing regulatory T-cell function. Whereas the majority of solid organ grafts are matched with the recipient based on blood group, this is not required for HSCT donors. Thus, the majority of hematopoietic transplants are probably not matched with the solid organ graft, and this could affect the survival of the organ graft. Moreover, in most patients, the immunosuppressive regimen is changed after allogeneic HSCT from a regimen typical for SOT to an HSCT-specific regimen, which could be suboptimal in this situation.
Experience has been reported to show that tolerance to kidney transplant may occasionally occur after preceding HSCT, the organ graft surviving without any immunosuppression [4,9,18]. Some groups have worked for many years to develop methods to achieve tolerance to solid organ transplants by hematopoietic cell transplantation, aiming at mixed or complete chimerism [7,8,14,19,22-24,28,29]. The aim would be to avoid life-long immunosuppressive treatment with its adverse effects. After a long period of preclinical work some clinical experience is now available, mainly in kidney transplant patients [12,14,19]. It has been possible to discontinue immunosuppressive treatment permanently or for a long period of time in a significant proportion of organ transplant recipients after tolerance induction. The balance between the conditioning treatment, donor, chimerism, immunosuppressive treatment, organ rejection and graft-versus-host diseases is delicate, but this approach is of great importance with potentially major clinical consequences.
In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. In stringently selected young patients, the overall and organ survivals appear to be comparable to patients undergoing SOT for other causes. Complications, such as infections and graft rejection are frequent but usually manageable. Thus, SOT offers a viable therapeutic option for selected patients with single organ failure after HSCT. Also, selected SOT recipients suffering from hematologic disorders may benefit from allogeneic HSCT and experience long-term survival without loss of organ function.
Conflict of interests
No conflict of interests is declared.
References
- Basak GW, Wiktor-Jedrzejczak W, Labopin M, Schoemans H, Ljungman P, Kobbe G, Beguin Y, Lang P, Koenecke C, Sykora KW, Te Boome L, van Biezen A, van der Werf S, Mohty M, de Witte T, Marsh J, Dreger P, Kröger N, Duarte R, Ruutu T. Allogeneic hematopoietic stem cell transplantation in solid organ transplant recipients: a retrospective, multicenter study of the EBMT. Am J Transplant. 2015; 15 (3): 705-714.
- Cheng G-S, Edelman JD, Madts DK, Martin PJ, Flowers MED: Outcomes of lung transplantation after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014; 20: 1169-1175.
- Chiang KY, Lazarus HM. Should we be performing more combined hematopoietic stem cell plus solid organ transplants? Bone Marrow Transplant 2003; 31: 633-642.
- Dey B, Sykes M, Spitzer T. Outcomes of recipients of both bone marrow and solid organ transplants: a review. Medicine 1998; 77: 355-369.
- Doney KC, Mielcarek M, Stewart FM, Appelbaum FR. Hematopoietic cell transplantation after solid organ transplantation. Biol Blood Marrow Transplant 2015; 21 (12): 2123-2128.
- EDQM, European Directorate for the Quality of Medicines & Health Care of the Council of Europe. Newsletter Transplant. International figures on donation and transplantation 2014.
- Fuchs EJ. Transplantation tolerance: from theory to clinic. Immunol Rev 2014; 258 (1): 64-79.
- Granados JMM, Benichou G, Kawai T. Hematopoietic stem cell infusion/ transplantation. Curr Opin Organ Transplant 2015, 20: 49-56.
- Helg C, Chapius B, Bolle JF, Morel P, Salomon D, Roux E, Antonioli V, Jeannet M, Leski M. Renal transplantation wihout immunosuppression in a shot with tolerance induced by allogeneic bone marrow transplantation. Transplantation 1994; 58: 1320-1421.
- Holm AM, Riise GC, Hansson L, Brinch L, Bjørtuft O, Iversen M, Simonsen S, Fløisand Y. Lung transplantation for bronchiolitis obliterans syndrome after allo-SCT. Bone Marrow Transplant. 2013; 48 (5): 703-707.
- Jung HS, Lee JG, Yu WS, Lee CY, Haam SJ, Paik HC. Early outcomes of lung transplantation for bronchiolitis obliterans syndrome after allogeneic haematopoietic stem cell transplantation; a single-centre experience. Interact CardioVasc Thorac Surg 2016; doi: 10.1093/icvts/ivw231
- Kawai T, Sachs DH, Sprangers B, Spitzer TR, Saidman SL, Zorn E, Tolkoff-Rubin N, Preffer F, Crisalli K, Gao B, Wong W, Morris H, LoCascio SA, Sayre P, Shonts B, Williams WW Jr, Smith RN, Colvin RB, Sykes M, Cosimi AB. Long-term results in recipients of combined HLA-mismatched kidney and bone marrowtransplantation without maintenance immunosuppression. Am J Transplant. 2014; 14 (7): 1599-1611.
- Koenecke C, Hertenstein B, Schetelig J, van Biezen A, Dammann E, Gratwohl A, Ganser A, Schleuning M, Bornhäuser M, Jacobsen N, Kröger N, Niederwieser D, de Witte T, Ruutu T. Solid organ transplantation after allogeneic hematopoietic stem cell transplantation: a retrospective, multicenter study of the EBMT. Am J Transplant. 2010; 10 (8): 1897-1906.
- Leventhal JR, Elliott MJ, Yolcu ES, Bozulic LD, Tollerud DJ, Mathew JM, Konieczna I, Ison MG, Galcin J, Mehta J, BadderMD, Abecassis MMI, Miller J, Gallon L, Ildstad ST. Immune reconstitution/ Immunocompetence in recipients of kidney plus hematopoietic stem/ facilitating cell transplants. Transplantation 2015; 99: 288-298.
- Lin RJ, Larson RA, van Besien K, Rich ES. Allogeneic hematopoietic cell transplantation for therapy-related myeloid leukemia following orthotopic cardiac transplantation. Case Reports in Hematology 2013; article ID 140138.
- Lister J, Simpson JK, deMagalhaes-Silverman MM, Rybka WB, Donnenberg AD, Myers DJ, Ball ED. Allogeneic peripheral blood stem cell transplant for myelodysplasia after chemotherapy for post-transplant lymphoma in a cardiac transplant recipient at 10 years. Bone Marrow Transplant 1997; 19: 943-945.
- Passweg JR, Baldomero H, Bader P, Bonini C, Cesaro S, Dreger P, Duarte RF, Dufour C, Kuball J, Farge-Bancel D, Gennery A, Kröger N, Lanza F, Nagler A, Sureda A, Mohty M for the European Society for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 2016; 51: 786-792.
- Sayegh MH, Fine NA, Smith JL, Rennke HG, Milford EL, Tilney NL. Immunological tolerance to renal allografts after bone marrow transplants from the same donors Ann Intern Med 1991; 114: 954-955.
- Scandling JD, Busque S, Shizuru JA, Lowsky R, Hoppe R, Dejbakhsh-Jones S, Jensen K, Shore A, Strober JA, Lavori P, Turnbull BB, Engleman EG, Strober S. Chimerism, graft survival, and withdrawal of immunosuppressive drugs in HLA matched and mismatched patients after living donor kidney and hematopoietic cell transplantation. Am J Transplant 2015: 15: 695-704.
- Schechter T, Gassas A, Weitzman S, Grant D, Pollock-BarZiv S, Dipchand A, Alexander S, Ali M, Avitzur Y, Doyle J. Hematopoietic stem-cell transplantation following solid-organ transplantation in children. Bone Marrow Transplant 2011; 46: 1321-1325.
- Soubani AO, Kingah P, Alshabani K, Muma G, Haq A. Lung transplantation following hematopoietic stem cell transplantation: report of two cases and systematic review of literature. Clin Transplant 2014; 28: 772-782.
- Spitzer TR: Intersection of hematopoietic cell and solid organ transplantation: lessons learned and unanswered questions. Biol Blood Marrow Transplant 2015; 21: 2037-2038.
- Strober S. Use of hematopoietic cell transplants to achieve tolerance in patients with solid organ transplants. Blood 2016; 127 (12): 1539-1543.
- Sykes M. Immune tolerance in recipients of combined haploidentical bone marrow and kidney transplantation. Bone Marrow Transplant 2015; 50: S82-S86.
- Thomas SE, Hutchinson RJ, DebRoy M, Magee JC. Successful renal transplantation following prior bone marrow transplantation in pediatric patients. Pediatr Transplant. 2004; 8 (5): 507-512.
- Upadhyay K, Fine RN. Solid organ transplantation following end-organ failure in recipients of hematopoietic stem cell transplantation in children. Pediatr Nephrol. 2014; 29 (8): 1337-1347.
- Vogl UM, Nagayama K, Bojic M, Hoda MA, Klepetko W, Jaksch P, Dekan S, Siersch V, Mitterbauer M, Schellongowski P, Greinix HT, Petkov V, Schulenburg A, Kalhs P, Rabitsch W. Lung transplantation for bronchiolitis obliterans afer allogeneic hemaopoietic stem cell transplantation: a single- center experience. Transplantation 2013; 95:623-628.
- Yolcu ES, Leventhal JR, Ildstad S. Facilitating cells in tolerance induction for kidney transplantation. Curr Opin Organ Transplant 2015; 20: 57-63.
- Zakrzewski JL, van den Brink MRM, Hubbell JA. Overcoming immunological barriers in regenerative medicine. Nature Biotechnol 2014; 32 (8): 786-794.
Introduction
Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. About 32000 SOTs were performed in the European Union in 2014, comprising approximately 20000 kidney, 7400 liver, 2100 heart, 1800 lung and 900 other transplants [6]. Allogeneic hematopoietic stem cell transplantation (HSCT) is the only cure or the most effective treatment for a large number of malignant and non-malignant diseases of the lympho-hematopoietic system with an otherwise poor prognosis. At present, the number of allogeneic HSCTs in Europe is more than 15000 per year [17]. With these large numbers of patients, the likelihood of coincidental occurrence of disorders potentially indicating SOT and HSCT in the same patient is increasing. Such transplantation combinations can be problematic for many reasons.
Allogeneic HSCT is an intensive treatment which frequently causes organ injury and failure, resulting in a potential indication for a SOT. The mechanisms of organ injury include cytotoxic drugs and radiotherapy in the pre-transplant conditioning, organ-toxic agents in supportive care, and immunological mechanisms. The organs most often affected are kidney, liver, lung and heart. Factors that may complicate a SOT in this setting are the fragility of the patient as a consequence of the toxicity of HSCT, immunosuppressive treatment for the HSCT which may not be optimal for the SOT, potential immunological problems caused by tissue type differences between the recipient, the hematopoietic graft and the solid organ graft, and infections. In SOT recipients, the long-lasting immunosuppressive medication may affect the function of organs, and increase the risk of infections. SOT can be associated with the development of hematological disorders comprising aplastic anemia, post-transplant lymphoproliferative disease, acute myeloid leukemia and myelodysplastic syndromes which can form a potential indication for allogeneic HSCT.
As the clinical situation is often found to be complicated and the risk of treatment failure regarded as high, the numbers of SOTs to HSCT recipients and those of HSCTs to SOT recipients have been low as compared to the number of patients who would have a potential indication for such treatment. Until recent years the literature of such transplantations consisted mainly of case reports as reviewed elsewhere [1,3,13], with the obvious risk of publication bias. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients.
Experience with organ transplantation in hematopoietic stem cell transplant recipients
There is a rather limited published experience available for the assessment of the indications for SOT in HSCT recipients. Koenecke et al [13] carried out a survey among 107 EBMT (European Society for Blood and Marrow Transplantation) centres, with 67000 allogeneic transplantations performed during the study period of 1984-2007. In 28 centres a total of 45 SOTs had been carried out in 40 patients who had previously undergone allogeneic HSCT, of them 15 renal, 15 liver, 13 lung, 1 heart and 1 skin transplantation. Hence, the approximate frequency of these interventions was 67 SOTs per 100 000 HSCTs. Moreover, seven additional SOTs were identified from case reports or from centres that decided not to participate, altogether bringing the total number to 52 SOTs. The median age of the patients was between 20 and 35 years, depending of the organ graft type. The liver transplant patients were the youngest ones. Most of the patients had leukaemia, but about 15% had aplastic anaemia and another 15% an inherited disorder. A large majority of the patients received myeloablative conditioning. The graft had been bone marrow in 60%, and the donor was a matched related donor in 54% of the cases. Approximately half of the patients developed acute and/or chronic graft-versus-host disease. The median time from the HSCT to the organ transplantation ranged from 34 (liver transplants) to 84 (kidney transplants) months.
In kidney transplantations, the most frequent cause of renal failure was drug treatment or radiotherapy. All patients were on dialysis at the time of the SOT. The donor was unrelated in four cases and related in nine cases; of the latter four were the same as in the blood stem cell donation.
Patients receiving a liver transplantation could be divided into two groups, early and late transplantations after HSCT. Early liver transplantation (before six months post-HSCT) was performed because of acute liver failure due to veno-occlusive disease (VOD) or acute GVHD, late transplantation (usually 2-5 years post-HSCT) mostly because of chronic liver-GVHD or cirrhosis. In 75% of the transplantations an unrelated deceased donor was used.
In all lung transplantations the cause of the respiratory failure was bronchiolitis obliterans. In ten of the twelve cases the donor was a deceased unrelated donor. One of the two related donors was the HSCT donor.
The overall 5-year survival after SOT was 78%, in renal transplantations 100%, in liver transplantations 71% and in lung transplantations 63%. None of the renal transplantation patients was on dialysis at the end of the follow-up. The 2-year incidence of SOT failure was overall 16%, 20% in patients with GVHD preceding the SOT and 7% in those with no GVHD. Graft rejection occurred in approximately 30% of the patients with each type of transplantation, but it could usually be treated. Two patients with renal transplantation, 2 with liver transplantation and 4 with lung transplantation experienced graft failure. The relapse incidence of the underlying malignant disease was 4% at 5 years after the SOT. Of the six patients who received the SOT from the original HSCT donor (4 renal, 1 liver, 1 lung transplantation), all but the lung transplantation patient were alive at the time of the analysis.
Bronchiolitis obliterans has been the main indication for lung transplantation after allogeneic HSCT. Holm et al [10] reported on 13 such patients from the Nordic countries. Their median age was 34 (range 16-55) years. The indication for the HSCT had been a malignant hematological disease in all but two patients. All suffered from chronic GvHD. The median time from the HSCT to the lung transplantation was 8.2 (range 0.7-16) years. All had a bilateral lung transplantation. With a median follow-up time of 4.2 years from the lung transplantation, the survival of the patients was 90% at one year and 75% at 5 years. Of the two patients who died, one died of relapse of the underlying disease and one of infection. These results were compared to data obtained from the Scandiatransplant registry and found not to be differ ent from the general population of lung transplant patients. Four patients developed BOS in the transplanted lung; in one case, a second transplantation was carried out. Vogl and coworkers [27] reported a cohort of seven patients who received lung transplantation for bronchiolitis obliterans after allogeneic HSCT. In this cohort there was a case fatality rate of 57% and the median survival was 24 months after the lung transplantation. In this group of patients the interval between the HSCT and the lung transplantation was much shorter than in the study of Holm et al, median 18 (6-120) months. Cheng et al [2] reported on the outcome of nine patients with lung transplantation after allogeneic HSCT with a survival of 89 and 37% at 1 and 5 years, respectively. Jung ad coworkers [11] published recently their experience with nine patients treated with lung transplantation for BOS. After a relatively short median follow-up of 17 months, six of the nine patients were in good health. Soubani et al [21] reviewed recently the literature and identified 79 recipients of lung transplantation after allogeneic HSCT, including the patients of most of the reports referred to above. The median time from the HSCT to the lung transplantation was 52 months, and at three years the survival was 79%. Thirty per cent of the patients had developed BOS after the transplantation. The risk of relapse of the underlying hematological disease did not seem to be increased.
Upadhyay and Fine [26] have presented a review on solid organ transplantation following end-organ failure in recipients of hematopoietic stem cell transplantation in children. The results have been dependent on various risk factors, but especially the kidney transplant patients have fared well, and the authors recommend transplantation of the failed organs to be considered as potential treatment in selected patients.
Experience with allogeneic HSCT in solid organ transplant recipients
Basak et al [1] reported a survey of allogeneic HSCT carried out after SOT at EBMT centres. Thirty-one HSCTs had been performed in 28 SOT recipients. Thirteen patients had a preceding liver transplantation, 12 kidney transplantation, and 3 heart transplantation. The indication for the HSCT was a malignant hematological disorder in 22/28 patients, mostly acute leukemia. The diagnosis leading to HSCT was known before the SOT in 8/28 patients. The median time from SOT to HSCT was 37 (range 1-315 months). Two thirds of the hematopoietic grafts were from peripheral blood. Approximately one half of the donors were HLA-identical siblings, the rest unrelated or haploidentical. Half of the patients received myeloablative, the other half reduced intensity conditioning. All evaluable patients except one showed engraftment, one graft was lost later. The incidence of acute and chronic GVHD was not clearly different from what could be expected in a general HSCT population. Despite the often advanced state of the underlying disease at the time of HSCT, the relapse rate was low, 22%. With a 5-year follow-up, solid organ graft failure occurred in 9 of 31 patients, in 5/13 (38%) of the renal transplantations, 3/15 (20%) of the liver transplantations, and in one of the three heart transplantations. Five of the nine failures were defined as graft rejection. The median time to graft failure was 1.8 months with a range of 0 to 131 months. The TRM at three months was 25%. Infection was the most common cause of death. The overall survival at five years after the allogeneic HSCT was 40% for all patients, 51% for liver transplant recipients and 42% for renal transplant recipients. There were no significant differences in the outcomes between different types of HSCT donors or grafts, or the conditioning intensity.
Doney et al [5] reported on eight patients who had received an allogeneic HSCT after SOT at their center. Four of the eight patients were alive after a median follow-up of 8.7 years post-transplantation. Schechter et al [20] published four pediatric patients, two of whom had been treated with HSCT for PTLD after heart transplantation, and two patients transplanted for aplastic anemia after liver transplantation. All four fared poorly and died within one year.
Discussion
Although a considerable number of reports describing SOT following HSCT have been published, the number of such transplantations has been small in relation to the frequent problems of organ injury and failure seen in clinical practice. There are apparently many reasons to this, including the often complicated clinical situation, poor general condition of the patient, and possibly a certain resistance of clinicians to consider such a treatment approach, but also insufficient information of the results of such transplantations. The described recent publications show that the outcomes can be quite good. In a previous survey [13], the 5-year survival of patients receiving a renal transplantation was 100 %, with no patient being on dialysis. The 5-year survival for liver transplantation was 71% and that for lung transplantation 63%. These figures are not markedly different from the outcomes in general SOT populations. It is clear that the patient populations in dual transplantations have been selected, with young patient age, benign diseases somewhat overrepresented, and underlying malignant disease under good control, but the results show that SOT carried out for organ failure after HSCT is a feasible treatment option in carefully selected patients. There is no indication that prolonged immunosuppressive treatment due to the SOT would significantly increase the risk of disease relapse. However, it has to be noted that in a large proportion of the patients in the survey [13], the SOTs following HSCT were carried out late, when the risk of relapse is already relatively low.
Bronchiolitis obliterans is a serious and problematic complication of allogeneic HSCT, for which there is no effective treatment. Therefore the good results of lung transplantations for this disorder, reported in some studies, are encouraging. In the report of Holm et al [10], the 5-year OS was 75%, in the report by Koenecke [13], 63%. In contrast, in the report of Vogl et al [27], the median survival in a group of seven patients was only 24 months and the case fatality rate 57%. As discussed by Holm et al [10], a major difference between the patient materials between their study and that of Vogl et al was the timing of the lung transplantation, the median time from the HSCT being 8.2 years in the Holm study vs. 18 months in the study by Vogl [27]. This proba bly reflects, at least in part, the general importance of patient selection; patients undergoing SOT early after HSCT being more likely to be at a high risk of failure because of a rapidly progressive complication and a more fragile general condition due to the recent HSCT.
The experience with dual transplantations including a cardiac transplantation published is small, consisting mainly of case reports [1,3,13,15,16,20]. Therefore, no general conclusions of heart transplantations in combination with HSCT can be drawn.
In a HSCT recipient treated with a SOT, the immunological conditions may be complicated. The immunosuppressive strategy applied in the two types of transplantations is somewhat different. These factors might affect the risk of solid organ graft rejection. In the survey [13], graft rejection was seen in renal transplantations in 4/13 cases, leading to kidney failure in two patients. These were treated with a second renal transplantation. In 2/14 liver transplantations rejection led to graft failure. In 2/10 lung transplantations rejection resulted in respiratory failure, in one case this was treated with retransplantation. Among the patients treated with lung transplantation in the study [10], no rejection leading to organ failure took place. Therefore, also given the small numbers of patients, the incidences of graft rejection were not markedly different from those that can be seen in non-HSCT SOT patients. The occurrence or absence of GVHD did not significantly affect the outcome.
The number of reports of HSCT in SOT recipients in the literature is rather limited. In such HSCTs the function and fate of the solid organ graft would be a concern. As shown in the survey [1], graft failure is a concrete risk; this was seen in 9/31 patients. There was a renal graft failure in 38%, liver graft failure in 20% and heart graft failure in 1/3 of the patients. In approximately half of the cases this was reported as being the result of rejection. There were no major problems with hematopoietic engraftment; with one exception, engraftment took place in all evaluable patients.
There are many possible mechanism of graft rejection in this transplant setting. An immune response can be directed against mismatched HLA molecules, because the hematopoietic graft is HLA-matched with the recipient rather than the solid organ. Conditioning prior to allogeneic HSCT and infection may increase the immunogenicity of the organ graft by enhancing antigen presentation, increasing costimulatory signals, changing the properties of the vascular endothelium, and suppressing regulatory T-cell function. Whereas the majority of solid organ grafts are matched with the recipient based on blood group, this is not required for HSCT donors. Thus, the majority of hematopoietic transplants are probably not matched with the solid organ graft, and this could affect the survival of the organ graft. Moreover, in most patients, the immunosuppressive regimen is changed after allogeneic HSCT from a regimen typical for SOT to an HSCT-specific regimen, which could be suboptimal in this situation.
Experience has been reported to show that tolerance to kidney transplant may occasionally occur after preceding HSCT, the organ graft surviving without any immunosuppression [4,9,18]. Some groups have worked for many years to develop methods to achieve tolerance to solid organ transplants by hematopoietic cell transplantation, aiming at mixed or complete chimerism [7,8,14,19,22-24,28,29]. The aim would be to avoid life-long immunosuppressive treatment with its adverse effects. After a long period of preclinical work some clinical experience is now available, mainly in kidney transplant patients [12,14,19]. It has been possible to discontinue immunosuppressive treatment permanently or for a long period of time in a significant proportion of organ transplant recipients after tolerance induction. The balance between the conditioning treatment, donor, chimerism, immunosuppressive treatment, organ rejection and graft-versus-host diseases is delicate, but this approach is of great importance with potentially major clinical consequences.
In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. In stringently selected young patients, the overall and organ survivals appear to be comparable to patients undergoing SOT for other causes. Complications, such as infections and graft rejection are frequent but usually manageable. Thus, SOT offers a viable therapeutic option for selected patients with single organ failure after HSCT. Also, selected SOT recipients suffering from hematologic disorders may benefit from allogeneic HSCT and experience long-term survival without loss of organ function.
Conflict of interests
No conflict of interests is declared.
References
- Basak GW, Wiktor-Jedrzejczak W, Labopin M, Schoemans H, Ljungman P, Kobbe G, Beguin Y, Lang P, Koenecke C, Sykora KW, Te Boome L, van Biezen A, van der Werf S, Mohty M, de Witte T, Marsh J, Dreger P, Kröger N, Duarte R, Ruutu T. Allogeneic hematopoietic stem cell transplantation in solid organ transplant recipients: a retrospective, multicenter study of the EBMT. Am J Transplant. 2015; 15 (3): 705-714.
- Cheng G-S, Edelman JD, Madts DK, Martin PJ, Flowers MED: Outcomes of lung transplantation after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2014; 20: 1169-1175.
- Chiang KY, Lazarus HM. Should we be performing more combined hematopoietic stem cell plus solid organ transplants? Bone Marrow Transplant 2003; 31: 633-642.
- Dey B, Sykes M, Spitzer T. Outcomes of recipients of both bone marrow and solid organ transplants: a review. Medicine 1998; 77: 355-369.
- Doney KC, Mielcarek M, Stewart FM, Appelbaum FR. Hematopoietic cell transplantation after solid organ transplantation. Biol Blood Marrow Transplant 2015; 21 (12): 2123-2128.
- EDQM, European Directorate for the Quality of Medicines & Health Care of the Council of Europe. Newsletter Transplant. International figures on donation and transplantation 2014.
- Fuchs EJ. Transplantation tolerance: from theory to clinic. Immunol Rev 2014; 258 (1): 64-79.
- Granados JMM, Benichou G, Kawai T. Hematopoietic stem cell infusion/ transplantation. Curr Opin Organ Transplant 2015, 20: 49-56.
- Helg C, Chapius B, Bolle JF, Morel P, Salomon D, Roux E, Antonioli V, Jeannet M, Leski M. Renal transplantation wihout immunosuppression in a shot with tolerance induced by allogeneic bone marrow transplantation. Transplantation 1994; 58: 1320-1421.
- Holm AM, Riise GC, Hansson L, Brinch L, Bjørtuft O, Iversen M, Simonsen S, Fløisand Y. Lung transplantation for bronchiolitis obliterans syndrome after allo-SCT. Bone Marrow Transplant. 2013; 48 (5): 703-707.
- Jung HS, Lee JG, Yu WS, Lee CY, Haam SJ, Paik HC. Early outcomes of lung transplantation for bronchiolitis obliterans syndrome after allogeneic haematopoietic stem cell transplantation; a single-centre experience. Interact CardioVasc Thorac Surg 2016; doi: 10.1093/icvts/ivw231
- Kawai T, Sachs DH, Sprangers B, Spitzer TR, Saidman SL, Zorn E, Tolkoff-Rubin N, Preffer F, Crisalli K, Gao B, Wong W, Morris H, LoCascio SA, Sayre P, Shonts B, Williams WW Jr, Smith RN, Colvin RB, Sykes M, Cosimi AB. Long-term results in recipients of combined HLA-mismatched kidney and bone marrowtransplantation without maintenance immunosuppression. Am J Transplant. 2014; 14 (7): 1599-1611.
- Koenecke C, Hertenstein B, Schetelig J, van Biezen A, Dammann E, Gratwohl A, Ganser A, Schleuning M, Bornhäuser M, Jacobsen N, Kröger N, Niederwieser D, de Witte T, Ruutu T. Solid organ transplantation after allogeneic hematopoietic stem cell transplantation: a retrospective, multicenter study of the EBMT. Am J Transplant. 2010; 10 (8): 1897-1906.
- Leventhal JR, Elliott MJ, Yolcu ES, Bozulic LD, Tollerud DJ, Mathew JM, Konieczna I, Ison MG, Galcin J, Mehta J, BadderMD, Abecassis MMI, Miller J, Gallon L, Ildstad ST. Immune reconstitution/ Immunocompetence in recipients of kidney plus hematopoietic stem/ facilitating cell transplants. Transplantation 2015; 99: 288-298.
- Lin RJ, Larson RA, van Besien K, Rich ES. Allogeneic hematopoietic cell transplantation for therapy-related myeloid leukemia following orthotopic cardiac transplantation. Case Reports in Hematology 2013; article ID 140138.
- Lister J, Simpson JK, deMagalhaes-Silverman MM, Rybka WB, Donnenberg AD, Myers DJ, Ball ED. Allogeneic peripheral blood stem cell transplant for myelodysplasia after chemotherapy for post-transplant lymphoma in a cardiac transplant recipient at 10 years. Bone Marrow Transplant 1997; 19: 943-945.
- Passweg JR, Baldomero H, Bader P, Bonini C, Cesaro S, Dreger P, Duarte RF, Dufour C, Kuball J, Farge-Bancel D, Gennery A, Kröger N, Lanza F, Nagler A, Sureda A, Mohty M for the European Society for Blood and Marrow Transplantation (EBMT). Bone Marrow Transplant 2016; 51: 786-792.
- Sayegh MH, Fine NA, Smith JL, Rennke HG, Milford EL, Tilney NL. Immunological tolerance to renal allografts after bone marrow transplants from the same donors Ann Intern Med 1991; 114: 954-955.
- Scandling JD, Busque S, Shizuru JA, Lowsky R, Hoppe R, Dejbakhsh-Jones S, Jensen K, Shore A, Strober JA, Lavori P, Turnbull BB, Engleman EG, Strober S. Chimerism, graft survival, and withdrawal of immunosuppressive drugs in HLA matched and mismatched patients after living donor kidney and hematopoietic cell transplantation. Am J Transplant 2015: 15: 695-704.
- Schechter T, Gassas A, Weitzman S, Grant D, Pollock-BarZiv S, Dipchand A, Alexander S, Ali M, Avitzur Y, Doyle J. Hematopoietic stem-cell transplantation following solid-organ transplantation in children. Bone Marrow Transplant 2011; 46: 1321-1325.
- Soubani AO, Kingah P, Alshabani K, Muma G, Haq A. Lung transplantation following hematopoietic stem cell transplantation: report of two cases and systematic review of literature. Clin Transplant 2014; 28: 772-782.
- Spitzer TR: Intersection of hematopoietic cell and solid organ transplantation: lessons learned and unanswered questions. Biol Blood Marrow Transplant 2015; 21: 2037-2038.
- Strober S. Use of hematopoietic cell transplants to achieve tolerance in patients with solid organ transplants. Blood 2016; 127 (12): 1539-1543.
- Sykes M. Immune tolerance in recipients of combined haploidentical bone marrow and kidney transplantation. Bone Marrow Transplant 2015; 50: S82-S86.
- Thomas SE, Hutchinson RJ, DebRoy M, Magee JC. Successful renal transplantation following prior bone marrow transplantation in pediatric patients. Pediatr Transplant. 2004; 8 (5): 507-512.
- Upadhyay K, Fine RN. Solid organ transplantation following end-organ failure in recipients of hematopoietic stem cell transplantation in children. Pediatr Nephrol. 2014; 29 (8): 1337-1347.
- Vogl UM, Nagayama K, Bojic M, Hoda MA, Klepetko W, Jaksch P, Dekan S, Siersch V, Mitterbauer M, Schellongowski P, Greinix HT, Petkov V, Schulenburg A, Kalhs P, Rabitsch W. Lung transplantation for bronchiolitis obliterans afer allogeneic hemaopoietic stem cell transplantation: a single- center experience. Transplantation 2013; 95:623-628.
- Yolcu ES, Leventhal JR, Ildstad S. Facilitating cells in tolerance induction for kidney transplantation. Curr Opin Organ Transplant 2015; 20: 57-63.
- Zakrzewski JL, van den Brink MRM, Hubbell JA. Overcoming immunological barriers in regenerative medicine. Nature Biotechnol 2014; 32 (8): 786-794.
Трансплантация органов (Tx) широко применяется для лечения тяжелой органной недостаточности. Аллогенная трансплантация гемопоэтических клеток (алло-ТГСК), как таковая, часто вызывает повреждение органов, что может быть показанием к Тх. В связи с возрастающим числом обоих видов лечения растет вероятность комбинированных состояний, требующих, как Тх, так и ТГСК у одного и того же пациента. Эти ситуации приводят к ряду клинических проблем. Так, факторами риска, способными повлиять на исходы Тх после ТГСК, могут быть: токсичность процедуры ТГСК, последующее иммуносупрессивное лечение, не адекватное режиму после Тх, возможные проблемы, связанные с тканевой несовместимостью больного, гемопоэтических клеток донора и органного трансплантата, а также развитие инфекций. В то же время иммуносупрессивная терапия больных после Тх может нарушать функции органа и повышать риск инфекций. Кроме того, Тх иногда ассоциирована с развитием гематологических заболеваний, как, например, апластической анемии, посттрансплантационных лимфопролиферативных заболеваний, острых лейкозов и др. Цель настоящей статьи состоит в анализе существующего клинического опыта в области алло-ТГСК и Тх у одних и тех же пациентов. В частности, рассматривается европейское исследование с участием 107 центров EBMT и клинические данные о 45 Тх, проведенных пациентам, которым ранее выполнялась ТГСК. Им проводили, главным образом трансплантацию почек из-за повреждений после химио- или радиотерапии, тогда как пересадки печени делали в ранние сроки после ТГСК в связи с тяжелой вено-окклюзионной болезнью или оРТПХб или позже, по поводу хронической РТПХ или цирроза. Выживаемость и клинические исходы анализировали для каждой клинической ситуации. Особое внимание уделяли больным, получавшим трансплантаты легких при облитерирующем альвеолите после алло-ТГСК, где наблюдались худшие клинические исходы и меньшие сроки выживания.
Ряд свежих работ касается алло-ТГСК, проведенных после Тх. Частота гематологических рецидивов составила 22%, что позволило длительно наблюдать пациентов. Пятилетняя выживаемость пересаженных органов была около 30%, в основном – из-за отторжения трансплантата.
Таким образом, Тх может быть ценной стратегией при лечении реципиентов гемопоэтических клеток в случае развития органной недостаточности. Инфекции, отторжение трансплантата и другие осложнения довольно часты, но обычно поддаются лечению. Кроме того, некоторые реципиенты органов, у которых развились гематологические заболевания, могут выиграть от алло-ТГСК.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["DOI"]=> array(36) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8145" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-15-20" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-15-20" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_EN"]=> array(36) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8146" ["VALUE"]=> array(2) { ["TEXT"]=> string(12) "Tapani Ruutu" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(12) "Tapani Ruutu" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_EN"]=> array(36) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8147" ["VALUE"]=> array(2) { ["TEXT"]=> string(308) "Tapani Ruutu, Helsinki University Hospital, Biomedicum<br> 2C, POB 705, FIN-00029 HUS, Helsinki, Finland<br> <br> Phone: +358 50 555 1279<br> Fax: +358 9 4717 1506<br> E-mail: <a href="mailto:tapani.ruutu@hus.fi">tapani.ruutu@hus.fi</a><br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(250) "Tapani Ruutu, Helsinki University Hospital, Biomedicum2C, POB 705, FIN-00029 HUS, Helsinki, Finland
Phone: +358 50 555 1279
Fax: +358 9 4717 1506
E-mail: tapani.ruutu@hus.fi
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_EN"]=> array(36) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8148" ["VALUE"]=> array(2) { ["TEXT"]=> string(2517) "<p> Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. Meanwhile, allogeneic hematopoietic stem cell transplantation (HSCT), by itself, causes frequent organ injury, thus being a potential indication for SOT. Due to increasing numbers of these interventions worldwide, a probability of combined disorders requiring both SOT and HSCT in the same patient is increasing. These transplantation combinations represent some clinical problems. Appropriate risk factors that may affect subsequent SOT outcomes post-HSCT include toxicity of HSCT procedure, immunosuppressive treatment for HSCT patients which may not be optimal for the SOT, potential problems caused by tissue incompatibility between host tissues, hematopoietic graft, and the solid organ graft, and emerging infections. Vice versa, in SOT recipients, the immunosuppressive medication may affect the organ functions, and increase the risk of infections. Moreover, SOT can be associated with development of hematological disorders, such as aplastic anemia, post-transplant lymphoproliferative disease, acute leukemias, etc. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients. Here we discuss the survey which enrolled patients from 107 EBMT centres and clinical data on 45 SOTs carried out in the patients who previously underwent allo-HSCT. Kidney transplantations were performed, mainly, because of the drug-induced organ affection, or radiotherapy, whereas liver transplantations were made either early, for severe VOD or aGvHD, or later, due to chronic liver GVHD or cirrhosis. Survival rates and clinical outcomes are analyzed for each clinical situation in the groups. A special attention is given to the patients who received lung transplants in cases of bronchiolitis obliterans following allo-HSCT, characterized by inferior clinical outcomes and shorter survival time. </p> <p> Some recent data concern allo-HSCT carried out after SOT. The hematological relapse rate was 22 %, thus allowing long-term observations. The 5-year rate for the solid organ failure was about 30%, mostly, due to graft rejection. </p> <p> In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. Infections, graft rejection and other complications are frequent but usually manageable. Also, selected SOT recipients developing hematological disorders may benefit from allo-HSCT. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2481) "
Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. Meanwhile, allogeneic hematopoietic stem cell transplantation (HSCT), by itself, causes frequent organ injury, thus being a potential indication for SOT. Due to increasing numbers of these interventions worldwide, a probability of combined disorders requiring both SOT and HSCT in the same patient is increasing. These transplantation combinations represent some clinical problems. Appropriate risk factors that may affect subsequent SOT outcomes post-HSCT include toxicity of HSCT procedure, immunosuppressive treatment for HSCT patients which may not be optimal for the SOT, potential problems caused by tissue incompatibility between host tissues, hematopoietic graft, and the solid organ graft, and emerging infections. Vice versa, in SOT recipients, the immunosuppressive medication may affect the organ functions, and increase the risk of infections. Moreover, SOT can be associated with development of hematological disorders, such as aplastic anemia, post-transplant lymphoproliferative disease, acute leukemias, etc. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients. Here we discuss the survey which enrolled patients from 107 EBMT centres and clinical data on 45 SOTs carried out in the patients who previously underwent allo-HSCT. Kidney transplantations were performed, mainly, because of the drug-induced organ affection, or radiotherapy, whereas liver transplantations were made either early, for severe VOD or aGvHD, or later, due to chronic liver GVHD or cirrhosis. Survival rates and clinical outcomes are analyzed for each clinical situation in the groups. A special attention is given to the patients who received lung transplants in cases of bronchiolitis obliterans following allo-HSCT, characterized by inferior clinical outcomes and shorter survival time.
Some recent data concern allo-HSCT carried out after SOT. The hematological relapse rate was 22 %, thus allowing long-term observations. The 5-year rate for the solid organ failure was about 30%, mostly, due to graft rejection.
In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. Infections, graft rejection and other complications are frequent but usually manageable. Also, selected SOT recipients developing hematological disorders may benefit from allo-HSCT.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["NAME_EN"]=> array(36) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8149" ["VALUE"]=> string(101) "Allogeneic transplantation of hematopoietic stem cells and solid organ in the same patient: an update" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(101) "Allogeneic transplantation of hematopoietic stem cells and solid organ in the same patient: an update" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" } ["FULL_TEXT_RU"]=> array(36) { ["ID"]=> string(2) "42" ["TIMESTAMP_X"]=> string(19) "2015-09-07 20:29:18" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(23) "Полный текст" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(12) "FULL_TEXT_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "42" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(23) "Полный текст" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["PDF_RU"]=> array(36) { ["ID"]=> string(2) "43" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF RUS" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_RU" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "43" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8150" ["VALUE"]=> string(3) "246" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "246" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF RUS" ["~DEFAULT_VALUE"]=> string(0) "" } ["PDF_EN"]=> array(36) { ["ID"]=> string(2) "44" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF ENG" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "44" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8151" ["VALUE"]=> string(3) "247" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "247" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF ENG" ["~DEFAULT_VALUE"]=> string(0) "" } ["NAME_LONG"]=> array(36) { ["ID"]=> string(2) "45" ["TIMESTAMP_X"]=> string(19) "2023-04-13 00:55:00" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(72) "Название (для очень длинных заголовков)" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "NAME_LONG" ["DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "45" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(80) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(72) "Название (для очень длинных заголовков)" ["~DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } } } ["DISPLAY_PROPERTIES"]=> array(13) { ["AUTHOR_EN"]=> array(37) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8146" ["VALUE"]=> array(2) { ["TEXT"]=> string(12) "Tapani Ruutu" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(12) "Tapani Ruutu" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(12) "Tapani Ruutu" } ["SUMMARY_EN"]=> array(37) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8148" ["VALUE"]=> array(2) { ["TEXT"]=> string(2517) "<p> Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. Meanwhile, allogeneic hematopoietic stem cell transplantation (HSCT), by itself, causes frequent organ injury, thus being a potential indication for SOT. Due to increasing numbers of these interventions worldwide, a probability of combined disorders requiring both SOT and HSCT in the same patient is increasing. These transplantation combinations represent some clinical problems. Appropriate risk factors that may affect subsequent SOT outcomes post-HSCT include toxicity of HSCT procedure, immunosuppressive treatment for HSCT patients which may not be optimal for the SOT, potential problems caused by tissue incompatibility between host tissues, hematopoietic graft, and the solid organ graft, and emerging infections. Vice versa, in SOT recipients, the immunosuppressive medication may affect the organ functions, and increase the risk of infections. Moreover, SOT can be associated with development of hematological disorders, such as aplastic anemia, post-transplant lymphoproliferative disease, acute leukemias, etc. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients. Here we discuss the survey which enrolled patients from 107 EBMT centres and clinical data on 45 SOTs carried out in the patients who previously underwent allo-HSCT. Kidney transplantations were performed, mainly, because of the drug-induced organ affection, or radiotherapy, whereas liver transplantations were made either early, for severe VOD or aGvHD, or later, due to chronic liver GVHD or cirrhosis. Survival rates and clinical outcomes are analyzed for each clinical situation in the groups. A special attention is given to the patients who received lung transplants in cases of bronchiolitis obliterans following allo-HSCT, characterized by inferior clinical outcomes and shorter survival time. </p> <p> Some recent data concern allo-HSCT carried out after SOT. The hematological relapse rate was 22 %, thus allowing long-term observations. The 5-year rate for the solid organ failure was about 30%, mostly, due to graft rejection. </p> <p> In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. Infections, graft rejection and other complications are frequent but usually manageable. Also, selected SOT recipients developing hematological disorders may benefit from allo-HSCT. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2481) "Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. Meanwhile, allogeneic hematopoietic stem cell transplantation (HSCT), by itself, causes frequent organ injury, thus being a potential indication for SOT. Due to increasing numbers of these interventions worldwide, a probability of combined disorders requiring both SOT and HSCT in the same patient is increasing. These transplantation combinations represent some clinical problems. Appropriate risk factors that may affect subsequent SOT outcomes post-HSCT include toxicity of HSCT procedure, immunosuppressive treatment for HSCT patients which may not be optimal for the SOT, potential problems caused by tissue incompatibility between host tissues, hematopoietic graft, and the solid organ graft, and emerging infections. Vice versa, in SOT recipients, the immunosuppressive medication may affect the organ functions, and increase the risk of infections. Moreover, SOT can be associated with development of hematological disorders, such as aplastic anemia, post-transplant lymphoproliferative disease, acute leukemias, etc. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients. Here we discuss the survey which enrolled patients from 107 EBMT centres and clinical data on 45 SOTs carried out in the patients who previously underwent allo-HSCT. Kidney transplantations were performed, mainly, because of the drug-induced organ affection, or radiotherapy, whereas liver transplantations were made either early, for severe VOD or aGvHD, or later, due to chronic liver GVHD or cirrhosis. Survival rates and clinical outcomes are analyzed for each clinical situation in the groups. A special attention is given to the patients who received lung transplants in cases of bronchiolitis obliterans following allo-HSCT, characterized by inferior clinical outcomes and shorter survival time.
Some recent data concern allo-HSCT carried out after SOT. The hematological relapse rate was 22 %, thus allowing long-term observations. The 5-year rate for the solid organ failure was about 30%, mostly, due to graft rejection.
In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. Infections, graft rejection and other complications are frequent but usually manageable. Also, selected SOT recipients developing hematological disorders may benefit from allo-HSCT.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(2481) "Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. Meanwhile, allogeneic hematopoietic stem cell transplantation (HSCT), by itself, causes frequent organ injury, thus being a potential indication for SOT. Due to increasing numbers of these interventions worldwide, a probability of combined disorders requiring both SOT and HSCT in the same patient is increasing. These transplantation combinations represent some clinical problems. Appropriate risk factors that may affect subsequent SOT outcomes post-HSCT include toxicity of HSCT procedure, immunosuppressive treatment for HSCT patients which may not be optimal for the SOT, potential problems caused by tissue incompatibility between host tissues, hematopoietic graft, and the solid organ graft, and emerging infections. Vice versa, in SOT recipients, the immunosuppressive medication may affect the organ functions, and increase the risk of infections. Moreover, SOT can be associated with development of hematological disorders, such as aplastic anemia, post-transplant lymphoproliferative disease, acute leukemias, etc. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients. Here we discuss the survey which enrolled patients from 107 EBMT centres and clinical data on 45 SOTs carried out in the patients who previously underwent allo-HSCT. Kidney transplantations were performed, mainly, because of the drug-induced organ affection, or radiotherapy, whereas liver transplantations were made either early, for severe VOD or aGvHD, or later, due to chronic liver GVHD or cirrhosis. Survival rates and clinical outcomes are analyzed for each clinical situation in the groups. A special attention is given to the patients who received lung transplants in cases of bronchiolitis obliterans following allo-HSCT, characterized by inferior clinical outcomes and shorter survival time.
Some recent data concern allo-HSCT carried out after SOT. The hematological relapse rate was 22 %, thus allowing long-term observations. The 5-year rate for the solid organ failure was about 30%, mostly, due to graft rejection.
In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. Infections, graft rejection and other complications are frequent but usually manageable. Also, selected SOT recipients developing hematological disorders may benefit from allo-HSCT.
" } ["DOI"]=> array(37) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8145" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-15-20" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-15-20" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-15-20" } ["NAME_EN"]=> array(37) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8149" ["VALUE"]=> string(101) "Allogeneic transplantation of hematopoietic stem cells and solid organ in the same patient: an update" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(101) "Allogeneic transplantation of hematopoietic stem cells and solid organ in the same patient: an update" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(101) "Allogeneic transplantation of hematopoietic stem cells and solid organ in the same patient: an update" } ["ORGANIZATION_EN"]=> array(37) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8147" ["VALUE"]=> array(2) { ["TEXT"]=> string(308) "Tapani Ruutu, Helsinki University Hospital, Biomedicum<br> 2C, POB 705, FIN-00029 HUS, Helsinki, Finland<br> <br> Phone: +358 50 555 1279<br> Fax: +358 9 4717 1506<br> E-mail: <a href="mailto:tapani.ruutu@hus.fi">tapani.ruutu@hus.fi</a><br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(250) "Tapani Ruutu, Helsinki University Hospital, Biomedicum2C, POB 705, FIN-00029 HUS, Helsinki, Finland
Phone: +358 50 555 1279
Fax: +358 9 4717 1506
E-mail: tapani.ruutu@hus.fi
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(250) "Tapani Ruutu, Helsinki University Hospital, Biomedicum
2C, POB 705, FIN-00029 HUS, Helsinki, Finland
Phone: +358 50 555 1279
Fax: +358 9 4717 1506
E-mail: tapani.ruutu@hus.fi
" } ["AUTHORS"]=> array(38) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(1) { [0]=> string(4) "8158" } ["VALUE"]=> array(1) { [0]=> string(1) "7" } ["DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(1) { [0]=> string(1) "7" } ["~DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(53) "Tapani Ruutu" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["AUTHOR_RU"]=> array(37) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8142" ["VALUE"]=> array(2) { ["TEXT"]=> string(23) "Тапани Рууту" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(23) "Тапани Рууту" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(23) "Тапани Рууту" } ["SUBMITTED"]=> array(37) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8138" ["VALUE"]=> string(22) "09/21/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "09/21/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "09/21/2016 12:00:00 am" } ["ACCEPTED"]=> array(37) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8139" ["VALUE"]=> string(22) "10/28/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "10/28/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "10/28/2016 12:00:00 am" } ["KEYWORDS"]=> array(38) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(6) { [0]=> string(4) "8152" [1]=> string(4) "8153" [2]=> string(4) "8154" [3]=> string(4) "8155" [4]=> string(4) "8156" [5]=> string(4) "8157" } ["VALUE"]=> array(6) { [0]=> string(3) "105" [1]=> string(3) "298" [2]=> string(3) "659" [3]=> string(3) "660" [4]=> string(2) "18" [5]=> string(3) "661" } ["DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(6) { [0]=> string(3) "105" [1]=> string(3) "298" [2]=> string(3) "659" [3]=> string(3) "660" [4]=> string(2) "18" [5]=> string(3) "661" } ["~DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(6) { [0]=> string(116) "трансплантация гемопоэтических клеток" [1]=> string(72) "трансплантация" [2]=> string(91) "последовательное лечение" [3]=> string(62) "показания" [4]=> string(78) "клинические исходы" [5]=> string(75) "органная функция" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["CONTACT"]=> array(38) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8140" ["VALUE"]=> string(1) "7" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(1) "7" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(53) "Tapani Ruutu" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["SUMMARY_RU"]=> array(37) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8144" ["VALUE"]=> array(2) { ["TEXT"]=> string(4693) "<p>Трансплантация органов (Tx) широко применяется для лечения тяжелой органной недостаточности. Аллогенная трансплантация гемопоэтических клеток (алло-ТГСК), как таковая, часто вызывает повреждение органов, что может быть показанием к Тх. В связи с возрастающим числом обоих видов лечения растет вероятность комбинированных состояний, требующих, как Тх, так и ТГСК у одного и того же пациента. Эти ситуации приводят к ряду клинических проблем. Так, факторами риска, способными повлиять на исходы Тх после ТГСК, могут быть: токсичность процедуры ТГСК, последующее иммуносупрессивное лечение, не адекватное режиму после Тх, возможные проблемы, связанные с тканевой несовместимостью больного, гемопоэтических клеток донора и органного трансплантата, а также развитие инфекций. В то же время иммуносупрессивная терапия больных после Тх может нарушать функции органа и повышать риск инфекций. Кроме того, Тх иногда ассоциирована с развитием гематологических заболеваний, как, например, апластической анемии, посттрансплантационных лимфопролиферативных заболеваний, острых лейкозов и др. Цель настоящей статьи состоит в анализе существующего клинического опыта в области алло-ТГСК и Тх у одних и тех же пациентов. В частности, рассматривается европейское исследование с участием 107 центров EBMT и клинические данные о 45 Тх, проведенных пациентам, которым ранее выполнялась ТГСК. Им проводили, главным образом трансплантацию почек из-за повреждений после химио- или радиотерапии, тогда как пересадки печени делали в ранние сроки после ТГСК в связи с тяжелой вено-окклюзионной болезнью или оРТПХб или позже, по поводу хронической РТПХ или цирроза. Выживаемость и клинические исходы анализировали для каждой клинической ситуации. Особое внимание уделяли больным, получавшим трансплантаты легких при облитерирующем альвеолите после алло-ТГСК, где наблюдались худшие клинические исходы и меньшие сроки выживания.</p> <p>Ряд свежих работ касается алло-ТГСК, проведенных после Тх. Частота гематологических рецидивов составила 22%, что позволило длительно наблюдать пациентов. Пятилетняя выживаемость пересаженных органов была около 30%, в основном – из-за отторжения трансплантата.</p> <p>Таким образом, Тх может быть ценной стратегией при лечении реципиентов гемопоэтических клеток в случае развития органной недостаточности. Инфекции, отторжение трансплантата и другие осложнения довольно часты, но обычно поддаются лечению. Кроме того, некоторые реципиенты органов, у которых развились гематологические заболевания, могут выиграть от алло-ТГСК.</p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(4657) "
Трансплантация органов (Tx) широко применяется для лечения тяжелой органной недостаточности. Аллогенная трансплантация гемопоэтических клеток (алло-ТГСК), как таковая, часто вызывает повреждение органов, что может быть показанием к Тх. В связи с возрастающим числом обоих видов лечения растет вероятность комбинированных состояний, требующих, как Тх, так и ТГСК у одного и того же пациента. Эти ситуации приводят к ряду клинических проблем. Так, факторами риска, способными повлиять на исходы Тх после ТГСК, могут быть: токсичность процедуры ТГСК, последующее иммуносупрессивное лечение, не адекватное режиму после Тх, возможные проблемы, связанные с тканевой несовместимостью больного, гемопоэтических клеток донора и органного трансплантата, а также развитие инфекций. В то же время иммуносупрессивная терапия больных после Тх может нарушать функции органа и повышать риск инфекций. Кроме того, Тх иногда ассоциирована с развитием гематологических заболеваний, как, например, апластической анемии, посттрансплантационных лимфопролиферативных заболеваний, острых лейкозов и др. Цель настоящей статьи состоит в анализе существующего клинического опыта в области алло-ТГСК и Тх у одних и тех же пациентов. В частности, рассматривается европейское исследование с участием 107 центров EBMT и клинические данные о 45 Тх, проведенных пациентам, которым ранее выполнялась ТГСК. Им проводили, главным образом трансплантацию почек из-за повреждений после химио- или радиотерапии, тогда как пересадки печени делали в ранние сроки после ТГСК в связи с тяжелой вено-окклюзионной болезнью или оРТПХб или позже, по поводу хронической РТПХ или цирроза. Выживаемость и клинические исходы анализировали для каждой клинической ситуации. Особое внимание уделяли больным, получавшим трансплантаты легких при облитерирующем альвеолите после алло-ТГСК, где наблюдались худшие клинические исходы и меньшие сроки выживания.
Ряд свежих работ касается алло-ТГСК, проведенных после Тх. Частота гематологических рецидивов составила 22%, что позволило длительно наблюдать пациентов. Пятилетняя выживаемость пересаженных органов была около 30%, в основном – из-за отторжения трансплантата.
Таким образом, Тх может быть ценной стратегией при лечении реципиентов гемопоэтических клеток в случае развития органной недостаточности. Инфекции, отторжение трансплантата и другие осложнения довольно часты, но обычно поддаются лечению. Кроме того, некоторые реципиенты органов, у которых развились гематологические заболевания, могут выиграть от алло-ТГСК.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(4657) "Трансплантация органов (Tx) широко применяется для лечения тяжелой органной недостаточности. Аллогенная трансплантация гемопоэтических клеток (алло-ТГСК), как таковая, часто вызывает повреждение органов, что может быть показанием к Тх. В связи с возрастающим числом обоих видов лечения растет вероятность комбинированных состояний, требующих, как Тх, так и ТГСК у одного и того же пациента. Эти ситуации приводят к ряду клинических проблем. Так, факторами риска, способными повлиять на исходы Тх после ТГСК, могут быть: токсичность процедуры ТГСК, последующее иммуносупрессивное лечение, не адекватное режиму после Тх, возможные проблемы, связанные с тканевой несовместимостью больного, гемопоэтических клеток донора и органного трансплантата, а также развитие инфекций. В то же время иммуносупрессивная терапия больных после Тх может нарушать функции органа и повышать риск инфекций. Кроме того, Тх иногда ассоциирована с развитием гематологических заболеваний, как, например, апластической анемии, посттрансплантационных лимфопролиферативных заболеваний, острых лейкозов и др. Цель настоящей статьи состоит в анализе существующего клинического опыта в области алло-ТГСК и Тх у одних и тех же пациентов. В частности, рассматривается европейское исследование с участием 107 центров EBMT и клинические данные о 45 Тх, проведенных пациентам, которым ранее выполнялась ТГСК. Им проводили, главным образом трансплантацию почек из-за повреждений после химио- или радиотерапии, тогда как пересадки печени делали в ранние сроки после ТГСК в связи с тяжелой вено-окклюзионной болезнью или оРТПХб или позже, по поводу хронической РТПХ или цирроза. Выживаемость и клинические исходы анализировали для каждой клинической ситуации. Особое внимание уделяли больным, получавшим трансплантаты легких при облитерирующем альвеолите после алло-ТГСК, где наблюдались худшие клинические исходы и меньшие сроки выживания.
Ряд свежих работ касается алло-ТГСК, проведенных после Тх. Частота гематологических рецидивов составила 22%, что позволило длительно наблюдать пациентов. Пятилетняя выживаемость пересаженных органов была около 30%, в основном – из-за отторжения трансплантата.
Таким образом, Тх может быть ценной стратегией при лечении реципиентов гемопоэтических клеток в случае развития органной недостаточности. Инфекции, отторжение трансплантата и другие осложнения довольно часты, но обычно поддаются лечению. Кроме того, некоторые реципиенты органов, у которых развились гематологические заболевания, могут выиграть от алло-ТГСК.
" } ["ORGANIZATION_RU"]=> array(37) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8143" ["VALUE"]=> array(2) { ["TEXT"]=> string(155) "Институт клинических исследований, Университетский госпиталь, Хельсинки, Финляндия" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(155) "Институт клинических исследований, Университетский госпиталь, Хельсинки, Финляндия" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(155) "Институт клинических исследований, Университетский госпиталь, Хельсинки, Финляндия" } } } [2]=> array(49) { ["IBLOCK_SECTION_ID"]=> string(2) "20" ["~IBLOCK_SECTION_ID"]=> string(2) "20" ["ID"]=> string(3) "658" ["~ID"]=> string(3) "658" ["IBLOCK_ID"]=> string(1) "2" ["~IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["~NAME"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["ACTIVE_FROM"]=> NULL ["~ACTIVE_FROM"]=> NULL ["TIMESTAMP_X"]=> string(22) "03/29/2017 04:11:01 pm" ["~TIMESTAMP_X"]=> string(22) "03/29/2017 04:11:01 pm" ["DETAIL_PAGE_URL"]=> string(150) "/en/archive/vypusk-5-nomer-4/klinicheskie-raboty/cubklinicheskaya-alteratsiya-tubulyarnogo-epiteliya-i-ostroe-povrezhdenie-pochek-pri-allogennoy-tran/" ["~DETAIL_PAGE_URL"]=> string(150) "/en/archive/vypusk-5-nomer-4/klinicheskie-raboty/cubklinicheskaya-alteratsiya-tubulyarnogo-epiteliya-i-ostroe-povrezhdenie-pochek-pri-allogennoy-tran/" ["LIST_PAGE_URL"]=> string(12) "/en/archive/" ["~LIST_PAGE_URL"]=> string(12) "/en/archive/" ["DETAIL_TEXT"]=> string(16641) "Introduction
Acute kidney injury (AKI), being a common complication of various conditions, is associated with high mortality and became a substantial challenge for modern medicine [23]. AKI is known to be a risk factor for development and progression of chronic kidney disease (CKD) that represents serious medical and social problem [7]. Conventional criteria for AKI diagnostics and staging, according to current clinical practice guidelines, are serum creatinine concentration (PCr) elevation and decline of urine output [2]. These criteria are also used for AKI diagnostics in patients undergoing hematopoietic stem cell transplantation (HSCT) [9,18]. However, fluctuations of PCr and urine output, reflecting development of AKI, are not reliable markers of earlier, potentially reversible stages of tubular damage in patients following HSCT [4, 8, 12]. Noteworthy, a series of molecular events are associated with tubular epithelium damage and subsequent kidney dysfunction. These alterations occur in still viable cells and may be applied either for the evaluation of early subclinical tubular injury or for the prediction of AKI development [3, 19, 22]. Both resident kidney cells and local immune cells are shown to enhance expression of some molecules, which may serve as biomarkers of early organ damage with still normal glomerular filtration rate (GFR) and PCr values [19,20,22]. The efficacy of MBMs evaluation for the prediction of some etiological AKI variants has been already demonstrated [5, 6, 13, 21]. There are, however, only scarce data concerning AKI associated with HSCT [17]. The aims of present study were as follows: a) to define AKI incidence with routine clinical criteria and the occurrence of subclinical tubular injury on the basis of MBMs assessment following HSCT b) to evaluate the efficacy of MBMs measurement for the prediction of AKI.
Patients and Methods
Study design
Ninety HSCT recipients who underwent allogeneic HSCT (46 males, 44 females) were enrolled into the observational prospective study. None of them had a history of renal disorders. Main clinical and demographic parameters of the group are presented in Table 1.
The duration of follow up period was 6 weeks including week 0 (before conditioning and HSCT), and weeks 1 to 5 post-transplant. A weekly clinical examination included the following: arterial blood pressure measurements, diurnal fluid uptake (ΣVfluid), signs of mucositis, clinically significant infections (both local and systemic), acute graft-versus-host disease (aGVHD). Appliance of drugs with known nephrotoxic side effects (chemotherapeutic agents, CNI, antimycotics, antibiotics and antivirals) [16] was also monitored on each week. Over observational period, all the patients underwent routine monitoring of serum creatinine (PCr), alanine aminotransferase (ALT), albumin, lactate dehydrogenase (LDH), hemoglobin (Hb) as well as total RBC and leucocyte counts. AKI assessment and severity stratification was performed according to the KDIGO Guidelines (Kidney Disease Improving Global Outcomes, 2012) [9].
Biomarker assays
Urine samples were collected according to a standard procedure in the morning time, followed by centrifugation at 1500 rpm for 10 min. The supernates were aliquoted and stored at -80 °С until laboratory testing. Concentrations of urinary molecular biomarkers (MBM) were measured weekly in the urine specimens: calbindin, clusterin, IL-18 (interleukin-18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase- π) and MCP-1 (monocyte chemoattractant protein-1). Quantitative determination was performed with customized immunoassay kits using a Bio-Plex 200 analyzer (Bio-Rad Lab, Inc., USA). The MBM concentrations per sample were normalized for serum creatinine level in the given sample. Control group included thirty-three age- and gender-matched healthy volunteers.
Increased MBM level on week 0 was defined as MBM concentration exceeding upper limit of 95% CI for the control group. Elevation of MBM at the weeks 1 to 5 was determined when concentration on subsequent week increased 2-fold or more compared to MBM concentration at week 0, or previous week.
Statistics
Statistical analysis was performed with a licensed software package (SAS 9.4). Each observation time-point was included into analysis as a single case, with exception of censored observations (474 cases in total). For a comparative intergroup analysis, a one way dispersion analysis (ANOVA) was applied. Multiple linear regression was used to find correlations between the MBM values and continuous variables, e.g., Pcr. A multivariate logistic regression analysis was applied to assess predictive value of MBMs in which the MBMs were included as independent variables, along with other potential confounders. The data are presented as a М±SD, median with interquartile ranges [m (25-75%)], mean values, and 95% confidence interval (95% CI) for a mean value, as well as M±SEM. Any intergroup differences or regression coefficients (in multivariate analysis) were considered significant by р < 0.05.
Results
Incidence of AKI cases as assessed by KDIGO criteria (stage 1 to 3), was found to be increased early post-transplant, with a maximum frequency by the week 5. (Fig. 1). In parallel, a regular trend was noted for the PCr increment; however, the 95% CI values still remained within normal limits. Meanwhile, percentage of cases with increased MBM levels in urine sufficiently exceeded the AKI occurrence. The median of simultaneously increased MBMs was 3 (2 to 5) over the total observation period, with only minimal variations at different weeks (Table 2). For the most MBMs, urinary excretion exhibited a several-fold increase post-transplant, as compared to controls. Noteworthy, concentrations of the most MBMs before HSCT were higher than those in the control group (Fig. 2).
Clusterin, MCP-1, and KIM-1 levels showed direct significant and independent correlations with PCr in multiple linear regression models adjusted for other important clinical factors (Table 3). IL-18 excretion negatively correlated with PCr levels registered at the subsequent week. Increase of KIM-1 and/or MCP-1 levels was found to be the independent predictor of AKI at a week following the MBM assessment (Table 4). Relative risk for AKI increased 2.3-fold in case of KIM-1 or MCP-1 elevation, while 3.4-fold increase in RR was found when both MBMs (KIM-1 and MCP-1) were elevated. Other MBMs studied did not have any associations with AKI risk, neither separately, nor in combinations.
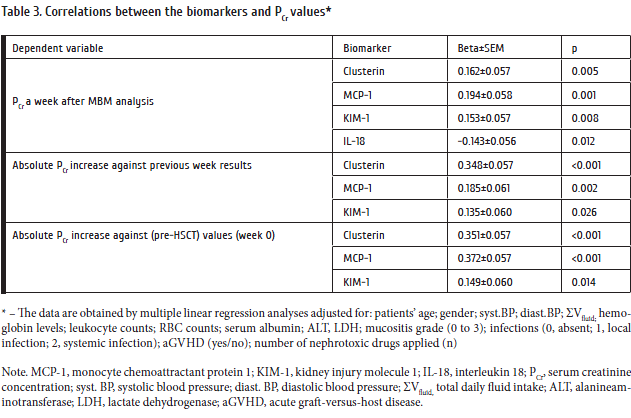
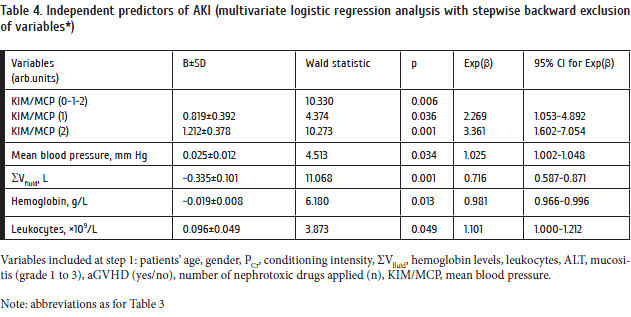
Discussion
AKI is a common complication of an early posttransplant period, which correlates with inferior short- and long-term outcomes of HSCT [11, 15]. A special feature of HSCT-associated renal dysfunction is a simultaneous action of multiple renotoxic factors including (but not limited to) nephrotoxic drugs, alterations of systemic circulation, infections, immune suppression, thus enabling different and complex mechanisms of tubular epithelium damage. Massive infusion therapy, drug-enhanced tubular secretion, body mass losses may contribute to low efficiency of routine AKI criteria in patients following HSCT [11]. Some existing data presume potential clinical value of molecular markers, expressed by resident renal cells and local immune cell populations in certain types of kidney damage [5, 6, 13, 19- 21]. Meanwhile, only scattered publications deal with comparative analysis of different MBMs in AKI following HSCT [17].
Unlike moderate PCr changes, the MBMs under study exhibited a marked increase which clearly preceded the emerging AKI.
Incidence of cases with increased MBMs significantly exceeded the incidence of AKI post-transplant. Moreover, based on these data one can presume that sub-clinical tubular damage affects the majority of patients in the setting of HSCT. Notably, simultaneous elevation of different MBMs found in our study may, probably, reflect different renal responses to multiple damaging factors following HSCT.
The results obtained with MBM panel are definitely in line with basic concept of consequences of acute kidney injury [3, 19]. In particular, this concept allows to discriminate two principal stages of cellular response: (1) an initial stage, when damaged resident tubular epithelial cells retain viability followed by (2) a phase of cellular death and subsequent renal dysfunction, i.e., AKI [19]. Study results have clearly shown that increased MBM levels anticipate Pcr increment and, thus, may define the subclinical kidney damage. Higher occurrence of increased MBMs as compared to increase of Pcr make it clear that tubular epithelial damage not always results in clinically significant renal dysfunction defined as AKI.
One may suggest that the imbalance of various adaptation mechanisms is significant for AKI development. Some of cellular molecular adaptation mechanisms are associated with unfavorable events causing apoptosis/necroptosis, whereas others may induce physiological responses oriented towards cell survival. In particular, increased levels of urinary KIM-1 and МСР-1, being inflammatory molecules, are significantly and independently associated with Pcr changes and higher risk of subsequent AKI. Meanwhile, the overall increase of clusterin, calbindin and GST-π excretion did not correlate with AKI development. One may, therefore, suggest that their higher secretion by viable tubular epithelium could reflect a physiologic response to the damaging factors. Moreover, IL-18 showed even a negative correlation with Pcr, thus pointing to probable association between its up-regulation and protective mechanisms within tubular epithelium in HSCT patients [1, 10].
Hence, we have shown that multiple nephrotoxic effects early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of clinical AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation.
Conflict of interests
No conflict of interests is declared.
The study was performed under financial support of the Committee for Science and Higher School of the St. Petersburg Government and The First St. Petersburg I. Pavlov State Medical University (Contest of innovative research projects – 2015).
References
- Alnasser HA, Guan Q, Zhang F, Gleave ME, Nguan CY, Du C. Requirment of clusterin expression for prosurvival autophagy in hypoxic kidney tubular epithelial cells. Am J Physiol Renal Physiol 2016; 310 (2): F160-73.
- Bellomo R, Kellum JA, Ronco C. Defining acute renal failure: physiological principles. Intensive Care Med 2004, 30 (1): 33-37.
- Basile DP, Anderson MD, Sutton TA. Pathophysiology of acute kidney injury. Compr Physiol. 2012; 2 (2): 1303-1353.
- Coca SG, Parikh CR. Urinary biomarkers for acute kidney injury: perspectives on translation. Clin J Am Soc Nephrol 2008; 3 (2): 481-490.
- Field M, Dronovalli V, Mistry P Drayson M, Ready A, Cobbold M, Inston N. Urinary biomarkers of acute kidney injury in diceased organ donors-kidney injury molecule-1 as an adjunct to predicting outcome. Clin Transplant 2014; 28 (7): 808-815.
- Ghatanatti R, Teli A, Tirkey SS Bhattacharya S, Sengupta G, Mondal A. Role of renal biomarkers as predictors of acute kidney injury in cardiac surgery. Asian Cardiovasc Thorac Ann 2014; 22 (2): 234-241.
- Haase M, Kellum JA, Ronco C. Subclinical AKI - an emerging syndrome with important consequences. Nat Rev Nephrol 2012, 8 (12): 735-739.
- Hjortrup PB, Haase N Wetterslev M, Perner A.Clinical review: Predictive value of neutrophil gelatinase-associated lipocalin for acute kidney injury in intensive care patients. Critical Care. 2013, 17 (2): 211.
- Kellum JA, Aspelin P, Barsoum RS et al. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int (Suppl.) 2012; (2): 1.
- Kuwata K, Nakamura I, Ide M, Sato H, Nishikawa S, Tanaka M. Comparison of changes in urinary and blood levels of biomarkers associated with proximal tubular injury in rat models. J Toxicol Pathol 2015; 28 (3): 151-164.
- Lopes JA, Jorge S Acute kidney injury following HCT: incidence, risk factors and outcome. Bone Marrow Transplant 2011; 46 (11): 1399-1408.
- Martensson J, Martling CR, Bell M Novel Biomarkers of acute kidney injury and failure: clinical applicability. Br J Anaesth 2012; 109 (6): 843-850.
- Menzorov MV, Shutov AM, Makeeva ER, Mikhailova EV, Parfenova EA The role of neutrophil gelatinase-associated lipocalin for the early prediction of acute kidney injury in patients with acute coronary syndrome. Fundamental’nye issledovaniya. 2013; 9 (4): 698-702. (In Russian)
- Metzger J, Kirsch T, Schiffer E Ulger P, Mentes E, Brand K, Weissinger EM, Haubitz M, Mischak H, Herget-Rosenthal S. Urinary excretion of twenty peptides forms an early and accurate diagnostic pattern of acute kidney injury. Kidney Int 2010; 78 (12): 1252-1262.
- Sawinski D. The kidney effects of hematopoietic stem cell transplantation. Adv Chronic Kidney Dis 2014;21(1):96-105.
- Schrier RW, Coffman TM, Falk RJ, Molitoris BA, Neilson EG. In: Schrier’s Diseases of the Kidney 2013; 868-1086.
- Shingai N, Morito T. Urinary liver-type fatty acid-binding protein linked with increased risk of acute kidney injury after allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2014; 20 (12): 2010-2014.
- Smirnov AV, Dobronravov VA, Rumyantsev AS, Shilov EM, Vatazin AV et al. National guidelines acute kidney injury: basic principles of diagnosis, prevention and therapy. Part 1. Nefrologija. 2016, 20 (1): 79-104. (In Russian)
- Smirnov AV, Dobronravov VA, Rumyantsev AS, Kayukov IG. Acute kidney injury. M.: MIA Publishers 2015: 30-79. (In Russian)
- Smirnov AV, Kayukov IG, Dobronravov VA, Kucher AG. Acute kidney injury and acute renal failure: some lessons from international initiatives. Nefrologija. 2008; 12 (3): 7-12. (In Russian)
- Soni SS, Cruz D, Bobek I. NGAL: a biomarker of acute kidney injury and other systemic conditions. Int Urol Nephrol 2010; 42 (1): 141-150.
- Vaidya VS, Ferguson MA, Bonventre JV. Biomarkers of acute kidney injury. Annu Rev Pharmacol Toxicol. 2008; 48: 463-493.
- Zeng X, McMahon GM, Brunelli SM, Bates DW, Waikar SS.Incidence, outcomes, and comparisons across definitions of AKI in hospitalized individuals. Clin J Am Soc Nephrol. 2014, 9 (1): 12-20.
" ["~DETAIL_TEXT"]=> string(16641) "
Introduction
Acute kidney injury (AKI), being a common complication of various conditions, is associated with high mortality and became a substantial challenge for modern medicine [23]. AKI is known to be a risk factor for development and progression of chronic kidney disease (CKD) that represents serious medical and social problem [7]. Conventional criteria for AKI diagnostics and staging, according to current clinical practice guidelines, are serum creatinine concentration (PCr) elevation and decline of urine output [2]. These criteria are also used for AKI diagnostics in patients undergoing hematopoietic stem cell transplantation (HSCT) [9,18]. However, fluctuations of PCr and urine output, reflecting development of AKI, are not reliable markers of earlier, potentially reversible stages of tubular damage in patients following HSCT [4, 8, 12]. Noteworthy, a series of molecular events are associated with tubular epithelium damage and subsequent kidney dysfunction. These alterations occur in still viable cells and may be applied either for the evaluation of early subclinical tubular injury or for the prediction of AKI development [3, 19, 22]. Both resident kidney cells and local immune cells are shown to enhance expression of some molecules, which may serve as biomarkers of early organ damage with still normal glomerular filtration rate (GFR) and PCr values [19,20,22]. The efficacy of MBMs evaluation for the prediction of some etiological AKI variants has been already demonstrated [5, 6, 13, 21]. There are, however, only scarce data concerning AKI associated with HSCT [17]. The aims of present study were as follows: a) to define AKI incidence with routine clinical criteria and the occurrence of subclinical tubular injury on the basis of MBMs assessment following HSCT b) to evaluate the efficacy of MBMs measurement for the prediction of AKI.
Patients and Methods
Study design
Ninety HSCT recipients who underwent allogeneic HSCT (46 males, 44 females) were enrolled into the observational prospective study. None of them had a history of renal disorders. Main clinical and demographic parameters of the group are presented in Table 1.

The duration of follow up period was 6 weeks including week 0 (before conditioning and HSCT), and weeks 1 to 5 post-transplant. A weekly clinical examination included the following: arterial blood pressure measurements, diurnal fluid uptake (ΣVfluid), signs of mucositis, clinically significant infections (both local and systemic), acute graft-versus-host disease (aGVHD). Appliance of drugs with known nephrotoxic side effects (chemotherapeutic agents, CNI, antimycotics, antibiotics and antivirals) [16] was also monitored on each week. Over observational period, all the patients underwent routine monitoring of serum creatinine (PCr), alanine aminotransferase (ALT), albumin, lactate dehydrogenase (LDH), hemoglobin (Hb) as well as total RBC and leucocyte counts. AKI assessment and severity stratification was performed according to the KDIGO Guidelines (Kidney Disease Improving Global Outcomes, 2012) [9].
Biomarker assays
Urine samples were collected according to a standard procedure in the morning time, followed by centrifugation at 1500 rpm for 10 min. The supernates were aliquoted and stored at -80 °С until laboratory testing. Concentrations of urinary molecular biomarkers (MBM) were measured weekly in the urine specimens: calbindin, clusterin, IL-18 (interleukin-18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase- π) and MCP-1 (monocyte chemoattractant protein-1). Quantitative determination was performed with customized immunoassay kits using a Bio-Plex 200 analyzer (Bio-Rad Lab, Inc., USA). The MBM concentrations per sample were normalized for serum creatinine level in the given sample. Control group included thirty-three age- and gender-matched healthy volunteers.
Increased MBM level on week 0 was defined as MBM concentration exceeding upper limit of 95% CI for the control group. Elevation of MBM at the weeks 1 to 5 was determined when concentration on subsequent week increased 2-fold or more compared to MBM concentration at week 0, or previous week.
Statistics
Statistical analysis was performed with a licensed software package (SAS 9.4). Each observation time-point was included into analysis as a single case, with exception of censored observations (474 cases in total). For a comparative intergroup analysis, a one way dispersion analysis (ANOVA) was applied. Multiple linear regression was used to find correlations between the MBM values and continuous variables, e.g., Pcr. A multivariate logistic regression analysis was applied to assess predictive value of MBMs in which the MBMs were included as independent variables, along with other potential confounders. The data are presented as a М±SD, median with interquartile ranges [m (25-75%)], mean values, and 95% confidence interval (95% CI) for a mean value, as well as M±SEM. Any intergroup differences or regression coefficients (in multivariate analysis) were considered significant by р < 0.05.
Results
Incidence of AKI cases as assessed by KDIGO criteria (stage 1 to 3), was found to be increased early post-transplant, with a maximum frequency by the week 5. (Fig. 1). In parallel, a regular trend was noted for the PCr increment; however, the 95% CI values still remained within normal limits. Meanwhile, percentage of cases with increased MBM levels in urine sufficiently exceeded the AKI occurrence. The median of simultaneously increased MBMs was 3 (2 to 5) over the total observation period, with only minimal variations at different weeks (Table 2). For the most MBMs, urinary excretion exhibited a several-fold increase post-transplant, as compared to controls. Noteworthy, concentrations of the most MBMs before HSCT were higher than those in the control group (Fig. 2).


Clusterin, MCP-1, and KIM-1 levels showed direct significant and independent correlations with PCr in multiple linear regression models adjusted for other important clinical factors (Table 3). IL-18 excretion negatively correlated with PCr levels registered at the subsequent week. Increase of KIM-1 and/or MCP-1 levels was found to be the independent predictor of AKI at a week following the MBM assessment (Table 4). Relative risk for AKI increased 2.3-fold in case of KIM-1 or MCP-1 elevation, while 3.4-fold increase in RR was found when both MBMs (KIM-1 and MCP-1) were elevated. Other MBMs studied did not have any associations with AKI risk, neither separately, nor in combinations.


Discussion
AKI is a common complication of an early posttransplant period, which correlates with inferior short- and long-term outcomes of HSCT [11, 15]. A special feature of HSCT-associated renal dysfunction is a simultaneous action of multiple renotoxic factors including (but not limited to) nephrotoxic drugs, alterations of systemic circulation, infections, immune suppression, thus enabling different and complex mechanisms of tubular epithelium damage. Massive infusion therapy, drug-enhanced tubular secretion, body mass losses may contribute to low efficiency of routine AKI criteria in patients following HSCT [11]. Some existing data presume potential clinical value of molecular markers, expressed by resident renal cells and local immune cell populations in certain types of kidney damage [5, 6, 13, 19- 21]. Meanwhile, only scattered publications deal with comparative analysis of different MBMs in AKI following HSCT [17].
Unlike moderate PCr changes, the MBMs under study exhibited a marked increase which clearly preceded the emerging AKI.
Incidence of cases with increased MBMs significantly exceeded the incidence of AKI post-transplant. Moreover, based on these data one can presume that sub-clinical tubular damage affects the majority of patients in the setting of HSCT. Notably, simultaneous elevation of different MBMs found in our study may, probably, reflect different renal responses to multiple damaging factors following HSCT.
The results obtained with MBM panel are definitely in line with basic concept of consequences of acute kidney injury [3, 19]. In particular, this concept allows to discriminate two principal stages of cellular response: (1) an initial stage, when damaged resident tubular epithelial cells retain viability followed by (2) a phase of cellular death and subsequent renal dysfunction, i.e., AKI [19]. Study results have clearly shown that increased MBM levels anticipate Pcr increment and, thus, may define the subclinical kidney damage. Higher occurrence of increased MBMs as compared to increase of Pcr make it clear that tubular epithelial damage not always results in clinically significant renal dysfunction defined as AKI.
One may suggest that the imbalance of various adaptation mechanisms is significant for AKI development. Some of cellular molecular adaptation mechanisms are associated with unfavorable events causing apoptosis/necroptosis, whereas others may induce physiological responses oriented towards cell survival. In particular, increased levels of urinary KIM-1 and МСР-1, being inflammatory molecules, are significantly and independently associated with Pcr changes and higher risk of subsequent AKI. Meanwhile, the overall increase of clusterin, calbindin and GST-π excretion did not correlate with AKI development. One may, therefore, suggest that their higher secretion by viable tubular epithelium could reflect a physiologic response to the damaging factors. Moreover, IL-18 showed even a negative correlation with Pcr, thus pointing to probable association between its up-regulation and protective mechanisms within tubular epithelium in HSCT patients [1, 10].
Hence, we have shown that multiple nephrotoxic effects early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of clinical AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation.
Conflict of interests
No conflict of interests is declared.
The study was performed under financial support of the Committee for Science and Higher School of the St. Petersburg Government and The First St. Petersburg I. Pavlov State Medical University (Contest of innovative research projects – 2015).
References
- Alnasser HA, Guan Q, Zhang F, Gleave ME, Nguan CY, Du C. Requirment of clusterin expression for prosurvival autophagy in hypoxic kidney tubular epithelial cells. Am J Physiol Renal Physiol 2016; 310 (2): F160-73.
- Bellomo R, Kellum JA, Ronco C. Defining acute renal failure: physiological principles. Intensive Care Med 2004, 30 (1): 33-37.
- Basile DP, Anderson MD, Sutton TA. Pathophysiology of acute kidney injury. Compr Physiol. 2012; 2 (2): 1303-1353.
- Coca SG, Parikh CR. Urinary biomarkers for acute kidney injury: perspectives on translation. Clin J Am Soc Nephrol 2008; 3 (2): 481-490.
- Field M, Dronovalli V, Mistry P Drayson M, Ready A, Cobbold M, Inston N. Urinary biomarkers of acute kidney injury in diceased organ donors-kidney injury molecule-1 as an adjunct to predicting outcome. Clin Transplant 2014; 28 (7): 808-815.
- Ghatanatti R, Teli A, Tirkey SS Bhattacharya S, Sengupta G, Mondal A. Role of renal biomarkers as predictors of acute kidney injury in cardiac surgery. Asian Cardiovasc Thorac Ann 2014; 22 (2): 234-241.
- Haase M, Kellum JA, Ronco C. Subclinical AKI - an emerging syndrome with important consequences. Nat Rev Nephrol 2012, 8 (12): 735-739.
- Hjortrup PB, Haase N Wetterslev M, Perner A.Clinical review: Predictive value of neutrophil gelatinase-associated lipocalin for acute kidney injury in intensive care patients. Critical Care. 2013, 17 (2): 211.
- Kellum JA, Aspelin P, Barsoum RS et al. KDIGO Clinical Practice Guideline for Acute Kidney Injury. Kidney Int (Suppl.) 2012; (2): 1.
- Kuwata K, Nakamura I, Ide M, Sato H, Nishikawa S, Tanaka M. Comparison of changes in urinary and blood levels of biomarkers associated with proximal tubular injury in rat models. J Toxicol Pathol 2015; 28 (3): 151-164.
- Lopes JA, Jorge S Acute kidney injury following HCT: incidence, risk factors and outcome. Bone Marrow Transplant 2011; 46 (11): 1399-1408.
- Martensson J, Martling CR, Bell M Novel Biomarkers of acute kidney injury and failure: clinical applicability. Br J Anaesth 2012; 109 (6): 843-850.
- Menzorov MV, Shutov AM, Makeeva ER, Mikhailova EV, Parfenova EA The role of neutrophil gelatinase-associated lipocalin for the early prediction of acute kidney injury in patients with acute coronary syndrome. Fundamental’nye issledovaniya. 2013; 9 (4): 698-702. (In Russian)
- Metzger J, Kirsch T, Schiffer E Ulger P, Mentes E, Brand K, Weissinger EM, Haubitz M, Mischak H, Herget-Rosenthal S. Urinary excretion of twenty peptides forms an early and accurate diagnostic pattern of acute kidney injury. Kidney Int 2010; 78 (12): 1252-1262.
- Sawinski D. The kidney effects of hematopoietic stem cell transplantation. Adv Chronic Kidney Dis 2014;21(1):96-105.
- Schrier RW, Coffman TM, Falk RJ, Molitoris BA, Neilson EG. In: Schrier’s Diseases of the Kidney 2013; 868-1086.
- Shingai N, Morito T. Urinary liver-type fatty acid-binding protein linked with increased risk of acute kidney injury after allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2014; 20 (12): 2010-2014.
- Smirnov AV, Dobronravov VA, Rumyantsev AS, Shilov EM, Vatazin AV et al. National guidelines acute kidney injury: basic principles of diagnosis, prevention and therapy. Part 1. Nefrologija. 2016, 20 (1): 79-104. (In Russian)
- Smirnov AV, Dobronravov VA, Rumyantsev AS, Kayukov IG. Acute kidney injury. M.: MIA Publishers 2015: 30-79. (In Russian)
- Smirnov AV, Kayukov IG, Dobronravov VA, Kucher AG. Acute kidney injury and acute renal failure: some lessons from international initiatives. Nefrologija. 2008; 12 (3): 7-12. (In Russian)
- Soni SS, Cruz D, Bobek I. NGAL: a biomarker of acute kidney injury and other systemic conditions. Int Urol Nephrol 2010; 42 (1): 141-150.
- Vaidya VS, Ferguson MA, Bonventre JV. Biomarkers of acute kidney injury. Annu Rev Pharmacol Toxicol. 2008; 48: 463-493.
- Zeng X, McMahon GM, Brunelli SM, Bates DW, Waikar SS.Incidence, outcomes, and comparisons across definitions of AKI in hospitalized individuals. Clin J Am Soc Nephrol. 2014, 9 (1): 12-20.
" ["DETAIL_TEXT_TYPE"]=> string(4) "html" ["~DETAIL_TEXT_TYPE"]=> string(4) "html" ["PREVIEW_TEXT"]=> string(0) "" ["~PREVIEW_TEXT"]=> string(0) "" ["PREVIEW_TEXT_TYPE"]=> string(4) "text" ["~PREVIEW_TEXT_TYPE"]=> string(4) "text" ["PREVIEW_PICTURE"]=> NULL ["~PREVIEW_PICTURE"]=> NULL ["LANG_DIR"]=> string(4) "/ru/" ["~LANG_DIR"]=> string(4) "/ru/" ["SORT"]=> string(3) "500" ["~SORT"]=> string(3) "500" ["CODE"]=> string(100) "cubklinicheskaya-alteratsiya-tubulyarnogo-epiteliya-i-ostroe-povrezhdenie-pochek-pri-allogennoy-tran" ["~CODE"]=> string(100) "cubklinicheskaya-alteratsiya-tubulyarnogo-epiteliya-i-ostroe-povrezhdenie-pochek-pri-allogennoy-tran" ["EXTERNAL_ID"]=> string(3) "658" ["~EXTERNAL_ID"]=> string(3) "658" ["IBLOCK_TYPE_ID"]=> string(7) "journal" ["~IBLOCK_TYPE_ID"]=> string(7) "journal" ["IBLOCK_CODE"]=> string(7) "volumes" ["~IBLOCK_CODE"]=> string(7) "volumes" ["IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["~IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["LID"]=> string(2) "s2" ["~LID"]=> string(2) "s2" ["EDIT_LINK"]=> NULL ["DELETE_LINK"]=> NULL ["DISPLAY_ACTIVE_FROM"]=> string(0) "" ["IPROPERTY_VALUES"]=> array(18) { ["ELEMENT_META_TITLE"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["ELEMENT_META_KEYWORDS"]=> string(159) "Острое повреждение почек трансплантация гемопоэтических стволовых клеток биомаркеры" ["ELEMENT_META_DESCRIPTION"]=> string(380) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клетокSubclinical tubular epithelium damage and acute kidney injury following allogeneic hematopoietic stem cell transplantation" ["ELEMENT_PREVIEW_PICTURE_FILE_ALT"]=> string(3641) "<h3>ЦЕЛЬ ИССЛЕДОВАНИЯ</h3> <p>определение значения молекулярных биомаркеров (МБМ), ассоциированных с альтерацией клеток тубулярного эпителия, в прогнозировании острого повреждения почек после трансплантации гемопоэтических стволовых клеток (ТГСК).</p> <h3>ПАЦИЕНТЫ И МЕТОДЫ </h3> <p>В открытое обзервационное проспективное исследование включены 90 больных (46 мужчин и 44 женщины), которым была выполнена ТГСК. В образцах мочи до ТГСК и на пяти первых неделях раннего посттрансплантационного периода определены концентрации МБМ (кальбиндина, кластерина, интерлейкина-18 (IL-18), молекулы повреждения почек-1 (KIM-1), глютатион-S-трансферазы (π-класс) (GST-π), протеина хемотаксиса моноцитов-1 (MCP-1). Параллельно мониторировали основные клинические показатели. Диагностику и стратификацию тяжести острого повреждения почек (ОПП) проводили согласно рекомендациям KDIGO (Kidney Disease Improving Global Outcomes).</p> <h3>РЕЗУЛЬТАТЫ</h3> <p>Доля случаев с ОПП прогрессивно увеличивалась в раннем посттрансплантационном периоде, достигнув максимума к неделе 5 после ТГСК. Повышение содержания МБМ выявляли существенно чаще случаев ОПП, причем повышение МБМ предшествовало формированию дисфункции почек (ОПП). При множественном регрессионном анализе кластерин, MCP-1 и KIM-1 имели прямую, достоверную, независимую от других анализируемых факторов связь с креатинином сыворотки крови на неделе, следующей за определением МБМ. Повышение мочевой экскреции KIM-1 и (или) MCP-1 было независимо от других клинических факторов ассоциировано с увеличением относительного риска (ОР) развития ОПП.</p> <h3>ЗАКЛЮЧЕНИЕ</h3> <p>Множественные ренотоксичные воздействия при ТГСК приводят к существенному и одновременному повышению экскреции с мочой БМ тубулярного повреждения, отражающему субклиническое повреждение клеток тубулярного эпителия в отсутствии критериев ОПП; оценка мочевой экскреции KIM-1 и MCP-1 представляется наиболее подходящим методом предиктивной диагностики ОПП ассоциированного с ТГСК.</p>" ["ELEMENT_PREVIEW_PICTURE_FILE_TITLE"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["ELEMENT_DETAIL_PICTURE_FILE_ALT"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["ELEMENT_DETAIL_PICTURE_FILE_TITLE"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["SECTION_META_TITLE"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["SECTION_META_KEYWORDS"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["SECTION_META_DESCRIPTION"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["SECTION_PICTURE_FILE_ALT"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["SECTION_PICTURE_FILE_TITLE"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["SECTION_PICTURE_FILE_NAME"]=> string(100) "cubklinicheskaya-alteratsiya-tubulyarnogo-epiteliya-i-ostroe-povrezhdenie-pochek-pri-allogennoy-tran" ["SECTION_DETAIL_PICTURE_FILE_ALT"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["SECTION_DETAIL_PICTURE_FILE_TITLE"]=> string(258) "Cубклиническая альтерация тубулярного эпителия и острое повреждение почек при аллогенной трансплантации гемопоэтических стволовых клеток" ["SECTION_DETAIL_PICTURE_FILE_NAME"]=> string(100) "cubklinicheskaya-alteratsiya-tubulyarnogo-epiteliya-i-ostroe-povrezhdenie-pochek-pri-allogennoy-tran" ["ELEMENT_PREVIEW_PICTURE_FILE_NAME"]=> string(100) "cubklinicheskaya-alteratsiya-tubulyarnogo-epiteliya-i-ostroe-povrezhdenie-pochek-pri-allogennoy-tran" ["ELEMENT_DETAIL_PICTURE_FILE_NAME"]=> string(100) "cubklinicheskaya-alteratsiya-tubulyarnogo-epiteliya-i-ostroe-povrezhdenie-pochek-pri-allogennoy-tran" } ["FIELDS"]=> array(1) { ["IBLOCK_SECTION_ID"]=> string(2) "20" } ["PROPERTIES"]=> array(18) { ["KEYWORDS"]=> array(36) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(3) { [0]=> string(4) "8120" [1]=> string(4) "8121" [2]=> string(4) "8122" } ["VALUE"]=> array(3) { [0]=> string(3) "655" [1]=> string(2) "15" [2]=> string(3) "657" } ["DESCRIPTION"]=> array(3) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(3) { [0]=> string(3) "655" [1]=> string(2) "15" [2]=> string(3) "657" } ["~DESCRIPTION"]=> array(3) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" } ["SUBMITTED"]=> array(36) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8000" ["VALUE"]=> string(22) "11/10/2016 02:55:00 pm" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/10/2016 02:55:00 pm" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL } ["ACCEPTED"]=> array(36) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8001" ["VALUE"]=> string(22) "12/09/2016 02:55:00 pm" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "12/09/2016 02:55:00 pm" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL } ["PUBLISHED"]=> array(36) { ["ID"]=> string(2) "22" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Дата публикации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "PUBLISHED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "22" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Дата публикации" ["~DEFAULT_VALUE"]=> NULL } ["CONTACT"]=> array(36) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8002" ["VALUE"]=> string(3) "390" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "390" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHORS"]=> array(36) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(4) { [0]=> string(4) "8123" [1]=> string(4) "8124" [2]=> string(4) "8125" [3]=> string(4) "8126" } ["VALUE"]=> array(4) { [0]=> string(3) "390" [1]=> string(3) "389" [2]=> string(3) "654" [3]=> string(3) "391" } ["DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(4) { [0]=> string(3) "390" [1]=> string(3) "389" [2]=> string(3) "654" [3]=> string(3) "391" } ["~DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_RU"]=> array(36) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8007" ["VALUE"]=> array(2) { ["TEXT"]=> string(306) "Владимир А. Добронравов<sup>1,2</sup>, Кирилл А. Смирнов<sup>1,2</sup>, Борис В. Афанасьев<sup>1,3</sup>, Ольга В. Галкина<sup>1,2</sup>, Алексей В. Смирнов<sup>1</sup>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(246) "Владимир А. Добронравов1,2, Кирилл А. Смирнов1,2, Борис В. Афанасьев1,3, Ольга В. Галкина1,2, Алексей В. Смирнов1" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_RU"]=> array(36) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8008" ["VALUE"]=> array(2) { ["TEXT"]=> string(558) "<sup>1</sup>Первый Санкт-Петербургский государственный медицинский университет им. акад. И. П. Павлова, Россия <br> <sup>2</sup>Научно-исследовательский институт нефрологии <br> <sup>3</sup>Научно-исследовательский институт детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(510) "1Первый Санкт-Петербургский государственный медицинский университет им. акад. И. П. Павлова, Россия
2Научно-исследовательский институт нефрологии
3Научно-исследовательский институт детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_RU"]=> array(36) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8009" ["VALUE"]=> array(2) { ["TEXT"]=> string(3641) "<h3>ЦЕЛЬ ИССЛЕДОВАНИЯ</h3> <p>определение значения молекулярных биомаркеров (МБМ), ассоциированных с альтерацией клеток тубулярного эпителия, в прогнозировании острого повреждения почек после трансплантации гемопоэтических стволовых клеток (ТГСК).</p> <h3>ПАЦИЕНТЫ И МЕТОДЫ </h3> <p>В открытое обзервационное проспективное исследование включены 90 больных (46 мужчин и 44 женщины), которым была выполнена ТГСК. В образцах мочи до ТГСК и на пяти первых неделях раннего посттрансплантационного периода определены концентрации МБМ (кальбиндина, кластерина, интерлейкина-18 (IL-18), молекулы повреждения почек-1 (KIM-1), глютатион-S-трансферазы (π-класс) (GST-π), протеина хемотаксиса моноцитов-1 (MCP-1). Параллельно мониторировали основные клинические показатели. Диагностику и стратификацию тяжести острого повреждения почек (ОПП) проводили согласно рекомендациям KDIGO (Kidney Disease Improving Global Outcomes).</p> <h3>РЕЗУЛЬТАТЫ</h3> <p>Доля случаев с ОПП прогрессивно увеличивалась в раннем посттрансплантационном периоде, достигнув максимума к неделе 5 после ТГСК. Повышение содержания МБМ выявляли существенно чаще случаев ОПП, причем повышение МБМ предшествовало формированию дисфункции почек (ОПП). При множественном регрессионном анализе кластерин, MCP-1 и KIM-1 имели прямую, достоверную, независимую от других анализируемых факторов связь с креатинином сыворотки крови на неделе, следующей за определением МБМ. Повышение мочевой экскреции KIM-1 и (или) MCP-1 было независимо от других клинических факторов ассоциировано с увеличением относительного риска (ОР) развития ОПП.</p> <h3>ЗАКЛЮЧЕНИЕ</h3> <p>Множественные ренотоксичные воздействия при ТГСК приводят к существенному и одновременному повышению экскреции с мочой БМ тубулярного повреждения, отражающему субклиническое повреждение клеток тубулярного эпителия в отсутствии критериев ОПП; оценка мочевой экскреции KIM-1 и MCP-1 представляется наиболее подходящим методом предиктивной диагностики ОПП ассоциированного с ТГСК.</p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(3545) "
ЦЕЛЬ ИССЛЕДОВАНИЯ
определение значения молекулярных биомаркеров (МБМ), ассоциированных с альтерацией клеток тубулярного эпителия, в прогнозировании острого повреждения почек после трансплантации гемопоэтических стволовых клеток (ТГСК).
ПАЦИЕНТЫ И МЕТОДЫ
В открытое обзервационное проспективное исследование включены 90 больных (46 мужчин и 44 женщины), которым была выполнена ТГСК. В образцах мочи до ТГСК и на пяти первых неделях раннего посттрансплантационного периода определены концентрации МБМ (кальбиндина, кластерина, интерлейкина-18 (IL-18), молекулы повреждения почек-1 (KIM-1), глютатион-S-трансферазы (π-класс) (GST-π), протеина хемотаксиса моноцитов-1 (MCP-1). Параллельно мониторировали основные клинические показатели. Диагностику и стратификацию тяжести острого повреждения почек (ОПП) проводили согласно рекомендациям KDIGO (Kidney Disease Improving Global Outcomes).
РЕЗУЛЬТАТЫ
Доля случаев с ОПП прогрессивно увеличивалась в раннем посттрансплантационном периоде, достигнув максимума к неделе 5 после ТГСК. Повышение содержания МБМ выявляли существенно чаще случаев ОПП, причем повышение МБМ предшествовало формированию дисфункции почек (ОПП). При множественном регрессионном анализе кластерин, MCP-1 и KIM-1 имели прямую, достоверную, независимую от других анализируемых факторов связь с креатинином сыворотки крови на неделе, следующей за определением МБМ. Повышение мочевой экскреции KIM-1 и (или) MCP-1 было независимо от других клинических факторов ассоциировано с увеличением относительного риска (ОР) развития ОПП.
ЗАКЛЮЧЕНИЕ
Множественные ренотоксичные воздействия при ТГСК приводят к существенному и одновременному повышению экскреции с мочой БМ тубулярного повреждения, отражающему субклиническое повреждение клеток тубулярного эпителия в отсутствии критериев ОПП; оценка мочевой экскреции KIM-1 и MCP-1 представляется наиболее подходящим методом предиктивной диагностики ОПП ассоциированного с ТГСК.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["DOI"]=> array(36) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8010" ["VALUE"]=> string(36) "10.18620/ctt-1866-8836-2016-5-4-8-14" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(36) "10.18620/ctt-1866-8836-2016-5-4-8-14" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_EN"]=> array(36) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8011" ["VALUE"]=> array(2) { ["TEXT"]=> string(233) "Vladimir A. Dobronravov <sup>1,2</sup>, Kirill A. Smirnov <sup>1,2</sup>, Boris V. Afanasiev <sup>1,3</sup>, Olga V. Galkina <sup>1,2</sup>, Alexey V. Smirnov <sup>1,2</sup>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(173) "Vladimir A. Dobronravov 1,2, Kirill A. Smirnov 1,2, Boris V. Afanasiev 1,3, Olga V. Galkina 1,2, Alexey V. Smirnov 1,2" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_EN"]=> array(36) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8012" ["VALUE"]=> array(2) { ["TEXT"]=> string(557) "Prof. Vladimir A. Dobronravov, MD, PhD, DSc, Research Institute of Nephrology, The First St. Petersburg I. Pavlov State<br> Medical University, Russia, L.Tolstoy St. 17, 197089, St. Petersburg, Russia<br> <br> <sup>1</sup>First St. Petersburg I. Pavlov State Medical University, Russia,<br> <sup>2</sup>Nephrology Research Institute,<br> <sup>3</sup>Raisa Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(485) "Prof. Vladimir A. Dobronravov, MD, PhD, DSc, Research Institute of Nephrology, The First St. Petersburg I. Pavlov StateMedical University, Russia, L.Tolstoy St. 17, 197089, St. Petersburg, Russia
1First St. Petersburg I. Pavlov State Medical University, Russia,
2Nephrology Research Institute,
3Raisa Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_EN"]=> array(36) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8049" ["VALUE"]=> array(2) { ["TEXT"]=> string(1814) "<p> The aim of this study was to determine clinical value of molecular biomarkers (MBM), associated with tubular epithelium damage, for the prediction of acute kidney injury (AKI) in the setting of hematopoietic stem cell transplantation (HSCT). </p> <h3>Patients and methods</h3> <p> Ninety HSCT recipients (46 males, 44 females) were enrolled into the observational prospective study. Urinary concentrations of calbindin, clusterin, IL-18 (interleukin- 18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase-π) and MCP-1 (monocyte chemoattractant protein-1) were measured in all patients before HSCT and at 5 consequent time points during early post-transplant period, along with routine clinical monitoring. AKI was defined according to the KDIGO (Kidney Disease Improving Global Outcomes) Guidelines. </p> <h3>Results</h3> <p> The incidence of AKI cases constantly increased during the observation period and reached the maximum level by the week 5 following HSCT. MBM elevation was observed more frequently than AKI and preceded the latter. Clusterin, MCP-1 and KIM-1 levels significantly correlated with subsequent serum creatinine values, measured a week after the MBM’s analysis according to multivariate linear regression models adjusted for other confounders. An increase in KIM-1 and/or MCP-1 urinary excretion was independently associated with a relative risk of AKI development. In summary, multiple renotoxic events early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(1754) "
The aim of this study was to determine clinical value of molecular biomarkers (MBM), associated with tubular epithelium damage, for the prediction of acute kidney injury (AKI) in the setting of hematopoietic stem cell transplantation (HSCT).
Patients and methods
Ninety HSCT recipients (46 males, 44 females) were enrolled into the observational prospective study. Urinary concentrations of calbindin, clusterin, IL-18 (interleukin- 18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase-π) and MCP-1 (monocyte chemoattractant protein-1) were measured in all patients before HSCT and at 5 consequent time points during early post-transplant period, along with routine clinical monitoring. AKI was defined according to the KDIGO (Kidney Disease Improving Global Outcomes) Guidelines.
Results
The incidence of AKI cases constantly increased during the observation period and reached the maximum level by the week 5 following HSCT. MBM elevation was observed more frequently than AKI and preceded the latter. Clusterin, MCP-1 and KIM-1 levels significantly correlated with subsequent serum creatinine values, measured a week after the MBM’s analysis according to multivariate linear regression models adjusted for other confounders. An increase in KIM-1 and/or MCP-1 urinary excretion was independently associated with a relative risk of AKI development. In summary, multiple renotoxic events early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["NAME_EN"]=> array(36) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8013" ["VALUE"]=> string(122) "Subclinical tubular epithelium damage and acute kidney injury following allogeneic hematopoietic stem cell transplantation" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(122) "Subclinical tubular epithelium damage and acute kidney injury following allogeneic hematopoietic stem cell transplantation" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" } ["FULL_TEXT_RU"]=> &array(36) { ["ID"]=> string(2) "42" ["TIMESTAMP_X"]=> string(19) "2015-09-07 20:29:18" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(23) "Полный текст" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(12) "FULL_TEXT_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "42" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(23) "Полный текст" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["PDF_RU"]=> array(36) { ["ID"]=> string(2) "43" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF RUS" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_RU" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "43" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8127" ["VALUE"]=> string(3) "244" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "244" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(7) "PDF RUS" ["~DEFAULT_VALUE"]=> string(0) "" } ["PDF_EN"]=> array(36) { ["ID"]=> string(2) "44" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF ENG" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "44" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8128" ["VALUE"]=> string(3) "245" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "245" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(7) "PDF ENG" ["~DEFAULT_VALUE"]=> string(0) "" } ["NAME_LONG"]=> array(36) { ["ID"]=> string(2) "45" ["TIMESTAMP_X"]=> string(19) "2023-04-13 00:55:00" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(72) "Название (для очень длинных заголовков)" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "NAME_LONG" ["DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "45" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(80) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(72) "Название (для очень длинных заголовков)" ["~DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } } } ["DISPLAY_PROPERTIES"]=> array(13) { ["AUTHOR_EN"]=> array(37) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8011" ["VALUE"]=> array(2) { ["TEXT"]=> string(233) "Vladimir A. Dobronravov <sup>1,2</sup>, Kirill A. Smirnov <sup>1,2</sup>, Boris V. Afanasiev <sup>1,3</sup>, Olga V. Galkina <sup>1,2</sup>, Alexey V. Smirnov <sup>1,2</sup>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(173) "Vladimir A. Dobronravov 1,2, Kirill A. Smirnov 1,2, Boris V. Afanasiev 1,3, Olga V. Galkina 1,2, Alexey V. Smirnov 1,2" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(173) "Vladimir A. Dobronravov 1,2, Kirill A. Smirnov 1,2, Boris V. Afanasiev 1,3, Olga V. Galkina 1,2, Alexey V. Smirnov 1,2" } ["SUMMARY_EN"]=> array(37) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8049" ["VALUE"]=> array(2) { ["TEXT"]=> string(1814) "<p> The aim of this study was to determine clinical value of molecular biomarkers (MBM), associated with tubular epithelium damage, for the prediction of acute kidney injury (AKI) in the setting of hematopoietic stem cell transplantation (HSCT). </p> <h3>Patients and methods</h3> <p> Ninety HSCT recipients (46 males, 44 females) were enrolled into the observational prospective study. Urinary concentrations of calbindin, clusterin, IL-18 (interleukin- 18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase-π) and MCP-1 (monocyte chemoattractant protein-1) were measured in all patients before HSCT and at 5 consequent time points during early post-transplant period, along with routine clinical monitoring. AKI was defined according to the KDIGO (Kidney Disease Improving Global Outcomes) Guidelines. </p> <h3>Results</h3> <p> The incidence of AKI cases constantly increased during the observation period and reached the maximum level by the week 5 following HSCT. MBM elevation was observed more frequently than AKI and preceded the latter. Clusterin, MCP-1 and KIM-1 levels significantly correlated with subsequent serum creatinine values, measured a week after the MBM’s analysis according to multivariate linear regression models adjusted for other confounders. An increase in KIM-1 and/or MCP-1 urinary excretion was independently associated with a relative risk of AKI development. In summary, multiple renotoxic events early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(1754) "The aim of this study was to determine clinical value of molecular biomarkers (MBM), associated with tubular epithelium damage, for the prediction of acute kidney injury (AKI) in the setting of hematopoietic stem cell transplantation (HSCT).
Patients and methods
Ninety HSCT recipients (46 males, 44 females) were enrolled into the observational prospective study. Urinary concentrations of calbindin, clusterin, IL-18 (interleukin- 18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase-π) and MCP-1 (monocyte chemoattractant protein-1) were measured in all patients before HSCT and at 5 consequent time points during early post-transplant period, along with routine clinical monitoring. AKI was defined according to the KDIGO (Kidney Disease Improving Global Outcomes) Guidelines.
Results
The incidence of AKI cases constantly increased during the observation period and reached the maximum level by the week 5 following HSCT. MBM elevation was observed more frequently than AKI and preceded the latter. Clusterin, MCP-1 and KIM-1 levels significantly correlated with subsequent serum creatinine values, measured a week after the MBM’s analysis according to multivariate linear regression models adjusted for other confounders. An increase in KIM-1 and/or MCP-1 urinary excretion was independently associated with a relative risk of AKI development. In summary, multiple renotoxic events early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(1754) "The aim of this study was to determine clinical value of molecular biomarkers (MBM), associated with tubular epithelium damage, for the prediction of acute kidney injury (AKI) in the setting of hematopoietic stem cell transplantation (HSCT).
Patients and methods
Ninety HSCT recipients (46 males, 44 females) were enrolled into the observational prospective study. Urinary concentrations of calbindin, clusterin, IL-18 (interleukin- 18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase-π) and MCP-1 (monocyte chemoattractant protein-1) were measured in all patients before HSCT and at 5 consequent time points during early post-transplant period, along with routine clinical monitoring. AKI was defined according to the KDIGO (Kidney Disease Improving Global Outcomes) Guidelines.
Results
The incidence of AKI cases constantly increased during the observation period and reached the maximum level by the week 5 following HSCT. MBM elevation was observed more frequently than AKI and preceded the latter. Clusterin, MCP-1 and KIM-1 levels significantly correlated with subsequent serum creatinine values, measured a week after the MBM’s analysis according to multivariate linear regression models adjusted for other confounders. An increase in KIM-1 and/or MCP-1 urinary excretion was independently associated with a relative risk of AKI development. In summary, multiple renotoxic events early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation.
" } ["DOI"]=> array(37) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8010" ["VALUE"]=> string(36) "10.18620/ctt-1866-8836-2016-5-4-8-14" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(36) "10.18620/ctt-1866-8836-2016-5-4-8-14" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(36) "10.18620/ctt-1866-8836-2016-5-4-8-14" } ["NAME_EN"]=> array(37) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8013" ["VALUE"]=> string(122) "Subclinical tubular epithelium damage and acute kidney injury following allogeneic hematopoietic stem cell transplantation" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(122) "Subclinical tubular epithelium damage and acute kidney injury following allogeneic hematopoietic stem cell transplantation" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(122) "Subclinical tubular epithelium damage and acute kidney injury following allogeneic hematopoietic stem cell transplantation" } ["ORGANIZATION_EN"]=> array(37) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8012" ["VALUE"]=> array(2) { ["TEXT"]=> string(557) "Prof. Vladimir A. Dobronravov, MD, PhD, DSc, Research Institute of Nephrology, The First St. Petersburg I. Pavlov State<br> Medical University, Russia, L.Tolstoy St. 17, 197089, St. Petersburg, Russia<br> <br> <sup>1</sup>First St. Petersburg I. Pavlov State Medical University, Russia,<br> <sup>2</sup>Nephrology Research Institute,<br> <sup>3</sup>Raisa Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(485) "Prof. Vladimir A. Dobronravov, MD, PhD, DSc, Research Institute of Nephrology, The First St. Petersburg I. Pavlov StateMedical University, Russia, L.Tolstoy St. 17, 197089, St. Petersburg, Russia
1First St. Petersburg I. Pavlov State Medical University, Russia,
2Nephrology Research Institute,
3Raisa Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(485) "Prof. Vladimir A. Dobronravov, MD, PhD, DSc, Research Institute of Nephrology, The First St. Petersburg I. Pavlov State
Medical University, Russia, L.Tolstoy St. 17, 197089, St. Petersburg, Russia
1First St. Petersburg I. Pavlov State Medical University, Russia,
2Nephrology Research Institute,
3Raisa Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
" } ["AUTHORS"]=> array(38) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(4) { [0]=> string(4) "8123" [1]=> string(4) "8124" [2]=> string(4) "8125" [3]=> string(4) "8126" } ["VALUE"]=> array(4) { [0]=> string(3) "390" [1]=> string(3) "389" [2]=> string(3) "654" [3]=> string(3) "391" } ["DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(4) { [0]=> string(3) "390" [1]=> string(3) "389" [2]=> string(3) "654" [3]=> string(3) "391" } ["~DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(4) { [0]=> string(66) "Vladimir A. Dobronravov" [1]=> string(60) "Kirill A. Smirnov" [2]=> string(61) "Boris V. Afanasiev" [3]=> string(58) "Olga V. Galkina" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["AUTHOR_RU"]=> array(37) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8007" ["VALUE"]=> array(2) { ["TEXT"]=> string(306) "Владимир А. Добронравов<sup>1,2</sup>, Кирилл А. Смирнов<sup>1,2</sup>, Борис В. Афанасьев<sup>1,3</sup>, Ольга В. Галкина<sup>1,2</sup>, Алексей В. Смирнов<sup>1</sup>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(246) "Владимир А. Добронравов1,2, Кирилл А. Смирнов1,2, Борис В. Афанасьев1,3, Ольга В. Галкина1,2, Алексей В. Смирнов1" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(246) "Владимир А. Добронравов1,2, Кирилл А. Смирнов1,2, Борис В. Афанасьев1,3, Ольга В. Галкина1,2, Алексей В. Смирнов1" } ["SUBMITTED"]=> array(37) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8000" ["VALUE"]=> string(22) "11/10/2016 02:55:00 pm" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/10/2016 02:55:00 pm" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "11/10/2016 02:55:00 pm" } ["ACCEPTED"]=> array(37) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8001" ["VALUE"]=> string(22) "12/09/2016 02:55:00 pm" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "12/09/2016 02:55:00 pm" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "12/09/2016 02:55:00 pm" } ["KEYWORDS"]=> array(38) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(3) { [0]=> string(4) "8120" [1]=> string(4) "8121" [2]=> string(4) "8122" } ["VALUE"]=> array(3) { [0]=> string(3) "655" [1]=> string(2) "15" [2]=> string(3) "657" } ["DESCRIPTION"]=> array(3) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(3) { [0]=> string(3) "655" [1]=> string(2) "15" [2]=> string(3) "657" } ["~DESCRIPTION"]=> array(3) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(3) { [0]=> string(90) "Острое повреждение почек" [1]=> string(134) "трансплантация гемопоэтических стволовых клеток" [2]=> string(64) "биомаркеры" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["CONTACT"]=> array(38) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8002" ["VALUE"]=> string(3) "390" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "390" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(66) "Vladimir A. Dobronravov" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["SUMMARY_RU"]=> array(37) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8009" ["VALUE"]=> array(2) { ["TEXT"]=> string(3641) "<h3>ЦЕЛЬ ИССЛЕДОВАНИЯ</h3> <p>определение значения молекулярных биомаркеров (МБМ), ассоциированных с альтерацией клеток тубулярного эпителия, в прогнозировании острого повреждения почек после трансплантации гемопоэтических стволовых клеток (ТГСК).</p> <h3>ПАЦИЕНТЫ И МЕТОДЫ </h3> <p>В открытое обзервационное проспективное исследование включены 90 больных (46 мужчин и 44 женщины), которым была выполнена ТГСК. В образцах мочи до ТГСК и на пяти первых неделях раннего посттрансплантационного периода определены концентрации МБМ (кальбиндина, кластерина, интерлейкина-18 (IL-18), молекулы повреждения почек-1 (KIM-1), глютатион-S-трансферазы (π-класс) (GST-π), протеина хемотаксиса моноцитов-1 (MCP-1). Параллельно мониторировали основные клинические показатели. Диагностику и стратификацию тяжести острого повреждения почек (ОПП) проводили согласно рекомендациям KDIGO (Kidney Disease Improving Global Outcomes).</p> <h3>РЕЗУЛЬТАТЫ</h3> <p>Доля случаев с ОПП прогрессивно увеличивалась в раннем посттрансплантационном периоде, достигнув максимума к неделе 5 после ТГСК. Повышение содержания МБМ выявляли существенно чаще случаев ОПП, причем повышение МБМ предшествовало формированию дисфункции почек (ОПП). При множественном регрессионном анализе кластерин, MCP-1 и KIM-1 имели прямую, достоверную, независимую от других анализируемых факторов связь с креатинином сыворотки крови на неделе, следующей за определением МБМ. Повышение мочевой экскреции KIM-1 и (или) MCP-1 было независимо от других клинических факторов ассоциировано с увеличением относительного риска (ОР) развития ОПП.</p> <h3>ЗАКЛЮЧЕНИЕ</h3> <p>Множественные ренотоксичные воздействия при ТГСК приводят к существенному и одновременному повышению экскреции с мочой БМ тубулярного повреждения, отражающему субклиническое повреждение клеток тубулярного эпителия в отсутствии критериев ОПП; оценка мочевой экскреции KIM-1 и MCP-1 представляется наиболее подходящим методом предиктивной диагностики ОПП ассоциированного с ТГСК.</p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(3545) "
ЦЕЛЬ ИССЛЕДОВАНИЯ
определение значения молекулярных биомаркеров (МБМ), ассоциированных с альтерацией клеток тубулярного эпителия, в прогнозировании острого повреждения почек после трансплантации гемопоэтических стволовых клеток (ТГСК).
ПАЦИЕНТЫ И МЕТОДЫ
В открытое обзервационное проспективное исследование включены 90 больных (46 мужчин и 44 женщины), которым была выполнена ТГСК. В образцах мочи до ТГСК и на пяти первых неделях раннего посттрансплантационного периода определены концентрации МБМ (кальбиндина, кластерина, интерлейкина-18 (IL-18), молекулы повреждения почек-1 (KIM-1), глютатион-S-трансферазы (π-класс) (GST-π), протеина хемотаксиса моноцитов-1 (MCP-1). Параллельно мониторировали основные клинические показатели. Диагностику и стратификацию тяжести острого повреждения почек (ОПП) проводили согласно рекомендациям KDIGO (Kidney Disease Improving Global Outcomes).
РЕЗУЛЬТАТЫ
Доля случаев с ОПП прогрессивно увеличивалась в раннем посттрансплантационном периоде, достигнув максимума к неделе 5 после ТГСК. Повышение содержания МБМ выявляли существенно чаще случаев ОПП, причем повышение МБМ предшествовало формированию дисфункции почек (ОПП). При множественном регрессионном анализе кластерин, MCP-1 и KIM-1 имели прямую, достоверную, независимую от других анализируемых факторов связь с креатинином сыворотки крови на неделе, следующей за определением МБМ. Повышение мочевой экскреции KIM-1 и (или) MCP-1 было независимо от других клинических факторов ассоциировано с увеличением относительного риска (ОР) развития ОПП.
ЗАКЛЮЧЕНИЕ
Множественные ренотоксичные воздействия при ТГСК приводят к существенному и одновременному повышению экскреции с мочой БМ тубулярного повреждения, отражающему субклиническое повреждение клеток тубулярного эпителия в отсутствии критериев ОПП; оценка мочевой экскреции KIM-1 и MCP-1 представляется наиболее подходящим методом предиктивной диагностики ОПП ассоциированного с ТГСК.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(3545) "ЦЕЛЬ ИССЛЕДОВАНИЯ
определение значения молекулярных биомаркеров (МБМ), ассоциированных с альтерацией клеток тубулярного эпителия, в прогнозировании острого повреждения почек после трансплантации гемопоэтических стволовых клеток (ТГСК).
ПАЦИЕНТЫ И МЕТОДЫ
В открытое обзервационное проспективное исследование включены 90 больных (46 мужчин и 44 женщины), которым была выполнена ТГСК. В образцах мочи до ТГСК и на пяти первых неделях раннего посттрансплантационного периода определены концентрации МБМ (кальбиндина, кластерина, интерлейкина-18 (IL-18), молекулы повреждения почек-1 (KIM-1), глютатион-S-трансферазы (π-класс) (GST-π), протеина хемотаксиса моноцитов-1 (MCP-1). Параллельно мониторировали основные клинические показатели. Диагностику и стратификацию тяжести острого повреждения почек (ОПП) проводили согласно рекомендациям KDIGO (Kidney Disease Improving Global Outcomes).
РЕЗУЛЬТАТЫ
Доля случаев с ОПП прогрессивно увеличивалась в раннем посттрансплантационном периоде, достигнув максимума к неделе 5 после ТГСК. Повышение содержания МБМ выявляли существенно чаще случаев ОПП, причем повышение МБМ предшествовало формированию дисфункции почек (ОПП). При множественном регрессионном анализе кластерин, MCP-1 и KIM-1 имели прямую, достоверную, независимую от других анализируемых факторов связь с креатинином сыворотки крови на неделе, следующей за определением МБМ. Повышение мочевой экскреции KIM-1 и (или) MCP-1 было независимо от других клинических факторов ассоциировано с увеличением относительного риска (ОР) развития ОПП.
ЗАКЛЮЧЕНИЕ
Множественные ренотоксичные воздействия при ТГСК приводят к существенному и одновременному повышению экскреции с мочой БМ тубулярного повреждения, отражающему субклиническое повреждение клеток тубулярного эпителия в отсутствии критериев ОПП; оценка мочевой экскреции KIM-1 и MCP-1 представляется наиболее подходящим методом предиктивной диагностики ОПП ассоциированного с ТГСК.
" } ["ORGANIZATION_RU"]=> array(37) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8008" ["VALUE"]=> array(2) { ["TEXT"]=> string(558) "<sup>1</sup>Первый Санкт-Петербургский государственный медицинский университет им. акад. И. П. Павлова, Россия <br> <sup>2</sup>Научно-исследовательский институт нефрологии <br> <sup>3</sup>Научно-исследовательский институт детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(510) "1Первый Санкт-Петербургский государственный медицинский университет им. акад. И. П. Павлова, Россия2Научно-исследовательский институт нефрологии
3Научно-исследовательский институт детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(510) "1Первый Санкт-Петербургский государственный медицинский университет им. акад. И. П. Павлова, Россия
2Научно-исследовательский институт нефрологии
3Научно-исследовательский институт детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой" } } } [3]=> array(49) { ["IBLOCK_SECTION_ID"]=> string(2) "20" ["~IBLOCK_SECTION_ID"]=> string(2) "20" ["ID"]=> string(3) "678" ["~ID"]=> string(3) "678" ["IBLOCK_ID"]=> string(1) "2" ["~IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["~NAME"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["ACTIVE_FROM"]=> NULL ["~ACTIVE_FROM"]=> NULL ["TIMESTAMP_X"]=> string(22) "03/29/2017 05:56:24 pm" ["~TIMESTAMP_X"]=> string(22) "03/29/2017 05:56:24 pm" ["DETAIL_PAGE_URL"]=> string(150) "/en/archive/vypusk-5-nomer-4/klinicheskie-raboty/vremennaya-dinamika-vosstanovleniya-immuniteta-i-virusnaya-reaktivatsiya-posle-transplantatsii-gemop/" ["~DETAIL_PAGE_URL"]=> string(150) "/en/archive/vypusk-5-nomer-4/klinicheskie-raboty/vremennaya-dinamika-vosstanovleniya-immuniteta-i-virusnaya-reaktivatsiya-posle-transplantatsii-gemop/" ["LIST_PAGE_URL"]=> string(12) "/en/archive/" ["~LIST_PAGE_URL"]=> string(12) "/en/archive/" ["DETAIL_TEXT"]=> string(46061) "
Introduction
Hematopoietic stem cell transplantation (HSCT) is usually preceded by intensive chemo- an/or radiation therapy causing cytotoxic insult to hematopoietic progenitors and lymphoid cell populations, depletion of myeloid and lymphoid cell pools, thus resulting into severe temporary immune deficiency. Therefore, HSCT patients are at high risk for bacterial and viral infections, at least, at early terms (< 100 days) post-transplant (Fig.1). The organ-specific bacterial and virus-associated complications following intensive chemoand/ or radiotherapy are treated with antimicrobial and antiviral drugs, either in pre-emptive mode, or upon detection of the pathogens [6].
The aim of our review is to evaluate reconstitution terms and patterns for major immune populations following HSCT, comparing it with reactivation terms for the most common herpesviruses, i.e., cytomegalovirus (CMV), Epstein-Barr virus (EBV), and Herpes Simplex virus (HSV).
Recovery of myeloid populations post-HSCT
The granulocyte reconstitution depends on the so-called transit time, i.e., the total time period required for stem cell recruitment, differentiation, expansion, maturation and release to the bloodstream. Restoration terms for myeloid cells after hematopoietic stem cell transplantation (HSCT) are usually defined as the 1st day with neutrophil counts of >0.5x109/L, with mean recovery terms of 12 to 20 days [1]. The time to neutrophil engraftment depends on the type of graft, with a median time of ca. 30 days for umbilical stem cells, 21 days for bone marrow, and 14 days for peripheral blood stem cells [44]. Generally, the innate immune system including granulocytes, monocytes, and NK cells recovers within weeks after intensive cytostatic treatment and HSCT.
According to Bemark et al. [4], granulocytes and monocytes (along with blood platelets), are the first mature donor cells appearing in peripheral blood post-transplant. These early- produced cells are generated by means of stress- (or ineffective) hemopoiesis, thus being functionally impaired. E.g., blood granulocytes produced up to day +30 post-HSCT are relatively fragile and apoptosis-prone [7]. The neutrophil functions (e.g. chemotaxis, phagocytosis and bacterial killing) may be also attenuated, whereas monocytes may produce normal, or, sometimes, decreased amounts of IL-1, IL-6 and TNF-1 [39, 42, 55]. Generally, innate immune response of phagocytic cells (granulocytes and monocytes) is, recovered within 4-6 weeks after engraftment of hematopoietic cells.
Absolute numbers of natural killer (NK) cells are normalized rather soon after HSCT. The study by Nguyen et al. [35] included ten cases of haplo-HSCT traced at d+14 to d+90 post-transplant. In available cases No.8,9 and 10, the percentages of NK cells were high since d+14, followed by reconstitution of B cells by d+30, whereas T cells started to increase as late as at 3 months. The earliest reconstituted NK cells exhibit a CD56high phenotype, with high levels of NK G2A/CD94 and lower amounts of inhibitory KIR, thus reflecting their relative immaturity which may be traced up to 6 months post-HSCT [35, 51]. A more extensive study was performed in 56 patients subjected to haplo-HSCT [51]. Blood samples were analyzed from day +15 to >100 days post-HSCT for CD56bright/dim like as CD117, NKG2A, CD62L and other surface differentiation markers. It has been shown that restoration of functionally mature, NK-cell subsets after haploidentical HSCT proceeds for, at least, 3 months. Hence, phenotypic maturation of NK cells and their functional ability post-transplant confirms suboptimal levels of mature NK’s up to 3 months after HSCT [14]. Approximate terms of numeric and functional deficiencies for different blood leukocyte populations are summarized in Table.1.
Overview of T cell reconstitution post-HSCT
Adaptive immunity after HSCT recovers much slower than innate immune response. E.g., B- and T-cell counts may normalize during the first months after transplantation [47]. A comprehensive summary of data concerning lymphocyte subpopulations following allo-HSCT is presented by de Koning et al. [12]. The authors state, however, that available information on the post-HSCT recovery of generic T, B and NK cells is limited by later terms (>1 month post-transplant).
The numbers of CD8+ T cells post-transplant increase earlier than those of CD4+ T cells. Therefore, the CD4/CD8 ratio is initially reversed in these patients [15]. A sufficient number of T cell-related immune parameters were studied by [21] who evaluated immune recovery in 67 patients with acute myeloid leukemia (AML) following allo-HSCT. They studied CD3+, CD3+CD4+, CD3+CD8+, CD3+CD4+/CD3+CD8+ ratio, CD3-CD56+, and CD19+ cells on days +30…365. Peripheral blast counts >5% were related to lower number of CD3+CD4+ (day +30) and NK cells (day +180; p= 0.02). Intensity of conditioning did not have any significant impact on the kinetics of immune recovery. Patients with normal CD3+CD4+/CD3+CD8+ ratio (day +30) and NK cell counts (day +90; p < 0.05) experienced better survival than those with decreased parameters. Post-transplant sepsis or severe infections were associated with impaired CD3+CD8+ (day +90; p = 0.015) and CD19+ (day +90; p = 0.02) recovery. Relapse in patients following allo-SCT showed an association with decreased numbers of CD19+ (day +270) and NK cells (day +365). Acute GvHD (II-IV) was accompanied by reduced CD19+ and CD3+CD4+ cells. Thus, deficiencies of the major lymphoid populations correlated with common post-transplant complications.
There are two biological pathways for restoration of CD4+ Т cells in HSCT cases, i.e., thymus-dependent (central) and peripheral mechanisms, as discussed by Gress et al. [16]. In pediatric patients with chemotherapy-induced lymphopenia the recovery of naive, CD45RA+CD45RO- CD4+ T cells predominated within first six months, accompanied by marked increase of thymus volume [30]. These data again suggest the thymus-dependent T cell production to be primarily responsible for the repopulation of peripheral CD4+ T cells in young patients. Meanwhile, therapy-related lymphopenia in adults (ages over 30) was recovered by scarce production of CD45RA+ CD4+ cells during the first year post therapy. However, CD45RO+ CD4+ cells increased rapidly in number, repopulating the CD4+ T cells to pretreatment levels within the first 3 months after chemotherapy [17]. Thus, the “adult” type of CD4+ T cell recovery reflects a thymus-independent expansion pathway resulting into production of memory T cells. Therefore, in absence of effective thymopoiesis, the levels of total CD4 cells may remain low, even 4–5 years after transplant. Furthermore, thymus-dependent CD4 production caused renewal of central memory populations, identified as CD62L+ CCR7+ CD45RA– [18]. Hence, proper thymus functioning is necessary for rapid CD4+T cell restoration following HSCT. Therefore, pediatric patients have, potentially, more chances to restore their T cell pool within a shorter time period.
The immunological status of 37 adult patients with lymphoma after high-dose chemotherapy auto-SCT was studied by Dean et al. [11]. At a median follow-up of 10.5 years (range 2.2-20.2) following auto-SCT, the proportion of CD4(+) cells remained significantly reduced in patients compared with controls, predominantly a result of persistently reduced naive CD4(+) cell numbers. The numbers of naive CD8(+) lymphocytes (P = 0.014) and transitional B cells (P = 0.008) were also significantly reduced. Revaccination of the patients, as a test for antibody induction, showed ambiguous results. Hence, immunological deficiency of T cell subsets may be retained for many years after cytostatic treatment and auto-SCT.
In view of polyclonal T cell reconstitution post-HSCT, it, generally, may produce only minor fractions targeted for specific viral pathogens. Over last 10-15 years, a search for enrichment and expansion of virus-specific T cells (against CMV, EBV etc.) draws much attention. The CD8+T cell clones recognizing viruses are also present at the time of HSCT, being potentially able of selective expansion in cases of CMV or EBV activation. However, at the first months after transplantation, a narrow and skewed repertoire of T lymphocytes with memory-like phenotype is observed [5, 27]. Therefore, many groups have studied adoptive T-cell transfer, e.g., establishment of early virus-specific Т cell populations as a means of immune recovery acceleration and prevention of early viral infections [50]. Moreover, deficiency of such T-cell clones with virus-restricted specificity may be a sufficient clinical factor at remote terms, e.g., effects of sustained Epstein-Barr virus (EBV) activation, causing the so-called secondary lymphoproliferative disease (LPD) in HSCT patients. Both short- and long-term immunotherapeutic strategies, including virus-specific T cells, are widely discussed.
B cell recovery post-HSCT
An extensive review by Bemark et al. [4] discussed time course and reasons for altered antibody response and B cell evolution following HSCT, in context of peripheral B cell differentiation. The mature B cells and their precursors are known to be efficiently depleted during conditioning, the levels of circulating IgG antibodies drop slowly, due to long half-life of IgG in serum and the survival of many plasma cells after myelodepletion [46, 52]. Recipient plasma cells are lost relatively quickly post-HSCT, due to cytotoxicity of conditioning, or depletion of patients’ plasma cells by donor-derived T cells. Moreover, memory B cells replenish the pool of plasma cells which produce specific antibodies induced by vaccinations. Hence the loss of specific antibodies may require revaccination several months after HSCT.
The B cells are deficient in peripheral blood over the first months after HSCT, with gradual normalization of cell counts within 6–12 months [3]. Memory B cells expressing CD27 did not expand to full scale, remaining at subnormal levels for, at least, 2 years after transplantation, as shown by Avanzini et al. [2] in pediatric HSCT patients. The profiles of circulating B cells were studied at 3 to 24 months post-transplant. At any terms, the patients displayed a significantly higher percentage of naive (IgD+CD27-) B cells and showed significantly lower in vitro stimulation of Ig production, as compared to healthy controls. One should, however, consider an opportunity of donor memory B cell transfer, like as persistence of residual memory B cells early after HSCT, especially following reduced-intensity conditioning regimens.
Concerning mitogenic response of B cells from HSCT patients, a near-normal in vitro proliferation was shown after 8-day culturing with EBV particles, whereas response to pokeweed mitogen (PWM) was significantly decreased [29]. T cells from HSCT patients showed a decreased helper ability in PWM-induced B cell activation. Intrinsic deficiencies within the B cell compartment may inhibit their nonspecific mitogenic responses.
An alternative definition of human transitional memory B cells is that they are CD27 –IgM+ CD5- CD10-. These immature B cell subsets cells are abundant in young children and in cord blood, and constitute 25–50% of all B cells even 1 year after HSCT, thus depicting a long-term immaturity of B cell populations post-transplant [4].
Following HSCT, the cells producing different antibody subclasses emerge in a distinct order, with production of IgM antibodies within a few months, followed by IgG1/ IgG3, IgG2/IgG4 and finally IgA [13]. Thus, deficiencies of humoral immune response are common after HSCT, being lifelong in some cases, due to intrinsic defects in B cells as well as prolonged deficiency of T helpers (e.g., naïve CD4+ T cells) in peripheral blood. Follicular dendritic cells (FDC) in lymph nodes may be also damaged by cytotoxic chemotherapy thus impairing the production of T cell-dependent antibody responses with germinal centre reactions [4]. With respect to long-term clinical consequences, aside of chronic GVHD, one should consider a risk of recurrent bacterial infections and poor responses to polysaccharide vaccines in HSCT survivors.
Hence, long-term T- and B cell immune deficiencies in allo-transplanted patients, seem to be a regular condition caused by preceding cytostatic treatment, and long-lasting immunosuppressive treatment post-transplant. Gradual restoration of normal immune hierarchy may proceed for years in these cases, especially in adult and elderly patients, causing retarded recovery of the key antiviral cell populations (CD4+ lymphocytes, mature NK cells), as well as B cells, including those producing virus-specific antibodies.
Vaccine-induced antibody response post-transplant
It is well known that distinct lymphoid subpopulations recover at different rates post-HSCT, thus affecting humoral immune response. Specific antibody production upon vaccination is an integral index of immune response. Surprisingly, there are only sparsed data on immunization and antibody response in these patients, especially at early terms (< 6 mo) post-HSCT.
Generally, the post-vaccinal immunity in HSCT recipients is determined by the long-term antibody response which largely determines infectious safety for the children post-transplant. E.g., Inaba et al. [20] performed a study at the St. Jude Children’s Hospital among 210 pediatric allo-HSCT recipients one year after transplantation. All the patients were re-immunized with combined vaccines, i.e., diphtheria- tetanus (Td), measles, mumps and rubella (MMR), as well as against hepatitis B and poliovirus at their first annual post-transplantation visit. The authors evaluated quality of routine immunization against eight common infections diphtheria, tetanus, pertussis, measles, mumps, rubella, hepatitis B, and poliovirus). Long-term antibody responses (>5 years post-vaccination) were seen for tetanus (95.7%), rubella (92.3%), poliovirus (97.9%), and, in diphtheria-tetanus- acellular pertussis (DTaP) recipients, diphtheria (100%), whereas pertussis vaccines were less effective (25% of cases with protective antibody levels).
In particular, testing for measles antibodies (n=130) showed an increase of seropositivity rates from 30% to 67%. This response to vaccine retained for >5 years. Lower CD3 counts were significantly associated with negative titres.
Results of influenza vaccination were reviewed by Ljungman and Avetisyan [24]. They discussed different works on the influenza vaccination in HSCT recipients. Appropriate studies on antibody response were rather small in size and difficult to compare, due to varying vaccination timing post-HSCT. Generally, the post-vaccinal response proved to be lower in HSCT patients as compared to healthy individuals, however, providing certain protection levels for the patients receiving influenza vaccine at >6 months or later after HSCT.
Of the 133 patients tested for rubella antibodies before immunization, 44% were positive. The percentage of patients positive for rubella antibodies more than doubled (93%) within a year of vaccination, and this effect retained for more than 5 years. Among patients evaluated for hepatitis B surface antigen antibodies, 52% were seropositive for before vaccination. Following immunization, 77% of the patients with antibody responses became HBV-seropositive within 2 to 5 years. However, ca. 25% of the group did not develop detectable antibody response. The negative titers did correlate with lower CD4 counts and history of severe aGvHD.
Poliovirus type 1, 2, and 3 antibodies were found in 54-57% of cases before immunization. Within years after immunization, the percentages of seropositive patients gradually increased, up to 98% of patients having protective titres at >5 years. Negative titres were associated, e.g., with older age at immunization, negative initial poliovirus serum titres, the use of radiation during the preconditioning therapy, and positive recipient status.
Hence, the vaccination failure in different cohorts was associated with following factors: older age and negative titres at immunization; lower CD3, CD4 or CD19 counts; higher IgM concentrations; positive recipient CMV serology; acute or chronic graft-versus-host disease; and radiation during preconditioning. These data are of great value for re-immunization strategies in this cohort at infectious risk.
A real opportunity of anti-HBV immunity transfer via donor cells is shown in a study by Lindemann et al [23]. The workers selected a group of patients/matched donor pairs without pre-existing HBV immunity. Twenty-seven prospective donors were vaccinated against HBV. The donor vaccination produced HBs antibodies specific cellular responses in most of them. Interestingly, since 2 weeks post-HSCT, 86 and 67% of the recipients had positive humoral and cellular HBV reactions. Subsequently, HBV immunity re-appeared in 83% of the recipients without revaccination. These results show that a transfer of specific humoral and cellular immunity by allogeneic hematopoietic cell transplantation (HSCT) should, in principle, be possible.
Time course of latent virus activation in transplanted patients
High occurence of CMV in HSCT patients mostly results from reactivation of a latent virus which is acquired since early childhood. Many patients become seropositive at the age of 3 to 5 years, showing appreciable levels of specific IgG antibodies in most HSC recipients, thus suggestive for a primary infection in past time [41]. Most authors guess that CMV reactivation in most cases is observed in CMV-positive patients, being a clearly negative factor for clinical outcomes [25, 43]. CMV-associated clinical conditions are known since many years, especially, pneumonias, hepatitis, colitis, cystitis [49].
Time-course of CMV reactivation/reinfection may be determined either as higher CMV genome copies in the sample (quantitative DNA real-time assay), or as increased incidence of PCR-positive tests in subsequent sample series. The mean incidence of CMV positivity in peripheral blood after HSCT reaches its peak values 2 to 3 months following intensive therapy and transplantation [31].
CMV incidence and reactivation rates depend on intensity of conditioning treatment (non-myeloablative versus myeloablative therapy). E.g., a study of 537 CMV-seropositive patients has shown that a non-myeloablative conditioning was associated with lesser risk of subsequent CMV infection at early terms posttransplant [34]. However, nonmyeloablative regimen (TBI+Fludarabine) was, generally, followed by retarded CMV reactivation, as compared to myeloablative treatment [19]. Interestingly, a supplementary immunosuppression, e.g., treatment with antithymocyte globulin, or total lymphoid irradiation, correlated with earlier CMV viremia post-transplant, if compared with standard myeloablative therapy [43].
In fact, selective in vivo depletion of T cells, e.g., treatment with antithymocyte globulin prior to haplo-HSCT may cause a sufficient delay in CD4+T cell recovery, along with higher rates of CMV reactivation, as compared to the regimen with posttransplant cyclophosphamide [48]. These results suggest that the treatments with faster recovery of CD4+ T cells seem to better preserve antiviral immunity, thus allowing more efficient handling with herpesvirus infection after haplo-HSCT. Hence, targeted suppression of lymphoid cell populations pre-transplant is likely to cause higher incidence of CMV reactivation after HSCT.
Of special interest is early CMV activation in CMV-seropositive patients transplanted form CMV-negative donors. Since renewal of hematopoiesis in allo-HSCT setting proceeds with donor cells, one may expect decreased levels of anti-CMV specific T immunity, thus increasing risk of subsequent viremia [54].
In a similar HSCT cohort, we studied 143 patients with oncohematological disorders in a single-center study for herpesvirus reactivation post HSCT [9, 37]. The patients were subject to allogeneic transplantation of hematopoietic stem cells (HSCT). Occurrence and dynamics of acute GvHD was registered in posttransplant period, as well as other complications (pneumonia, neurological disorders, mucositis, cystitis). Reactivation of HSV, CMV and EBV was detectable in blood leukocytes after HSCT, respectively, in 51%, 57% and 45% of all cases (2.3-2.5-fold more common that pre-transplant). Primary diagnosis, gender of patients and type of transplant did not significantly influence these ratios.
Meanwhile, frequencies of CMV and HSV detection were dependent on patients’ age, with minimal detection rates at 1 to 4 years and a maximum at 10 to 20 years.
Prevalence of CMV positive tests was dependent upon the terms after HSCT, with a maximum at 2nd and 3rd months after transplantation (Fig.2A). As seen from previous data on cellular recovery, this time period is characterized by full restoration of innate immunity, and ongoing recovery maturation of adaptive lymphoid populations, e.g., CD4+ cells, and antibody-producing B cells. Similar post-transplant time dynamics was revealed for EBV reactivation (Fig 2B), thus presuming some analogies in reactivation mechanisms and type of interactions with host immune system. Herpes simplex (HSV) activation (Fig.2C) proceeded as soon as at 2nd- 3rd months after HSCT, probably, reflecting early affection of recipient epithelial cells, being its specific target population.
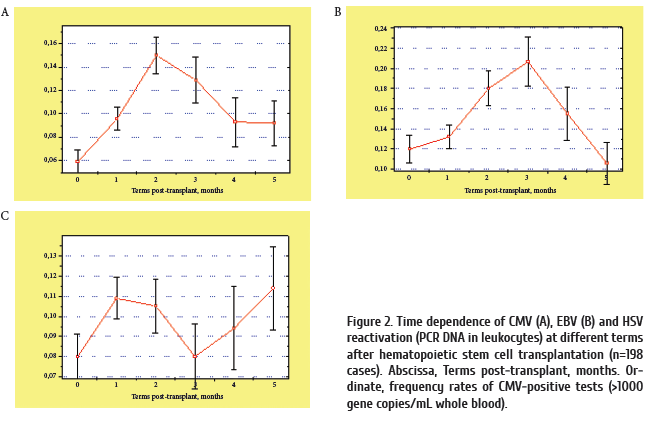
Finally, the incidence of post-transplant viremia depended on the patient’s age (Fig.3). I.e., in children of < 4 years old, the post-HSCT reactivation of cytomegalovirus was not frequent, being significantly increased (Р < 0.02) in older children (5 to 15 years old) post-transplant. Incidence of HSV reactivation was also maximal in adolescent recipients (Fig.4). These findings may be explained by lower infection levels in youngest children, maximal infection frequency in adolescents (6 to 15 years old), and sufficient antiviral immune response in older patients.
Like as recovery of immune populations, the CMV reactivation after HSCT may depend on a number of similar biological factors: 1) age of the patient; 3) intensity of conditioning regimen. Seropositivity of recipient is considered a special intrinsic factor for herpesvirus reactivation. All these factors should be taken into account when predicting risk for clinical CMV infection after intensive cytostatic treatment.
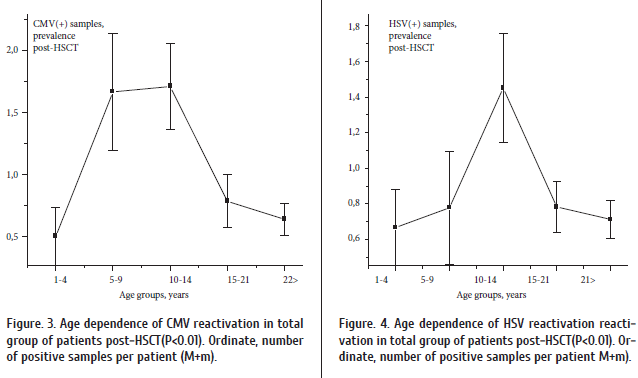
Along with studies of latent herpesviruses, some attention is now drawn to post-transplant reactivation of polyomaviruses (especially, BK and JC). E.g., BK virus is commonly found in urinary cell sediments by PCR approach, but it more frequently detected at 2-3 months post-HSCT [8]. Similar increase was observed for JC virus. Meanwhile, the BK and JC positivity in whole blood was not increased after HSCT, thus presuming only local reactivation of these viral pathogens.
Antibody response to cytomegalovirus reactivation
Since 1980s, it was proposed that both passive humoral immunity and specific T cell-mediated response contribute to the protective effect against CMV infection in allo-HSCT patients. Relative role of serological and cytotoxic immune responses in CMV infection was studied by Ludwig et al. (2006) who compared levels of glycoprotein B (gB)-specific antibodies and CMV-specific IFN-gamma producing CD4+ cells in CMV-positive SCT patients with/without CMV reactivation after SCT. The recipients without CMV reactivation showed a slow but steady increase in both parameters after SCT, indicating that initial high levels of gB specific antibodies or CMV specific CD4+ IFN-gamma+ cells are not necessary to prevent reactivation of CMV. Meanwhile, post-transplant CMV reactivation was associated with an increase in virus-specific CD4+ IFN-gamma+ cells prior to CMV activation, followed by a decline after the reactivation period. Patients who underwent only a single reactivation generated significant higher amounts of CD4+ IFN-gamma+ cells, than did patients with further reactivation episodes. Meanwhile, the course of gB specific antibodies for reactivating patients was something different, with significantly higher average values in the patients with CMV reactivation. This indicates that patients with a CMV reactivation exhibit a strong humoral dominated immune response.
Previous studies of protective anti-CMV antibodies in seropositive bone marrow recipients have shown induction of neutralizing anti-gB antibodies in cases of posttransplant CMV activation [40]. However, clinical significance of such neutralizing antibodies was not entirely confirmed in further studies [33]. The authors assessed time kinetics of the anti gB antibodies to CMV in 26 allo-HSC recipients. Neither absence, nor presence of CMV infection did correlate with serum concentrations of gB-specific and neutralizing antibodies. Recovery from CMV infection was not associated with antibody response to replication-associated antigens.
Potential clinical complications of EBV reactivation in transplanted patients
Among gamma-herpesviruses, the EBV is the mostly studied pathogen. Forty years ago, W and G. Henle had shown ability of EBV to infect B lymphocytes followed by their transformation to the lymphoblastoid virus-producing cells capable of long-term in vitro growth. Gamma-herpesviruses exhibit a well-known tropicity for lymphocytes, and, therefore, may induce lymphoproliferative disorders, as well as some muscular and endothelial neoplasias in humans [26]. The infected B-lymphoid cells reproduce normal stages of B cell differentiation in follicular germinal centers.
Latent EBV carriage is common to most adult subjects, as evidence by its common secretion with saliva. Uncontrolled proliferation of EBV-infected B cell in immunocompetent persons is blocked by cytotoxic lymphocytes. However, this mechanism of immune surveillance is largely failing in immunocompromised subjects [10, 36]. Therefore, EBV reactivation after transplantation of solid organs and hematopoietic stem cells is often followed by secondary posttransplant lymphoproliferative disorders (PTLD). Typically, the PTLDs emerge after long-term and massive immunosuppressive therapy which results into decreased amounts and functions of EBV-specific cytotoxic T cells, along with uncontrolled, EBV-driven B cell proliferation [10]. The EBV-positive malignancies observed in solid organ transplants, mostly originate from the recipient B cells, whereas 90% of PTLD in HSCT patients develop from the donor B cells [45].
Tischer et al. [48] have shown that 1/4 of patients pre-treated with ATG before HSCT developed PTLD, as compared to absence of this virally-induced condition among patients with presumably better preserved cytotoxic T cell populations.
Conclusions
Near-total depletion of innate and adaptive immune cell populations occurs due to intensive treatment followed by HSCT. Reconstitution of immune system from donor hematopoietic cells takes quite time periods needed for expansion, differentiation, migration and homing of granulocytes, monocytes, NK cells, T- and B cells. The intermediate maturation steps, especially, for lymphoid cell recovery, are known only to limited degree, and bear, mostly later terms (>1 month post-transplant).
Functional recovery of NK-cell subsets and blood monocytes may require up to 3 months; maturation and immune selection of CD8+ T cells proceeds for 3-4 months, whereas functional recovery of CD4+ T cells may be delayed for years, especially in older patients.
The B-cell ability to produce specific antibodies is partially retained, due to survival of resistant memory B cells post- HSCT, but differentiation and maturation of the donor-derived B cell hierarchy may require months and years.
Reactivation of herpesviruses in de novo produced donor cells, generally, proceeds in parallel with the periods of CD8+ and CD4+ T cell exhaustion, thus enabling expansion of latent herpesviruses, despite sufficient antibody response during CMV reactivation. Chronic EBV infection of B cells represents a more sufficient hazard, due to high risk of posttransplant lymphoproliferative disease at longer time range.
Hence, restoration of innate immunity, especially, monocytes/macrophages and NK cells originating from allografts, is most important at the early terms post-transplant, due to their immediate anti-infectious and GvL effects. At later terms (>100 d), deficient reconstitution of CD4+T cells may be responsible for deleterious infections with viruses and fungi, with older patients being at higher risk. To date, the delayed recovery of lymphoid subsets, especially, virus-specific T cells and memory B cells represents a major limitation for the long-term clinical outcomes in allo-transplanted patients. Therefore, current studies are aiming for enforcement of virus-targeted T-cell therapy, in order to accelerate clearance of cells with activated virus from affected tissues [32].
Conflict of interests
No conflict of interests is declared.
References
- Ali MY, Oyama Y, Monreal J, Winter J, Tallman M, Gordon LI, Williams S, Singhal S, Mehta J. Reassessing the definition of myeloid engraftment after autotransplantation: it is not necessary to see 0.5×10(9)/l neutrophils on 3 consecutive days to define myeloid recovery. Bone Marrow Transplant. 2002; 30 (11): 749-752.
- Avanzini MA, Locatelli F, Dos Santos C, Maccario R, Lenta E, Oliveri M, Giebel S, De Stefano P, Rossi F, Giorgiani G, Amendola G, Telli S, Marconi M. B lymphocyte reconstitution after hematopoietic stem cell transplantation: functional immaturity and slow recovery of memory CD27+ B cells. Exp Hematol 2005; 33 (4): 480-486.
- Baumgartner C, Morell A, Hirt A, Bucher U, Forster HK, Doran JE, Matter L, Brun del Re G, Wagner HP. Humoral immune function in pediatric patients treated with autologous bone marrow transplantation for B cell non-Hodgkin’s lymphoma. The influence of ex vivo marrow decontamination with anti-Y 29/55 monoclonal antibody and complement. Blood. 1988;71(5):1211-1217.
- Bemark M, Holmqvist J, Abrahamsson J, Mellgren K. Translational Mini-Review Series on B cell subsets in disease. Reconstitution after haematopoietic stem cell transplantation - revelation of B cell developmental pathways and lineage phenotypes. Clin Exp Immunol. 2012;167(1):15-25.
- Chalandon Y, Degermann S, Villard J, Arlettaz L, Kaiser L, Vischer S, Walter S, Heemskerk MH, van Lier RA, Helg C, Chapuis B, Roosnek E. Pre-transplantation CMV-specific T cells protect recipients of T-cell-depleted grafts against CMV-related complications. Blood 2006; 107:389–396.
- Chemaly RF, Shah DP, Boeckh MJ. Management of respiratory viral infections in hematopoietic cell transplant recipients and patients with hematologic malignancies. Clin Infect Dis. 2014; 59 (Suppl 5): S344-S351.
- Chukhlovin A. Enhanced ex vivo apoptosis of peripheral granulocytes is a sufficient factor of neutropenia following myeloablative chemotherapy. Leuk Res 2000; 24(6):507-509.
- Chukhlovin AB, Eismont YuA, Vavilov VN, Zubarovskaya LS, Afanasyev BV. Time- and sample-dependent differences in polyomavirus incidence following hematopoietic stem cell transplantation. Cell Ther Transplant. 2016; 5(1): 26-30.
- Chukhlovin A, Ovsyannikova M, Semenova E, Pankratova O, Zubarovskaya L, Afanasyev B Incidence of herpesvirus activation in young HSCT patients. 33 EBMT Meeting, Lyon, France, 2007. Abstr. P719.
- Cohen JI, Bollard CM, Khanna R, Pittaluga S. Current understanding of the role of Epstein-Barr virus (EBV) in lymphomagenesis and therapeutic approaches to EBV associated lymphomas. Leuk Lymphoma 2008;49(Suppl 1):27-34.
- Dean HF, Cazaly A, Hurlock C, Borras J, Williams AP, Johnson PW, Davies AJ. Defects in lymphocyte subsets and serological memory persist a median of 10 years after high-dose therapy and autologous progenitor cell rescue for malignant lymphoma. Bone Marrow Transplant. 2012;47(12):1545-1551.
- de Koning C, Plantinga M, Besseling P, Boelens JJ, Nierkens S. Immune reconstitution after allogeneic hematopoietic cell transplantation in children. Biol Blood Marrow Transplant. 2016; 22(2):195-206.
- Gerritsen EJ, van Tol MJ, Lankester AC, van der Weijden-Ragas CP, Jol-van der Zijde CM, Oudeman-Gruber NJ, Radl J, Vossen JM. Immunoglobulin levels and monoclonal gammopathies in children after bone marrow transplantation. Blood 1993; 82:3493–3502
- Gismondi A, Stabile H, Nisti P, Santoni A. Effector functions of natural killer cell subsets in the control of hematological malignancies. Front. Immunol. 6:567. doi: 10.3389/ fimmu.2015.00567
- Gratama JW, Naipal A, Oljans P, Zwaan FE, Verdonck LF, de Witte T, Vossen JM, Bolhuis RL, de Gast GC, Jansen J. T lymphocyte repopulation and differentiation after bone marrow transplantation. Early shifts in the ratio between T4+ and T8+ T lymphocytes correlate with the occurrence of acute graft-versus-host disease. Blood 1984;63:1416–1423.
- Gress RE, Emerson SG, Drobyski WR. Immune reconstitution: how it should work, what’s broken, and why it matters. Biol Blood Marrow Transplant. 2010; 16(1 Suppl):S133-137.
- Hakim FT, Cepeda R, Kaimei S, Mackall CL, McAtee N, Zujewski J, Cowan K, Gress RE. Constraints on CD4 recovery postchemotherapy in adults: Thymic insufficiency and apoptotic decline of expanded peripheral CD4 cells. Blood 1997;90 (9): 3789–3798
- Hakim FT, Memon SA, Cepeda R, Jones EC, Chow CK, Kasten-Sportes C, Odom J, Vance BA, Christensen BL, Mackall CL, Gress RE. Age-dependent incidence, time course, and consequences of thymic renewal in adults. J Clin Invest 2005;115(4):930–939
- Junghanss C, Boeckh M, Carter RA, Sandmaier BM, Maris MB, Maloney DG, Chauncey T, McSweeney PA, Little MT, Corey L, Storb R. Incidence and outcome of cytomegalovirus infections following nonmyeloablative compared with myeloablative allogeneic stem cell transplantation, a matched control study. Blood. 2002;99(6):1978-1985.
- Inaba H, Hartford CM, Pei D, Posner MJ, Yang J, Hayden RT, Srinivasan A, Triplett BM, McCulllers JA, Pui CH, Leung W. Longitudinal analysis of antibody response to immunization in paediatric survivors after allogeneic haematopoietic stem cell transplantation. Br J Haematol. 2012;156(1):109- 117.
- Klyuchnikov E, Asenova S, Kern W, Kilinc G, Ayuk F, Wiedemann B, Lioznov M, Freiberger P, Zalyalov Y, Zander AR, Kröger N, Bacher U. Post-transplant immune reconstitution after unrelated allogeneic stem cell transplant in patients with acute myeloid leukemia.Leuk Lymphoma. 2010;51(8):1450-1463.
- Koehne G, Zeller W, Stockschlaeder M, Zander AR. Phenotype of lymphocyte subsets after autologous peripheral blood stem cell transplantation. Bone Marrow Transplant. 1997;19(2):149-156.
- Lindemann M, Barsegian V, Runde V, Fiedler M, Heermann KH, Schaefer UW, Roggendorf M, Grosse-Wilde H. Transfer of humoral and cellular hepatitis B immunity by allogeneic hematopoietic cell transplantation. Transplantation. 2003 ;75(6):833-838.
- Ljungman P, Avetisyan G. Influenza vaccination in hematopoietic SCT recipients. Bone Marrow Transplant. 2008; 42(10): 637-641.
- Ljungman P. The role of cytomegalovirus serostatus on outcome of hematopoietic stem cell transplantation. Curr Opin Hematol. 2014 Nov;21(6):466-9.
- Longnecker R., Neipel F. Introduction to the human gamma-herpesviruses. In: Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis (Ed. A. Arvin, G. Campadelli- Fiume et al.). Cambridge University Press, 2007, Chapter 22.
- Lucas KG, Small TN, Heller G, Dupont B, O’Reilly RJ. The development of cellular immunity to Epstein–Barr virus after allogeneic bone marrow transplantation. Blood 1996;87:2594–2603.
- Ludwig B, Kraus FB, Kipp M, Preiser W, Schwerdtfeger R, Doerr HW, Buxbaum S. Cytomegalovirus-specific CD4 T-cell and glycoprotein B specific antibody response in recipients of allogenic stem cell transplantation. J Clin Virol. 2006; 35(2): 160-166.
- Lum LG, Seigneuret MC, Orcutt-Thordarson N, Noges JE, Storb R. The regulation of immunoglobulin synthesis after HLA-identical bone marrow transplantation: VI. Differential rates of maturation of distinct functional groups within lymphoid subpopulations in patients after human marrow grafting. Blood. 1985;65(6):1422-1433.
- Mackall CL, Fleisher TA, Brown MR, et al. Age, thymopoiesis, and CD4+ T-lymphocyte regeneration after intensive chemotherapy. New Engl J Med 1995; 332(3):143–149.
- Maeda Y, Teshima T, Yamada M, Shinagawa K, Nakao S, Ohno Y, Kojima K, Hara M, Nagafuji K, Hayashi S, Fukuda S, Sawada H, Matsue K, Takenaka K, Ishimaru F, Ikeda K, Niiya K, Harada M. Monitoring of human herpesviruses after allogeneic peripheral blood stem cell transplantation and bone marrow transplantation. Br J Haematol. 1999; 105(1):295-302.
- Moosmann A, Hammerschmidt W, Kolb HJ. Virus-specific T cells for therapy--approaches, problems, solutions. Eur J Cell Biol. 2012;91(1):97-101.
- Muñoz I, Gutiérrez A, Gimeno C, Farga A, Alberola J, Solano C, Prósper F, García-Conde J, Navarro D. Lack of association between the kinetics of human cytomegalovirus (HCMV) glycoprotein B (gB)-specific and neutralizing serum antibodies and development or recovery from HCMV active infection in patients undergoing allogeneic stem cell transplant. J Med Virol. 2001; 65(1): 77-84.
- Nakamae H, Kirby KA, Sandmaier BM, Norasetthada L, Maloney DG, Maris MB, Davis C, Corey L, Storb R, Boeckh M. Effect of Conditioning Regimen Intensity on CMV Infection in Allogeneic Hematopoietic Cell Transplantation. Biol Blood Marrow Transplant. 2009; 15(6): 694–703.
- Nguyen S, Dhedin N ,Vernant JP, Kuentz M, AlJijakli A, Rouas-Freiss N, et al. NK-cell reconstitution after haploidentical hematopoietic stem-cell transplantations: immaturity of NK cells and inhibitory effect of NKG2A override GvL effect. Blood 2005; 105:4135–4142.
- Nourse JP, Jones K, Gandhi MH Epstein-Barr-virus-related post-transplant lymphoproliferative disorders: pathogenetic insights for targeted therapy. Am J Transpl 2011; 11(5): 888-895.
- Pankratova OS, Chukhlovin AB, Zubarovskaya LS, Afanasyev BV. Incidence of herpesvirus detection and risk of common complications in allogeneic hematopoietic stem cell transplantation. Scientific Notes of the St.Petersburg State Medical University 2010; 17(1):56-60. (In Russian)
- Pankratova OS, Chukhlovin AB, Shiryaev SN, Eismont YA, Vavilov VN, Zubarovskaya LS, Afanasyev BV. Herpesviruses and oral ulcerations in hematopoietic SCT recipients. Bone Marrow Transplant. 2013; 48(10):1364-1365.
- Pechumer H, Leinisch E, Bender-Götze C, Ziegler-Heitbrock HW. Recovery of monocytes after bone marrow transplantation rapid reappearance of tumor necrosis factor alpha and interleukin 6 production. Transplantation. 1991; 52(4): 698-704.
- Roy DM, Brenner MK, Cook D, Duncan RI, Griffiths PD, Grundy JE. Transfer of humoral immunity against cytomegalovirus proteins following transplantation of T-cell-depleted allogeneic bone marrow from seropositive donors. J Med Virol. 1993; 41(2): 150-158.
- Ruell J, Barnes C, Mutton K, Foulkes B, Chang J, Cavet J, Guiver M, Menasce L, Dougal M, Chopra R. Active CMV disease does not always correlate with viral load detection. Bone Marrow Transplantation. 2007; 40: 55–61.
- Sahdev I, O’Reilly R, Black P, Heller G, HoffmannM. Interleukin-1 production following T-cell-depleted and unmodified marrow grafts. Pediatr Hematol Oncol 1996; 13:55–67.
- Schaenman JM, Shashidhar S, Rhee C, Wong J, Navato S, Wong RM, Ho DY, Arai S, Johnston L, Brown JM. Early CMV viremia is associated with impaired viral control following nonmyeloablative hematopoietic cell transplantation with a total lymphoid irradiation and antithymocyte globulin preparative regimen. Biol Blood Marrow Transplant 2011;17:693-702.
- Seggewiss R, Einsele H. Immune reconstitution after allogeneic transplantation and expanding options for immunomodulation: an update. Blood. 2010; 115(19): 3861–3868.
- Snow AL, Martinez OM. Epstein-Barr virus: evasive maneuvers in the development of PTLD. Am J Transplant 2007; 7(2):271–277.
- Storek J, Joseph A, Espino G, Dawson MA, Douek DC, Sullivan KM, Flowers ME, Martin P, Mathioudakis G, Nash RA, Storb R, Appelbaum FR, Maloney DG. Immunity of patients surviving 20 to 30 years after allogeneic or syngeneic bone marrow transplantation. Blood 2001; 98:3505–3512.
- Storek J, Geddes M, Khan F, Huard B, Helg C, Chalandon Y, Passweg J, Roosnek E. Reconstitution of the immune system after hematopoietic stem cell transplantation in humans. Semin Immunopathol. 2008; 30(4): 425-437.
- Tischer J, Engel N, Fritsch S, Prevalsek D, Hubmann M, Schulz C, Zoellner AK, Bücklein V, Reibke R, Mumm F, Rieger CT, Hill W, Ledderose G, Stemmler HJ, Köhnke T, Jäger G, Kolb HJ, Schmid C, Moosmann A, Hausmann A. Virus infection in HLA-haploidentical hematopoietic stem cell transplantation: incidence in the context of immune recovery in two different transplantation settings. Ann Hematol. 2015;94(10):1677-1688.
- Travi G, Pergam SA. Cytomegalovirus pneumonia in hematopoietic stem cell recipients. J Intensive Care Med. 2014; 29(4):200-212.
- Tzannou I, Leen AM. Accelerating immune reconstitution after hematopoietic stem cell transplantation. Clin Transl Immunology. 2014; 3(2):e11. doi: 10.1038/cti.2014.2. eCollection 2014.
- Vago L, Forno B, Sormani MP, Crocchiolo R, Zino E, DiTerlizzi S, etal. Temporal, quantitative, and functional characteristics of single-KIR-positive alloreactive natural killer cell recovery account for impaired graft-versus- leukemia activity after haploidentical hematopoietic stem cell transplantation. Blood. 2008; 112:3488–3499.
- van Tol MJ, Gerritsen EJ, de Lange GG, van Leeuwen AM, Jol-van der Zijde CM, Oudeman-Gruber NJ, de Vries E, Radl J, Vossen JM. The origin of IgG production and homogeneous IgG components after allogeneic bone marrow transplantation. Blood 1996; 87:818–826.
- Wald A, Corey L. Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis (ed. A. Arvin, G. Campadelli-Fiume et al.) Cambridge University Press. – 2007. – Chapter 36. URL: http://www.ncbi.nlm.nih.gov/books/NBK47376/
- Zhou W, Longmate J, Lacey SF, Palmer JM, Gallez-Hawkins G, Thao L, Spielberger R, Nakamura R, Forman SJ, Zaia JA, Diamond DJ. Impact of donor CMV status on viral infection and reconstitution of multifunction CMV-specific T cells in CMV-positive transplant recipients. Blood 2009;113(25):6465-6476.
- Zimmerli W, Zarth A, Gratwohl A, Speck B. Neutrophil function and pyogenic infections in bone marrow transplant recipients. Blood 1991; 77:393–399.
- Zucca E, Bertoni F, Vannata B, Cavalli F. Emerging role of infectious etiologies in the pathogenesis of marginal zone B-cell lymphomas. Clin Cancer Res. 2014; 20(20): 5207- 5216.
" ["~DETAIL_TEXT"]=> string(46061) "
Introduction
Hematopoietic stem cell transplantation (HSCT) is usually preceded by intensive chemo- an/or radiation therapy causing cytotoxic insult to hematopoietic progenitors and lymphoid cell populations, depletion of myeloid and lymphoid cell pools, thus resulting into severe temporary immune deficiency. Therefore, HSCT patients are at high risk for bacterial and viral infections, at least, at early terms (< 100 days) post-transplant (Fig.1). The organ-specific bacterial and virus-associated complications following intensive chemoand/ or radiotherapy are treated with antimicrobial and antiviral drugs, either in pre-emptive mode, or upon detection of the pathogens [6].
The aim of our review is to evaluate reconstitution terms and patterns for major immune populations following HSCT, comparing it with reactivation terms for the most common herpesviruses, i.e., cytomegalovirus (CMV), Epstein-Barr virus (EBV), and Herpes Simplex virus (HSV).

Recovery of myeloid populations post-HSCT
The granulocyte reconstitution depends on the so-called transit time, i.e., the total time period required for stem cell recruitment, differentiation, expansion, maturation and release to the bloodstream. Restoration terms for myeloid cells after hematopoietic stem cell transplantation (HSCT) are usually defined as the 1st day with neutrophil counts of >0.5x109/L, with mean recovery terms of 12 to 20 days [1]. The time to neutrophil engraftment depends on the type of graft, with a median time of ca. 30 days for umbilical stem cells, 21 days for bone marrow, and 14 days for peripheral blood stem cells [44]. Generally, the innate immune system including granulocytes, monocytes, and NK cells recovers within weeks after intensive cytostatic treatment and HSCT.
According to Bemark et al. [4], granulocytes and monocytes (along with blood platelets), are the first mature donor cells appearing in peripheral blood post-transplant. These early- produced cells are generated by means of stress- (or ineffective) hemopoiesis, thus being functionally impaired. E.g., blood granulocytes produced up to day +30 post-HSCT are relatively fragile and apoptosis-prone [7]. The neutrophil functions (e.g. chemotaxis, phagocytosis and bacterial killing) may be also attenuated, whereas monocytes may produce normal, or, sometimes, decreased amounts of IL-1, IL-6 and TNF-1 [39, 42, 55]. Generally, innate immune response of phagocytic cells (granulocytes and monocytes) is, recovered within 4-6 weeks after engraftment of hematopoietic cells.
Absolute numbers of natural killer (NK) cells are normalized rather soon after HSCT. The study by Nguyen et al. [35] included ten cases of haplo-HSCT traced at d+14 to d+90 post-transplant. In available cases No.8,9 and 10, the percentages of NK cells were high since d+14, followed by reconstitution of B cells by d+30, whereas T cells started to increase as late as at 3 months. The earliest reconstituted NK cells exhibit a CD56high phenotype, with high levels of NK G2A/CD94 and lower amounts of inhibitory KIR, thus reflecting their relative immaturity which may be traced up to 6 months post-HSCT [35, 51]. A more extensive study was performed in 56 patients subjected to haplo-HSCT [51]. Blood samples were analyzed from day +15 to >100 days post-HSCT for CD56bright/dim like as CD117, NKG2A, CD62L and other surface differentiation markers. It has been shown that restoration of functionally mature, NK-cell subsets after haploidentical HSCT proceeds for, at least, 3 months. Hence, phenotypic maturation of NK cells and their functional ability post-transplant confirms suboptimal levels of mature NK’s up to 3 months after HSCT [14]. Approximate terms of numeric and functional deficiencies for different blood leukocyte populations are summarized in Table.1.

Overview of T cell reconstitution post-HSCT
Adaptive immunity after HSCT recovers much slower than innate immune response. E.g., B- and T-cell counts may normalize during the first months after transplantation [47]. A comprehensive summary of data concerning lymphocyte subpopulations following allo-HSCT is presented by de Koning et al. [12]. The authors state, however, that available information on the post-HSCT recovery of generic T, B and NK cells is limited by later terms (>1 month post-transplant).
The numbers of CD8+ T cells post-transplant increase earlier than those of CD4+ T cells. Therefore, the CD4/CD8 ratio is initially reversed in these patients [15]. A sufficient number of T cell-related immune parameters were studied by [21] who evaluated immune recovery in 67 patients with acute myeloid leukemia (AML) following allo-HSCT. They studied CD3+, CD3+CD4+, CD3+CD8+, CD3+CD4+/CD3+CD8+ ratio, CD3-CD56+, and CD19+ cells on days +30…365. Peripheral blast counts >5% were related to lower number of CD3+CD4+ (day +30) and NK cells (day +180; p= 0.02). Intensity of conditioning did not have any significant impact on the kinetics of immune recovery. Patients with normal CD3+CD4+/CD3+CD8+ ratio (day +30) and NK cell counts (day +90; p < 0.05) experienced better survival than those with decreased parameters. Post-transplant sepsis or severe infections were associated with impaired CD3+CD8+ (day +90; p = 0.015) and CD19+ (day +90; p = 0.02) recovery. Relapse in patients following allo-SCT showed an association with decreased numbers of CD19+ (day +270) and NK cells (day +365). Acute GvHD (II-IV) was accompanied by reduced CD19+ and CD3+CD4+ cells. Thus, deficiencies of the major lymphoid populations correlated with common post-transplant complications.
There are two biological pathways for restoration of CD4+ Т cells in HSCT cases, i.e., thymus-dependent (central) and peripheral mechanisms, as discussed by Gress et al. [16]. In pediatric patients with chemotherapy-induced lymphopenia the recovery of naive, CD45RA+CD45RO- CD4+ T cells predominated within first six months, accompanied by marked increase of thymus volume [30]. These data again suggest the thymus-dependent T cell production to be primarily responsible for the repopulation of peripheral CD4+ T cells in young patients. Meanwhile, therapy-related lymphopenia in adults (ages over 30) was recovered by scarce production of CD45RA+ CD4+ cells during the first year post therapy. However, CD45RO+ CD4+ cells increased rapidly in number, repopulating the CD4+ T cells to pretreatment levels within the first 3 months after chemotherapy [17]. Thus, the “adult” type of CD4+ T cell recovery reflects a thymus-independent expansion pathway resulting into production of memory T cells. Therefore, in absence of effective thymopoiesis, the levels of total CD4 cells may remain low, even 4–5 years after transplant. Furthermore, thymus-dependent CD4 production caused renewal of central memory populations, identified as CD62L+ CCR7+ CD45RA– [18]. Hence, proper thymus functioning is necessary for rapid CD4+T cell restoration following HSCT. Therefore, pediatric patients have, potentially, more chances to restore their T cell pool within a shorter time period.
The immunological status of 37 adult patients with lymphoma after high-dose chemotherapy auto-SCT was studied by Dean et al. [11]. At a median follow-up of 10.5 years (range 2.2-20.2) following auto-SCT, the proportion of CD4(+) cells remained significantly reduced in patients compared with controls, predominantly a result of persistently reduced naive CD4(+) cell numbers. The numbers of naive CD8(+) lymphocytes (P = 0.014) and transitional B cells (P = 0.008) were also significantly reduced. Revaccination of the patients, as a test for antibody induction, showed ambiguous results. Hence, immunological deficiency of T cell subsets may be retained for many years after cytostatic treatment and auto-SCT.
In view of polyclonal T cell reconstitution post-HSCT, it, generally, may produce only minor fractions targeted for specific viral pathogens. Over last 10-15 years, a search for enrichment and expansion of virus-specific T cells (against CMV, EBV etc.) draws much attention. The CD8+T cell clones recognizing viruses are also present at the time of HSCT, being potentially able of selective expansion in cases of CMV or EBV activation. However, at the first months after transplantation, a narrow and skewed repertoire of T lymphocytes with memory-like phenotype is observed [5, 27]. Therefore, many groups have studied adoptive T-cell transfer, e.g., establishment of early virus-specific Т cell populations as a means of immune recovery acceleration and prevention of early viral infections [50]. Moreover, deficiency of such T-cell clones with virus-restricted specificity may be a sufficient clinical factor at remote terms, e.g., effects of sustained Epstein-Barr virus (EBV) activation, causing the so-called secondary lymphoproliferative disease (LPD) in HSCT patients. Both short- and long-term immunotherapeutic strategies, including virus-specific T cells, are widely discussed.
B cell recovery post-HSCT
An extensive review by Bemark et al. [4] discussed time course and reasons for altered antibody response and B cell evolution following HSCT, in context of peripheral B cell differentiation. The mature B cells and their precursors are known to be efficiently depleted during conditioning, the levels of circulating IgG antibodies drop slowly, due to long half-life of IgG in serum and the survival of many plasma cells after myelodepletion [46, 52]. Recipient plasma cells are lost relatively quickly post-HSCT, due to cytotoxicity of conditioning, or depletion of patients’ plasma cells by donor-derived T cells. Moreover, memory B cells replenish the pool of plasma cells which produce specific antibodies induced by vaccinations. Hence the loss of specific antibodies may require revaccination several months after HSCT.
The B cells are deficient in peripheral blood over the first months after HSCT, with gradual normalization of cell counts within 6–12 months [3]. Memory B cells expressing CD27 did not expand to full scale, remaining at subnormal levels for, at least, 2 years after transplantation, as shown by Avanzini et al. [2] in pediatric HSCT patients. The profiles of circulating B cells were studied at 3 to 24 months post-transplant. At any terms, the patients displayed a significantly higher percentage of naive (IgD+CD27-) B cells and showed significantly lower in vitro stimulation of Ig production, as compared to healthy controls. One should, however, consider an opportunity of donor memory B cell transfer, like as persistence of residual memory B cells early after HSCT, especially following reduced-intensity conditioning regimens.
Concerning mitogenic response of B cells from HSCT patients, a near-normal in vitro proliferation was shown after 8-day culturing with EBV particles, whereas response to pokeweed mitogen (PWM) was significantly decreased [29]. T cells from HSCT patients showed a decreased helper ability in PWM-induced B cell activation. Intrinsic deficiencies within the B cell compartment may inhibit their nonspecific mitogenic responses.
An alternative definition of human transitional memory B cells is that they are CD27 –IgM+ CD5- CD10-. These immature B cell subsets cells are abundant in young children and in cord blood, and constitute 25–50% of all B cells even 1 year after HSCT, thus depicting a long-term immaturity of B cell populations post-transplant [4].
Following HSCT, the cells producing different antibody subclasses emerge in a distinct order, with production of IgM antibodies within a few months, followed by IgG1/ IgG3, IgG2/IgG4 and finally IgA [13]. Thus, deficiencies of humoral immune response are common after HSCT, being lifelong in some cases, due to intrinsic defects in B cells as well as prolonged deficiency of T helpers (e.g., naïve CD4+ T cells) in peripheral blood. Follicular dendritic cells (FDC) in lymph nodes may be also damaged by cytotoxic chemotherapy thus impairing the production of T cell-dependent antibody responses with germinal centre reactions [4]. With respect to long-term clinical consequences, aside of chronic GVHD, one should consider a risk of recurrent bacterial infections and poor responses to polysaccharide vaccines in HSCT survivors.
Hence, long-term T- and B cell immune deficiencies in allo-transplanted patients, seem to be a regular condition caused by preceding cytostatic treatment, and long-lasting immunosuppressive treatment post-transplant. Gradual restoration of normal immune hierarchy may proceed for years in these cases, especially in adult and elderly patients, causing retarded recovery of the key antiviral cell populations (CD4+ lymphocytes, mature NK cells), as well as B cells, including those producing virus-specific antibodies.
Vaccine-induced antibody response post-transplant
It is well known that distinct lymphoid subpopulations recover at different rates post-HSCT, thus affecting humoral immune response. Specific antibody production upon vaccination is an integral index of immune response. Surprisingly, there are only sparsed data on immunization and antibody response in these patients, especially at early terms (< 6 mo) post-HSCT.
Generally, the post-vaccinal immunity in HSCT recipients is determined by the long-term antibody response which largely determines infectious safety for the children post-transplant. E.g., Inaba et al. [20] performed a study at the St. Jude Children’s Hospital among 210 pediatric allo-HSCT recipients one year after transplantation. All the patients were re-immunized with combined vaccines, i.e., diphtheria- tetanus (Td), measles, mumps and rubella (MMR), as well as against hepatitis B and poliovirus at their first annual post-transplantation visit. The authors evaluated quality of routine immunization against eight common infections diphtheria, tetanus, pertussis, measles, mumps, rubella, hepatitis B, and poliovirus). Long-term antibody responses (>5 years post-vaccination) were seen for tetanus (95.7%), rubella (92.3%), poliovirus (97.9%), and, in diphtheria-tetanus- acellular pertussis (DTaP) recipients, diphtheria (100%), whereas pertussis vaccines were less effective (25% of cases with protective antibody levels).
In particular, testing for measles antibodies (n=130) showed an increase of seropositivity rates from 30% to 67%. This response to vaccine retained for >5 years. Lower CD3 counts were significantly associated with negative titres.
Results of influenza vaccination were reviewed by Ljungman and Avetisyan [24]. They discussed different works on the influenza vaccination in HSCT recipients. Appropriate studies on antibody response were rather small in size and difficult to compare, due to varying vaccination timing post-HSCT. Generally, the post-vaccinal response proved to be lower in HSCT patients as compared to healthy individuals, however, providing certain protection levels for the patients receiving influenza vaccine at >6 months or later after HSCT.
Of the 133 patients tested for rubella antibodies before immunization, 44% were positive. The percentage of patients positive for rubella antibodies more than doubled (93%) within a year of vaccination, and this effect retained for more than 5 years. Among patients evaluated for hepatitis B surface antigen antibodies, 52% were seropositive for before vaccination. Following immunization, 77% of the patients with antibody responses became HBV-seropositive within 2 to 5 years. However, ca. 25% of the group did not develop detectable antibody response. The negative titers did correlate with lower CD4 counts and history of severe aGvHD.
Poliovirus type 1, 2, and 3 antibodies were found in 54-57% of cases before immunization. Within years after immunization, the percentages of seropositive patients gradually increased, up to 98% of patients having protective titres at >5 years. Negative titres were associated, e.g., with older age at immunization, negative initial poliovirus serum titres, the use of radiation during the preconditioning therapy, and positive recipient status.
Hence, the vaccination failure in different cohorts was associated with following factors: older age and negative titres at immunization; lower CD3, CD4 or CD19 counts; higher IgM concentrations; positive recipient CMV serology; acute or chronic graft-versus-host disease; and radiation during preconditioning. These data are of great value for re-immunization strategies in this cohort at infectious risk.
A real opportunity of anti-HBV immunity transfer via donor cells is shown in a study by Lindemann et al [23]. The workers selected a group of patients/matched donor pairs without pre-existing HBV immunity. Twenty-seven prospective donors were vaccinated against HBV. The donor vaccination produced HBs antibodies specific cellular responses in most of them. Interestingly, since 2 weeks post-HSCT, 86 and 67% of the recipients had positive humoral and cellular HBV reactions. Subsequently, HBV immunity re-appeared in 83% of the recipients without revaccination. These results show that a transfer of specific humoral and cellular immunity by allogeneic hematopoietic cell transplantation (HSCT) should, in principle, be possible.
Time course of latent virus activation in transplanted patients
High occurence of CMV in HSCT patients mostly results from reactivation of a latent virus which is acquired since early childhood. Many patients become seropositive at the age of 3 to 5 years, showing appreciable levels of specific IgG antibodies in most HSC recipients, thus suggestive for a primary infection in past time [41]. Most authors guess that CMV reactivation in most cases is observed in CMV-positive patients, being a clearly negative factor for clinical outcomes [25, 43]. CMV-associated clinical conditions are known since many years, especially, pneumonias, hepatitis, colitis, cystitis [49].
Time-course of CMV reactivation/reinfection may be determined either as higher CMV genome copies in the sample (quantitative DNA real-time assay), or as increased incidence of PCR-positive tests in subsequent sample series. The mean incidence of CMV positivity in peripheral blood after HSCT reaches its peak values 2 to 3 months following intensive therapy and transplantation [31].
CMV incidence and reactivation rates depend on intensity of conditioning treatment (non-myeloablative versus myeloablative therapy). E.g., a study of 537 CMV-seropositive patients has shown that a non-myeloablative conditioning was associated with lesser risk of subsequent CMV infection at early terms posttransplant [34]. However, nonmyeloablative regimen (TBI+Fludarabine) was, generally, followed by retarded CMV reactivation, as compared to myeloablative treatment [19]. Interestingly, a supplementary immunosuppression, e.g., treatment with antithymocyte globulin, or total lymphoid irradiation, correlated with earlier CMV viremia post-transplant, if compared with standard myeloablative therapy [43].
In fact, selective in vivo depletion of T cells, e.g., treatment with antithymocyte globulin prior to haplo-HSCT may cause a sufficient delay in CD4+T cell recovery, along with higher rates of CMV reactivation, as compared to the regimen with posttransplant cyclophosphamide [48]. These results suggest that the treatments with faster recovery of CD4+ T cells seem to better preserve antiviral immunity, thus allowing more efficient handling with herpesvirus infection after haplo-HSCT. Hence, targeted suppression of lymphoid cell populations pre-transplant is likely to cause higher incidence of CMV reactivation after HSCT.
Of special interest is early CMV activation in CMV-seropositive patients transplanted form CMV-negative donors. Since renewal of hematopoiesis in allo-HSCT setting proceeds with donor cells, one may expect decreased levels of anti-CMV specific T immunity, thus increasing risk of subsequent viremia [54].
In a similar HSCT cohort, we studied 143 patients with oncohematological disorders in a single-center study for herpesvirus reactivation post HSCT [9, 37]. The patients were subject to allogeneic transplantation of hematopoietic stem cells (HSCT). Occurrence and dynamics of acute GvHD was registered in posttransplant period, as well as other complications (pneumonia, neurological disorders, mucositis, cystitis). Reactivation of HSV, CMV and EBV was detectable in blood leukocytes after HSCT, respectively, in 51%, 57% and 45% of all cases (2.3-2.5-fold more common that pre-transplant). Primary diagnosis, gender of patients and type of transplant did not significantly influence these ratios.
Meanwhile, frequencies of CMV and HSV detection were dependent on patients’ age, with minimal detection rates at 1 to 4 years and a maximum at 10 to 20 years.
Prevalence of CMV positive tests was dependent upon the terms after HSCT, with a maximum at 2nd and 3rd months after transplantation (Fig.2A). As seen from previous data on cellular recovery, this time period is characterized by full restoration of innate immunity, and ongoing recovery maturation of adaptive lymphoid populations, e.g., CD4+ cells, and antibody-producing B cells. Similar post-transplant time dynamics was revealed for EBV reactivation (Fig 2B), thus presuming some analogies in reactivation mechanisms and type of interactions with host immune system. Herpes simplex (HSV) activation (Fig.2C) proceeded as soon as at 2nd- 3rd months after HSCT, probably, reflecting early affection of recipient epithelial cells, being its specific target population.

Finally, the incidence of post-transplant viremia depended on the patient’s age (Fig.3). I.e., in children of < 4 years old, the post-HSCT reactivation of cytomegalovirus was not frequent, being significantly increased (Р < 0.02) in older children (5 to 15 years old) post-transplant. Incidence of HSV reactivation was also maximal in adolescent recipients (Fig.4). These findings may be explained by lower infection levels in youngest children, maximal infection frequency in adolescents (6 to 15 years old), and sufficient antiviral immune response in older patients.
Like as recovery of immune populations, the CMV reactivation after HSCT may depend on a number of similar biological factors: 1) age of the patient; 3) intensity of conditioning regimen. Seropositivity of recipient is considered a special intrinsic factor for herpesvirus reactivation. All these factors should be taken into account when predicting risk for clinical CMV infection after intensive cytostatic treatment.

Along with studies of latent herpesviruses, some attention is now drawn to post-transplant reactivation of polyomaviruses (especially, BK and JC). E.g., BK virus is commonly found in urinary cell sediments by PCR approach, but it more frequently detected at 2-3 months post-HSCT [8]. Similar increase was observed for JC virus. Meanwhile, the BK and JC positivity in whole blood was not increased after HSCT, thus presuming only local reactivation of these viral pathogens.
Antibody response to cytomegalovirus reactivation
Since 1980s, it was proposed that both passive humoral immunity and specific T cell-mediated response contribute to the protective effect against CMV infection in allo-HSCT patients. Relative role of serological and cytotoxic immune responses in CMV infection was studied by Ludwig et al. (2006) who compared levels of glycoprotein B (gB)-specific antibodies and CMV-specific IFN-gamma producing CD4+ cells in CMV-positive SCT patients with/without CMV reactivation after SCT. The recipients without CMV reactivation showed a slow but steady increase in both parameters after SCT, indicating that initial high levels of gB specific antibodies or CMV specific CD4+ IFN-gamma+ cells are not necessary to prevent reactivation of CMV. Meanwhile, post-transplant CMV reactivation was associated with an increase in virus-specific CD4+ IFN-gamma+ cells prior to CMV activation, followed by a decline after the reactivation period. Patients who underwent only a single reactivation generated significant higher amounts of CD4+ IFN-gamma+ cells, than did patients with further reactivation episodes. Meanwhile, the course of gB specific antibodies for reactivating patients was something different, with significantly higher average values in the patients with CMV reactivation. This indicates that patients with a CMV reactivation exhibit a strong humoral dominated immune response.
Previous studies of protective anti-CMV antibodies in seropositive bone marrow recipients have shown induction of neutralizing anti-gB antibodies in cases of posttransplant CMV activation [40]. However, clinical significance of such neutralizing antibodies was not entirely confirmed in further studies [33]. The authors assessed time kinetics of the anti gB antibodies to CMV in 26 allo-HSC recipients. Neither absence, nor presence of CMV infection did correlate with serum concentrations of gB-specific and neutralizing antibodies. Recovery from CMV infection was not associated with antibody response to replication-associated antigens.
Potential clinical complications of EBV reactivation in transplanted patients
Among gamma-herpesviruses, the EBV is the mostly studied pathogen. Forty years ago, W and G. Henle had shown ability of EBV to infect B lymphocytes followed by their transformation to the lymphoblastoid virus-producing cells capable of long-term in vitro growth. Gamma-herpesviruses exhibit a well-known tropicity for lymphocytes, and, therefore, may induce lymphoproliferative disorders, as well as some muscular and endothelial neoplasias in humans [26]. The infected B-lymphoid cells reproduce normal stages of B cell differentiation in follicular germinal centers.
Latent EBV carriage is common to most adult subjects, as evidence by its common secretion with saliva. Uncontrolled proliferation of EBV-infected B cell in immunocompetent persons is blocked by cytotoxic lymphocytes. However, this mechanism of immune surveillance is largely failing in immunocompromised subjects [10, 36]. Therefore, EBV reactivation after transplantation of solid organs and hematopoietic stem cells is often followed by secondary posttransplant lymphoproliferative disorders (PTLD). Typically, the PTLDs emerge after long-term and massive immunosuppressive therapy which results into decreased amounts and functions of EBV-specific cytotoxic T cells, along with uncontrolled, EBV-driven B cell proliferation [10]. The EBV-positive malignancies observed in solid organ transplants, mostly originate from the recipient B cells, whereas 90% of PTLD in HSCT patients develop from the donor B cells [45].
Tischer et al. [48] have shown that 1/4 of patients pre-treated with ATG before HSCT developed PTLD, as compared to absence of this virally-induced condition among patients with presumably better preserved cytotoxic T cell populations.
Conclusions
Near-total depletion of innate and adaptive immune cell populations occurs due to intensive treatment followed by HSCT. Reconstitution of immune system from donor hematopoietic cells takes quite time periods needed for expansion, differentiation, migration and homing of granulocytes, monocytes, NK cells, T- and B cells. The intermediate maturation steps, especially, for lymphoid cell recovery, are known only to limited degree, and bear, mostly later terms (>1 month post-transplant).
Functional recovery of NK-cell subsets and blood monocytes may require up to 3 months; maturation and immune selection of CD8+ T cells proceeds for 3-4 months, whereas functional recovery of CD4+ T cells may be delayed for years, especially in older patients.
The B-cell ability to produce specific antibodies is partially retained, due to survival of resistant memory B cells post- HSCT, but differentiation and maturation of the donor-derived B cell hierarchy may require months and years.
Reactivation of herpesviruses in de novo produced donor cells, generally, proceeds in parallel with the periods of CD8+ and CD4+ T cell exhaustion, thus enabling expansion of latent herpesviruses, despite sufficient antibody response during CMV reactivation. Chronic EBV infection of B cells represents a more sufficient hazard, due to high risk of posttransplant lymphoproliferative disease at longer time range.
Hence, restoration of innate immunity, especially, monocytes/macrophages and NK cells originating from allografts, is most important at the early terms post-transplant, due to their immediate anti-infectious and GvL effects. At later terms (>100 d), deficient reconstitution of CD4+T cells may be responsible for deleterious infections with viruses and fungi, with older patients being at higher risk. To date, the delayed recovery of lymphoid subsets, especially, virus-specific T cells and memory B cells represents a major limitation for the long-term clinical outcomes in allo-transplanted patients. Therefore, current studies are aiming for enforcement of virus-targeted T-cell therapy, in order to accelerate clearance of cells with activated virus from affected tissues [32].
Conflict of interests
No conflict of interests is declared.
References
- Ali MY, Oyama Y, Monreal J, Winter J, Tallman M, Gordon LI, Williams S, Singhal S, Mehta J. Reassessing the definition of myeloid engraftment after autotransplantation: it is not necessary to see 0.5×10(9)/l neutrophils on 3 consecutive days to define myeloid recovery. Bone Marrow Transplant. 2002; 30 (11): 749-752.
- Avanzini MA, Locatelli F, Dos Santos C, Maccario R, Lenta E, Oliveri M, Giebel S, De Stefano P, Rossi F, Giorgiani G, Amendola G, Telli S, Marconi M. B lymphocyte reconstitution after hematopoietic stem cell transplantation: functional immaturity and slow recovery of memory CD27+ B cells. Exp Hematol 2005; 33 (4): 480-486.
- Baumgartner C, Morell A, Hirt A, Bucher U, Forster HK, Doran JE, Matter L, Brun del Re G, Wagner HP. Humoral immune function in pediatric patients treated with autologous bone marrow transplantation for B cell non-Hodgkin’s lymphoma. The influence of ex vivo marrow decontamination with anti-Y 29/55 monoclonal antibody and complement. Blood. 1988;71(5):1211-1217.
- Bemark M, Holmqvist J, Abrahamsson J, Mellgren K. Translational Mini-Review Series on B cell subsets in disease. Reconstitution after haematopoietic stem cell transplantation - revelation of B cell developmental pathways and lineage phenotypes. Clin Exp Immunol. 2012;167(1):15-25.
- Chalandon Y, Degermann S, Villard J, Arlettaz L, Kaiser L, Vischer S, Walter S, Heemskerk MH, van Lier RA, Helg C, Chapuis B, Roosnek E. Pre-transplantation CMV-specific T cells protect recipients of T-cell-depleted grafts against CMV-related complications. Blood 2006; 107:389–396.
- Chemaly RF, Shah DP, Boeckh MJ. Management of respiratory viral infections in hematopoietic cell transplant recipients and patients with hematologic malignancies. Clin Infect Dis. 2014; 59 (Suppl 5): S344-S351.
- Chukhlovin A. Enhanced ex vivo apoptosis of peripheral granulocytes is a sufficient factor of neutropenia following myeloablative chemotherapy. Leuk Res 2000; 24(6):507-509.
- Chukhlovin AB, Eismont YuA, Vavilov VN, Zubarovskaya LS, Afanasyev BV. Time- and sample-dependent differences in polyomavirus incidence following hematopoietic stem cell transplantation. Cell Ther Transplant. 2016; 5(1): 26-30.
- Chukhlovin A, Ovsyannikova M, Semenova E, Pankratova O, Zubarovskaya L, Afanasyev B Incidence of herpesvirus activation in young HSCT patients. 33 EBMT Meeting, Lyon, France, 2007. Abstr. P719.
- Cohen JI, Bollard CM, Khanna R, Pittaluga S. Current understanding of the role of Epstein-Barr virus (EBV) in lymphomagenesis and therapeutic approaches to EBV associated lymphomas. Leuk Lymphoma 2008;49(Suppl 1):27-34.
- Dean HF, Cazaly A, Hurlock C, Borras J, Williams AP, Johnson PW, Davies AJ. Defects in lymphocyte subsets and serological memory persist a median of 10 years after high-dose therapy and autologous progenitor cell rescue for malignant lymphoma. Bone Marrow Transplant. 2012;47(12):1545-1551.
- de Koning C, Plantinga M, Besseling P, Boelens JJ, Nierkens S. Immune reconstitution after allogeneic hematopoietic cell transplantation in children. Biol Blood Marrow Transplant. 2016; 22(2):195-206.
- Gerritsen EJ, van Tol MJ, Lankester AC, van der Weijden-Ragas CP, Jol-van der Zijde CM, Oudeman-Gruber NJ, Radl J, Vossen JM. Immunoglobulin levels and monoclonal gammopathies in children after bone marrow transplantation. Blood 1993; 82:3493–3502
- Gismondi A, Stabile H, Nisti P, Santoni A. Effector functions of natural killer cell subsets in the control of hematological malignancies. Front. Immunol. 6:567. doi: 10.3389/ fimmu.2015.00567
- Gratama JW, Naipal A, Oljans P, Zwaan FE, Verdonck LF, de Witte T, Vossen JM, Bolhuis RL, de Gast GC, Jansen J. T lymphocyte repopulation and differentiation after bone marrow transplantation. Early shifts in the ratio between T4+ and T8+ T lymphocytes correlate with the occurrence of acute graft-versus-host disease. Blood 1984;63:1416–1423.
- Gress RE, Emerson SG, Drobyski WR. Immune reconstitution: how it should work, what’s broken, and why it matters. Biol Blood Marrow Transplant. 2010; 16(1 Suppl):S133-137.
- Hakim FT, Cepeda R, Kaimei S, Mackall CL, McAtee N, Zujewski J, Cowan K, Gress RE. Constraints on CD4 recovery postchemotherapy in adults: Thymic insufficiency and apoptotic decline of expanded peripheral CD4 cells. Blood 1997;90 (9): 3789–3798
- Hakim FT, Memon SA, Cepeda R, Jones EC, Chow CK, Kasten-Sportes C, Odom J, Vance BA, Christensen BL, Mackall CL, Gress RE. Age-dependent incidence, time course, and consequences of thymic renewal in adults. J Clin Invest 2005;115(4):930–939
- Junghanss C, Boeckh M, Carter RA, Sandmaier BM, Maris MB, Maloney DG, Chauncey T, McSweeney PA, Little MT, Corey L, Storb R. Incidence and outcome of cytomegalovirus infections following nonmyeloablative compared with myeloablative allogeneic stem cell transplantation, a matched control study. Blood. 2002;99(6):1978-1985.
- Inaba H, Hartford CM, Pei D, Posner MJ, Yang J, Hayden RT, Srinivasan A, Triplett BM, McCulllers JA, Pui CH, Leung W. Longitudinal analysis of antibody response to immunization in paediatric survivors after allogeneic haematopoietic stem cell transplantation. Br J Haematol. 2012;156(1):109- 117.
- Klyuchnikov E, Asenova S, Kern W, Kilinc G, Ayuk F, Wiedemann B, Lioznov M, Freiberger P, Zalyalov Y, Zander AR, Kröger N, Bacher U. Post-transplant immune reconstitution after unrelated allogeneic stem cell transplant in patients with acute myeloid leukemia.Leuk Lymphoma. 2010;51(8):1450-1463.
- Koehne G, Zeller W, Stockschlaeder M, Zander AR. Phenotype of lymphocyte subsets after autologous peripheral blood stem cell transplantation. Bone Marrow Transplant. 1997;19(2):149-156.
- Lindemann M, Barsegian V, Runde V, Fiedler M, Heermann KH, Schaefer UW, Roggendorf M, Grosse-Wilde H. Transfer of humoral and cellular hepatitis B immunity by allogeneic hematopoietic cell transplantation. Transplantation. 2003 ;75(6):833-838.
- Ljungman P, Avetisyan G. Influenza vaccination in hematopoietic SCT recipients. Bone Marrow Transplant. 2008; 42(10): 637-641.
- Ljungman P. The role of cytomegalovirus serostatus on outcome of hematopoietic stem cell transplantation. Curr Opin Hematol. 2014 Nov;21(6):466-9.
- Longnecker R., Neipel F. Introduction to the human gamma-herpesviruses. In: Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis (Ed. A. Arvin, G. Campadelli- Fiume et al.). Cambridge University Press, 2007, Chapter 22.
- Lucas KG, Small TN, Heller G, Dupont B, O’Reilly RJ. The development of cellular immunity to Epstein–Barr virus after allogeneic bone marrow transplantation. Blood 1996;87:2594–2603.
- Ludwig B, Kraus FB, Kipp M, Preiser W, Schwerdtfeger R, Doerr HW, Buxbaum S. Cytomegalovirus-specific CD4 T-cell and glycoprotein B specific antibody response in recipients of allogenic stem cell transplantation. J Clin Virol. 2006; 35(2): 160-166.
- Lum LG, Seigneuret MC, Orcutt-Thordarson N, Noges JE, Storb R. The regulation of immunoglobulin synthesis after HLA-identical bone marrow transplantation: VI. Differential rates of maturation of distinct functional groups within lymphoid subpopulations in patients after human marrow grafting. Blood. 1985;65(6):1422-1433.
- Mackall CL, Fleisher TA, Brown MR, et al. Age, thymopoiesis, and CD4+ T-lymphocyte regeneration after intensive chemotherapy. New Engl J Med 1995; 332(3):143–149.
- Maeda Y, Teshima T, Yamada M, Shinagawa K, Nakao S, Ohno Y, Kojima K, Hara M, Nagafuji K, Hayashi S, Fukuda S, Sawada H, Matsue K, Takenaka K, Ishimaru F, Ikeda K, Niiya K, Harada M. Monitoring of human herpesviruses after allogeneic peripheral blood stem cell transplantation and bone marrow transplantation. Br J Haematol. 1999; 105(1):295-302.
- Moosmann A, Hammerschmidt W, Kolb HJ. Virus-specific T cells for therapy--approaches, problems, solutions. Eur J Cell Biol. 2012;91(1):97-101.
- Muñoz I, Gutiérrez A, Gimeno C, Farga A, Alberola J, Solano C, Prósper F, García-Conde J, Navarro D. Lack of association between the kinetics of human cytomegalovirus (HCMV) glycoprotein B (gB)-specific and neutralizing serum antibodies and development or recovery from HCMV active infection in patients undergoing allogeneic stem cell transplant. J Med Virol. 2001; 65(1): 77-84.
- Nakamae H, Kirby KA, Sandmaier BM, Norasetthada L, Maloney DG, Maris MB, Davis C, Corey L, Storb R, Boeckh M. Effect of Conditioning Regimen Intensity on CMV Infection in Allogeneic Hematopoietic Cell Transplantation. Biol Blood Marrow Transplant. 2009; 15(6): 694–703.
- Nguyen S, Dhedin N ,Vernant JP, Kuentz M, AlJijakli A, Rouas-Freiss N, et al. NK-cell reconstitution after haploidentical hematopoietic stem-cell transplantations: immaturity of NK cells and inhibitory effect of NKG2A override GvL effect. Blood 2005; 105:4135–4142.
- Nourse JP, Jones K, Gandhi MH Epstein-Barr-virus-related post-transplant lymphoproliferative disorders: pathogenetic insights for targeted therapy. Am J Transpl 2011; 11(5): 888-895.
- Pankratova OS, Chukhlovin AB, Zubarovskaya LS, Afanasyev BV. Incidence of herpesvirus detection and risk of common complications in allogeneic hematopoietic stem cell transplantation. Scientific Notes of the St.Petersburg State Medical University 2010; 17(1):56-60. (In Russian)
- Pankratova OS, Chukhlovin AB, Shiryaev SN, Eismont YA, Vavilov VN, Zubarovskaya LS, Afanasyev BV. Herpesviruses and oral ulcerations in hematopoietic SCT recipients. Bone Marrow Transplant. 2013; 48(10):1364-1365.
- Pechumer H, Leinisch E, Bender-Götze C, Ziegler-Heitbrock HW. Recovery of monocytes after bone marrow transplantation rapid reappearance of tumor necrosis factor alpha and interleukin 6 production. Transplantation. 1991; 52(4): 698-704.
- Roy DM, Brenner MK, Cook D, Duncan RI, Griffiths PD, Grundy JE. Transfer of humoral immunity against cytomegalovirus proteins following transplantation of T-cell-depleted allogeneic bone marrow from seropositive donors. J Med Virol. 1993; 41(2): 150-158.
- Ruell J, Barnes C, Mutton K, Foulkes B, Chang J, Cavet J, Guiver M, Menasce L, Dougal M, Chopra R. Active CMV disease does not always correlate with viral load detection. Bone Marrow Transplantation. 2007; 40: 55–61.
- Sahdev I, O’Reilly R, Black P, Heller G, HoffmannM. Interleukin-1 production following T-cell-depleted and unmodified marrow grafts. Pediatr Hematol Oncol 1996; 13:55–67.
- Schaenman JM, Shashidhar S, Rhee C, Wong J, Navato S, Wong RM, Ho DY, Arai S, Johnston L, Brown JM. Early CMV viremia is associated with impaired viral control following nonmyeloablative hematopoietic cell transplantation with a total lymphoid irradiation and antithymocyte globulin preparative regimen. Biol Blood Marrow Transplant 2011;17:693-702.
- Seggewiss R, Einsele H. Immune reconstitution after allogeneic transplantation and expanding options for immunomodulation: an update. Blood. 2010; 115(19): 3861–3868.
- Snow AL, Martinez OM. Epstein-Barr virus: evasive maneuvers in the development of PTLD. Am J Transplant 2007; 7(2):271–277.
- Storek J, Joseph A, Espino G, Dawson MA, Douek DC, Sullivan KM, Flowers ME, Martin P, Mathioudakis G, Nash RA, Storb R, Appelbaum FR, Maloney DG. Immunity of patients surviving 20 to 30 years after allogeneic or syngeneic bone marrow transplantation. Blood 2001; 98:3505–3512.
- Storek J, Geddes M, Khan F, Huard B, Helg C, Chalandon Y, Passweg J, Roosnek E. Reconstitution of the immune system after hematopoietic stem cell transplantation in humans. Semin Immunopathol. 2008; 30(4): 425-437.
- Tischer J, Engel N, Fritsch S, Prevalsek D, Hubmann M, Schulz C, Zoellner AK, Bücklein V, Reibke R, Mumm F, Rieger CT, Hill W, Ledderose G, Stemmler HJ, Köhnke T, Jäger G, Kolb HJ, Schmid C, Moosmann A, Hausmann A. Virus infection in HLA-haploidentical hematopoietic stem cell transplantation: incidence in the context of immune recovery in two different transplantation settings. Ann Hematol. 2015;94(10):1677-1688.
- Travi G, Pergam SA. Cytomegalovirus pneumonia in hematopoietic stem cell recipients. J Intensive Care Med. 2014; 29(4):200-212.
- Tzannou I, Leen AM. Accelerating immune reconstitution after hematopoietic stem cell transplantation. Clin Transl Immunology. 2014; 3(2):e11. doi: 10.1038/cti.2014.2. eCollection 2014.
- Vago L, Forno B, Sormani MP, Crocchiolo R, Zino E, DiTerlizzi S, etal. Temporal, quantitative, and functional characteristics of single-KIR-positive alloreactive natural killer cell recovery account for impaired graft-versus- leukemia activity after haploidentical hematopoietic stem cell transplantation. Blood. 2008; 112:3488–3499.
- van Tol MJ, Gerritsen EJ, de Lange GG, van Leeuwen AM, Jol-van der Zijde CM, Oudeman-Gruber NJ, de Vries E, Radl J, Vossen JM. The origin of IgG production and homogeneous IgG components after allogeneic bone marrow transplantation. Blood 1996; 87:818–826.
- Wald A, Corey L. Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis (ed. A. Arvin, G. Campadelli-Fiume et al.) Cambridge University Press. – 2007. – Chapter 36. URL: http://www.ncbi.nlm.nih.gov/books/NBK47376/
- Zhou W, Longmate J, Lacey SF, Palmer JM, Gallez-Hawkins G, Thao L, Spielberger R, Nakamura R, Forman SJ, Zaia JA, Diamond DJ. Impact of donor CMV status on viral infection and reconstitution of multifunction CMV-specific T cells in CMV-positive transplant recipients. Blood 2009;113(25):6465-6476.
- Zimmerli W, Zarth A, Gratwohl A, Speck B. Neutrophil function and pyogenic infections in bone marrow transplant recipients. Blood 1991; 77:393–399.
- Zucca E, Bertoni F, Vannata B, Cavalli F. Emerging role of infectious etiologies in the pathogenesis of marginal zone B-cell lymphomas. Clin Cancer Res. 2014; 20(20): 5207- 5216.
" ["DETAIL_TEXT_TYPE"]=> string(4) "html" ["~DETAIL_TEXT_TYPE"]=> string(4) "html" ["PREVIEW_TEXT"]=> string(0) "" ["~PREVIEW_TEXT"]=> string(0) "" ["PREVIEW_TEXT_TYPE"]=> string(4) "text" ["~PREVIEW_TEXT_TYPE"]=> string(4) "text" ["PREVIEW_PICTURE"]=> NULL ["~PREVIEW_PICTURE"]=> NULL ["LANG_DIR"]=> string(4) "/ru/" ["~LANG_DIR"]=> string(4) "/ru/" ["SORT"]=> string(3) "500" ["~SORT"]=> string(3) "500" ["CODE"]=> string(100) "vremennaya-dinamika-vosstanovleniya-immuniteta-i-virusnaya-reaktivatsiya-posle-transplantatsii-gemop" ["~CODE"]=> string(100) "vremennaya-dinamika-vosstanovleniya-immuniteta-i-virusnaya-reaktivatsiya-posle-transplantatsii-gemop" ["EXTERNAL_ID"]=> string(3) "678" ["~EXTERNAL_ID"]=> string(3) "678" ["IBLOCK_TYPE_ID"]=> string(7) "journal" ["~IBLOCK_TYPE_ID"]=> string(7) "journal" ["IBLOCK_CODE"]=> string(7) "volumes" ["~IBLOCK_CODE"]=> string(7) "volumes" ["IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["~IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["LID"]=> string(2) "s2" ["~LID"]=> string(2) "s2" ["EDIT_LINK"]=> NULL ["DELETE_LINK"]=> NULL ["DISPLAY_ACTIVE_FROM"]=> string(0) "" ["IPROPERTY_VALUES"]=> array(18) { ["ELEMENT_META_TITLE"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["ELEMENT_META_KEYWORDS"]=> string(266) "трансплантация гемопоэтических стволовых клеток восстановление иммунитета различия в созревании временная динамика герпесвирусы реактивация" ["ELEMENT_META_DESCRIPTION"]=> string(334) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клетокTime course of immune recovery and viral reactivation following hematopoietic stem cell transplantation" ["ELEMENT_PREVIEW_PICTURE_FILE_ALT"]=> string(5526) "<p> Полное истощение клеточных популяций врожденного и адаптивного иммунитета возникает после интенсивной химиотерапии и трансплантации гемопоэтических стволовых клеток (ТГСК), за чем следует постепенное восстановление иммунной системы за счет предшественников, образующихся из донорских гемопоэтических клеток, которые дифференцируются в миелоидный и лимфоидные ростки. Временная динамика восстановления иммунной системы и, в частности, отдельных клеточных популяций изучена лишь отчасти, особенно в ранние сроки после ТГСК. Например, популяции клеток врожденного иммунитета восстанавливаются в течение 1-го месяца после ТГСК, в связи с быстрым обновлением популяций гранулоцитов, моноцитов и НК-клеток. В то же время функциональное созревание субпопуляций НК-клеток и моноцитов крови может длиться до 3 мес. </p> <p> Как Т-, так и В-лимфоциты восстанавливаются гораздо медленнее, чем миеломоноцитарные популяции. Есть лишь ограниченная информация об их посттрансплантационном обновлении, так как большинство исследований проводились в поздние сроки (>1 мес. после ТГСК). Так, абсолютное число CD8+ T-клеток нормализуется примерно через 4 мес., при суженном спектре Т-лимфоцитов памяти. Темпы восстановления и созревания CD4+ T во многом зависят от остаточной функции тимуса, особенно в младших возрастах. Поэтому у младших пациентов преобладает популяция «наивных» CD4+ T cell в сроки >6 мес. после ТГСК. У лиц старшего возраста с инактивацией функции тимуса общее число CD4 клеток остается низким годами после ТГСК. В то же время антивирусный клеточный иммунитет активен, начиная с ранних сроков после трансплантации. Так, цитотоксические CD8+ клетки, специфичные в отношении ЦМВ или ВЭБ, быстро размножаются в случаях реактивации этих вирусов после ТГСК. </p> <p> Несмотря на восстановление абсолютных количеств В-клеток к 30-му дню после ТГСК, их функции (в т.ч. - антиген-специфическая продукция антител) снижены многие месяцы и годы после ТГСК из-за медленного восстановления зрелых популяций иммунных клеток, что напоминает постепенную эволюцию в иерархии В-клеток в организме человека. </p> <p> Реактивация герпесвирусов (в основном –ЦМВ, ВЭБ и ВПГ) является известным признаком иммунодефицита. Периоды максимального риска реактивации герпесвирусов (2-3 мес. после ТГСК) соответствуют периодам функционального дефицита CD8+ и CD4+ T-клеток, а также незрелости В-клеток, что отражает их недостаточную способность к устранению лейкоцитов, пораженных герпесвирусами. Индивидуальные сроки иммунного восстановления после ТГСК зависят от возраста больного, источника донорских клеток, развития острой РТПХ после трансплантации и др. Ответ на вакцинирование – важный критерии состояния иммунитета после ТГСК – также зависит от возраста больного и сохранности функций В-клеток. Временная динамика специфического антительного ответа показывает, что больные с латентной реактивацией ЦМВ могут позже проявить сильный гуморальный иммунитет, что ведет к ограничению инфекционного процесса. </p>" ["ELEMENT_PREVIEW_PICTURE_FILE_TITLE"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["ELEMENT_DETAIL_PICTURE_FILE_ALT"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["ELEMENT_DETAIL_PICTURE_FILE_TITLE"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["SECTION_META_TITLE"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["SECTION_META_KEYWORDS"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["SECTION_META_DESCRIPTION"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["SECTION_PICTURE_FILE_ALT"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["SECTION_PICTURE_FILE_TITLE"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["SECTION_PICTURE_FILE_NAME"]=> string(100) "vremennaya-dinamika-vosstanovleniya-immuniteta-i-virusnaya-reaktivatsiya-posle-transplantatsii-gemop" ["SECTION_DETAIL_PICTURE_FILE_ALT"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["SECTION_DETAIL_PICTURE_FILE_TITLE"]=> string(231) "Временная динамика восстановления иммунитета и вирусная реактивация после трансплантации гемопоэтических стволовых клеток" ["SECTION_DETAIL_PICTURE_FILE_NAME"]=> string(100) "vremennaya-dinamika-vosstanovleniya-immuniteta-i-virusnaya-reaktivatsiya-posle-transplantatsii-gemop" ["ELEMENT_PREVIEW_PICTURE_FILE_NAME"]=> string(100) "vremennaya-dinamika-vosstanovleniya-immuniteta-i-virusnaya-reaktivatsiya-posle-transplantatsii-gemop" ["ELEMENT_DETAIL_PICTURE_FILE_NAME"]=> string(100) "vremennaya-dinamika-vosstanovleniya-immuniteta-i-virusnaya-reaktivatsiya-posle-transplantatsii-gemop" } ["FIELDS"]=> array(1) { ["IBLOCK_SECTION_ID"]=> string(2) "20" } ["PROPERTIES"]=> array(18) { ["KEYWORDS"]=> array(36) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(6) { [0]=> string(4) "8318" [1]=> string(4) "8319" [2]=> string(4) "8320" [3]=> string(4) "8321" [4]=> string(4) "8322" [5]=> string(4) "8323" } ["VALUE"]=> array(6) { [0]=> string(2) "15" [1]=> string(3) "674" [2]=> string(3) "675" [3]=> string(3) "676" [4]=> string(3) "677" [5]=> string(3) "647" } ["DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(6) { [0]=> string(2) "15" [1]=> string(3) "674" [2]=> string(3) "675" [3]=> string(3) "676" [4]=> string(3) "677" [5]=> string(3) "647" } ["~DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" } ["SUBMITTED"]=> array(36) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8282" ["VALUE"]=> string(22) "11/02/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/02/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL } ["ACCEPTED"]=> array(36) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8283" ["VALUE"]=> string(22) "12/09/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "12/09/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL } ["PUBLISHED"]=> array(36) { ["ID"]=> string(2) "22" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Дата публикации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "PUBLISHED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "22" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Дата публикации" ["~DEFAULT_VALUE"]=> NULL } ["CONTACT"]=> array(36) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHORS"]=> array(36) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> bool(false) ["VALUE"]=> bool(false) ["DESCRIPTION"]=> bool(false) ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> bool(false) ["~DESCRIPTION"]=> bool(false) ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_RU"]=> array(36) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8284" ["VALUE"]=> array(2) { ["TEXT"]=> string(120) "Ольга С. Панкратова<sup>1</sup>, Алексей Б. Чухловин<sup>2</sup>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(96) "Ольга С. Панкратова1, Алексей Б. Чухловин2" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_RU"]=> array(36) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8285" ["VALUE"]=> array(2) { ["TEXT"]=> string(502) "<sup>1</sup>Госпиталь Университета г. Тампере, Финляндия <br> <sup>2</sup>НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский<br> государственный медицинский университет им. акад. И. П.Павлова, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(466) "1Госпиталь Университета г. Тампере, Финляндия
2НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. акад. И. П.Павлова, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_RU"]=> array(36) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8286" ["VALUE"]=> array(2) { ["TEXT"]=> string(5526) "<p> Полное истощение клеточных популяций врожденного и адаптивного иммунитета возникает после интенсивной химиотерапии и трансплантации гемопоэтических стволовых клеток (ТГСК), за чем следует постепенное восстановление иммунной системы за счет предшественников, образующихся из донорских гемопоэтических клеток, которые дифференцируются в миелоидный и лимфоидные ростки. Временная динамика восстановления иммунной системы и, в частности, отдельных клеточных популяций изучена лишь отчасти, особенно в ранние сроки после ТГСК. Например, популяции клеток врожденного иммунитета восстанавливаются в течение 1-го месяца после ТГСК, в связи с быстрым обновлением популяций гранулоцитов, моноцитов и НК-клеток. В то же время функциональное созревание субпопуляций НК-клеток и моноцитов крови может длиться до 3 мес. </p> <p> Как Т-, так и В-лимфоциты восстанавливаются гораздо медленнее, чем миеломоноцитарные популяции. Есть лишь ограниченная информация об их посттрансплантационном обновлении, так как большинство исследований проводились в поздние сроки (>1 мес. после ТГСК). Так, абсолютное число CD8+ T-клеток нормализуется примерно через 4 мес., при суженном спектре Т-лимфоцитов памяти. Темпы восстановления и созревания CD4+ T во многом зависят от остаточной функции тимуса, особенно в младших возрастах. Поэтому у младших пациентов преобладает популяция «наивных» CD4+ T cell в сроки >6 мес. после ТГСК. У лиц старшего возраста с инактивацией функции тимуса общее число CD4 клеток остается низким годами после ТГСК. В то же время антивирусный клеточный иммунитет активен, начиная с ранних сроков после трансплантации. Так, цитотоксические CD8+ клетки, специфичные в отношении ЦМВ или ВЭБ, быстро размножаются в случаях реактивации этих вирусов после ТГСК. </p> <p> Несмотря на восстановление абсолютных количеств В-клеток к 30-му дню после ТГСК, их функции (в т.ч. - антиген-специфическая продукция антител) снижены многие месяцы и годы после ТГСК из-за медленного восстановления зрелых популяций иммунных клеток, что напоминает постепенную эволюцию в иерархии В-клеток в организме человека. </p> <p> Реактивация герпесвирусов (в основном –ЦМВ, ВЭБ и ВПГ) является известным признаком иммунодефицита. Периоды максимального риска реактивации герпесвирусов (2-3 мес. после ТГСК) соответствуют периодам функционального дефицита CD8+ и CD4+ T-клеток, а также незрелости В-клеток, что отражает их недостаточную способность к устранению лейкоцитов, пораженных герпесвирусами. Индивидуальные сроки иммунного восстановления после ТГСК зависят от возраста больного, источника донорских клеток, развития острой РТПХ после трансплантации и др. Ответ на вакцинирование – важный критерии состояния иммунитета после ТГСК – также зависит от возраста больного и сохранности функций В-клеток. Временная динамика специфического антительного ответа показывает, что больные с латентной реактивацией ЦМВ могут позже проявить сильный гуморальный иммунитет, что ведет к ограничению инфекционного процесса. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(5470) "
Полное истощение клеточных популяций врожденного и адаптивного иммунитета возникает после интенсивной химиотерапии и трансплантации гемопоэтических стволовых клеток (ТГСК), за чем следует постепенное восстановление иммунной системы за счет предшественников, образующихся из донорских гемопоэтических клеток, которые дифференцируются в миелоидный и лимфоидные ростки. Временная динамика восстановления иммунной системы и, в частности, отдельных клеточных популяций изучена лишь отчасти, особенно в ранние сроки после ТГСК. Например, популяции клеток врожденного иммунитета восстанавливаются в течение 1-го месяца после ТГСК, в связи с быстрым обновлением популяций гранулоцитов, моноцитов и НК-клеток. В то же время функциональное созревание субпопуляций НК-клеток и моноцитов крови может длиться до 3 мес.
Как Т-, так и В-лимфоциты восстанавливаются гораздо медленнее, чем миеломоноцитарные популяции. Есть лишь ограниченная информация об их посттрансплантационном обновлении, так как большинство исследований проводились в поздние сроки (>1 мес. после ТГСК). Так, абсолютное число CD8+ T-клеток нормализуется примерно через 4 мес., при суженном спектре Т-лимфоцитов памяти. Темпы восстановления и созревания CD4+ T во многом зависят от остаточной функции тимуса, особенно в младших возрастах. Поэтому у младших пациентов преобладает популяция «наивных» CD4+ T cell в сроки >6 мес. после ТГСК. У лиц старшего возраста с инактивацией функции тимуса общее число CD4 клеток остается низким годами после ТГСК. В то же время антивирусный клеточный иммунитет активен, начиная с ранних сроков после трансплантации. Так, цитотоксические CD8+ клетки, специфичные в отношении ЦМВ или ВЭБ, быстро размножаются в случаях реактивации этих вирусов после ТГСК.
Несмотря на восстановление абсолютных количеств В-клеток к 30-му дню после ТГСК, их функции (в т.ч. - антиген-специфическая продукция антител) снижены многие месяцы и годы после ТГСК из-за медленного восстановления зрелых популяций иммунных клеток, что напоминает постепенную эволюцию в иерархии В-клеток в организме человека.
Реактивация герпесвирусов (в основном –ЦМВ, ВЭБ и ВПГ) является известным признаком иммунодефицита. Периоды максимального риска реактивации герпесвирусов (2-3 мес. после ТГСК) соответствуют периодам функционального дефицита CD8+ и CD4+ T-клеток, а также незрелости В-клеток, что отражает их недостаточную способность к устранению лейкоцитов, пораженных герпесвирусами. Индивидуальные сроки иммунного восстановления после ТГСК зависят от возраста больного, источника донорских клеток, развития острой РТПХ после трансплантации и др. Ответ на вакцинирование – важный критерии состояния иммунитета после ТГСК – также зависит от возраста больного и сохранности функций В-клеток. Временная динамика специфического антительного ответа показывает, что больные с латентной реактивацией ЦМВ могут позже проявить сильный гуморальный иммунитет, что ведет к ограничению инфекционного процесса.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["DOI"]=> array(36) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8287" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-32-43" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-32-43" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_EN"]=> array(36) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8288" ["VALUE"]=> array(2) { ["TEXT"]=> string(88) "<sup>1</sup>Olga S. Pankratova, <sup>2</sup>Alexei B. Chukhlovin" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(64) "1Olga S. Pankratova, 2Alexei B. Chukhlovin" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_EN"]=> array(36) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8289" ["VALUE"]=> array(2) { ["TEXT"]=> string(256) "<sup>1</sup>Tampere University Hospital, Tampere, Finland <br> <sup>2</sup>R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(226) "1Tampere University Hospital, Tampere, Finland2R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_EN"]=> array(36) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8290" ["VALUE"]=> array(2) { ["TEXT"]=> string(2944) "<p> Total depletion of innate and adaptive immune cell populations occurs after intensive chemotherapy and hematopoietic stem cell transplantation (HSCT) then followed by gradual recovery of immune populations, due to progenitors derived from donor hematopoietic cells which differentiate to myeloid and lymphoid lineages. Time dynamics of immune reconstitution and differential maturation of distinct immune populations is only partially evaluated, especially, at early terms post-transplant. E.g., innate immunity is restored within 1st month after HSCT, due to rapid reconstitution of granulocytes, monocytes, and natural killer (NK) cells. Meanwhile, functional recovery of mature NK-cell subsets and blood monocytes may continue for up to 3 months. </p> <p> Both T- and B-lymphocyte pools are restored much slower than myelomonocytic populations. The available information on their post-HSCT immune recovery is limited, since most studies are performed at later terms (>1 month post-transplant). Absolute numbers of CD8+ T cells return to control values ca. 4 months post-HSCT, however, exhibiting a skewed repertoire of memory T lymphocytes. Recovery and maturation rates of CD4+ T cells largely depend on residual thymus function, especially, in young subjects. Hence, a naïve CD4+ T cell population in pediatric patients predominates over 6 months post-HSCT. In older persons with inactive thymopoiesis the total CD4 cell counts remain low for years after HSCT. Meanwhile, antiviral cellular immunity is active since early terms post-transplant. E.g., cytotoxic CD8+ cells specific for cytomegalovirus (CMV), or Epstein- Barr virus (EBV) rapidly expand in cases of CMV or EBV activation. </p> <p> Despite recovery of absolute B-cell counts by day 30 post-HSCT, their functions, i.e., antigen-specific antibody production, are reduced for months and years after HSCT, due to slow restoration of mature immune cell populations, thus resemling normal evolution of B cell hierarchy in human organism. </p> <p> Reactivation of herpesviruses (mostly, CMV, EBV and Herpes Simplex) is a known feature of immune deficiency. Timing of maximal herpesvirus incidence (2-3 months post-HSCT) corresponds to the period of CD8+ and CD4+ T cell functional deficiency and B cell immaturity, thus reflecting their suboptimal ability to eliminate herpesvirus-affected leukocytes. Individual terms of immune recovery after allo-HSCT depend on the patients’ age, source of donor cells, acute GvHD post-HSCT etc. Vaccination response, being a potent in vivo criterion of immune recovery in post-transplant patients, is also dependent on the subjects’ age and restored B cell functions. Time dynamics of specific antibody response shows that the patients with latent CMV reactivation may later exhibit a strong humoral immune response, thus making the infection self-limiting. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2892) "
Total depletion of innate and adaptive immune cell populations occurs after intensive chemotherapy and hematopoietic stem cell transplantation (HSCT) then followed by gradual recovery of immune populations, due to progenitors derived from donor hematopoietic cells which differentiate to myeloid and lymphoid lineages. Time dynamics of immune reconstitution and differential maturation of distinct immune populations is only partially evaluated, especially, at early terms post-transplant. E.g., innate immunity is restored within 1st month after HSCT, due to rapid reconstitution of granulocytes, monocytes, and natural killer (NK) cells. Meanwhile, functional recovery of mature NK-cell subsets and blood monocytes may continue for up to 3 months.
Both T- and B-lymphocyte pools are restored much slower than myelomonocytic populations. The available information on their post-HSCT immune recovery is limited, since most studies are performed at later terms (>1 month post-transplant). Absolute numbers of CD8+ T cells return to control values ca. 4 months post-HSCT, however, exhibiting a skewed repertoire of memory T lymphocytes. Recovery and maturation rates of CD4+ T cells largely depend on residual thymus function, especially, in young subjects. Hence, a naïve CD4+ T cell population in pediatric patients predominates over 6 months post-HSCT. In older persons with inactive thymopoiesis the total CD4 cell counts remain low for years after HSCT. Meanwhile, antiviral cellular immunity is active since early terms post-transplant. E.g., cytotoxic CD8+ cells specific for cytomegalovirus (CMV), or Epstein- Barr virus (EBV) rapidly expand in cases of CMV or EBV activation.
Despite recovery of absolute B-cell counts by day 30 post-HSCT, their functions, i.e., antigen-specific antibody production, are reduced for months and years after HSCT, due to slow restoration of mature immune cell populations, thus resemling normal evolution of B cell hierarchy in human organism.
Reactivation of herpesviruses (mostly, CMV, EBV and Herpes Simplex) is a known feature of immune deficiency. Timing of maximal herpesvirus incidence (2-3 months post-HSCT) corresponds to the period of CD8+ and CD4+ T cell functional deficiency and B cell immaturity, thus reflecting their suboptimal ability to eliminate herpesvirus-affected leukocytes. Individual terms of immune recovery after allo-HSCT depend on the patients’ age, source of donor cells, acute GvHD post-HSCT etc. Vaccination response, being a potent in vivo criterion of immune recovery in post-transplant patients, is also dependent on the subjects’ age and restored B cell functions. Time dynamics of specific antibody response shows that the patients with latent CMV reactivation may later exhibit a strong humoral immune response, thus making the infection self-limiting.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["NAME_EN"]=> array(36) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8291" ["VALUE"]=> string(103) "Time course of immune recovery and viral reactivation following hematopoietic stem cell transplantation" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(103) "Time course of immune recovery and viral reactivation following hematopoietic stem cell transplantation" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" } ["FULL_TEXT_RU"]=> array(36) { ["ID"]=> string(2) "42" ["TIMESTAMP_X"]=> string(19) "2015-09-07 20:29:18" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(23) "Полный текст" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(12) "FULL_TEXT_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "42" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(23) "Полный текст" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["PDF_RU"]=> array(36) { ["ID"]=> string(2) "43" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF RUS" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_RU" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "43" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8292" ["VALUE"]=> string(3) "257" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "257" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF RUS" ["~DEFAULT_VALUE"]=> string(0) "" } ["PDF_EN"]=> array(36) { ["ID"]=> string(2) "44" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF ENG" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "44" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8293" ["VALUE"]=> string(3) "258" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "258" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF ENG" ["~DEFAULT_VALUE"]=> string(0) "" } ["NAME_LONG"]=> array(36) { ["ID"]=> string(2) "45" ["TIMESTAMP_X"]=> string(19) "2023-04-13 00:55:00" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(72) "Название (для очень длинных заголовков)" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "NAME_LONG" ["DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "45" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(80) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(72) "Название (для очень длинных заголовков)" ["~DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } } } ["DISPLAY_PROPERTIES"]=> array(11) { ["AUTHOR_EN"]=> array(37) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8288" ["VALUE"]=> array(2) { ["TEXT"]=> string(88) "<sup>1</sup>Olga S. Pankratova, <sup>2</sup>Alexei B. Chukhlovin" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(64) "1Olga S. Pankratova, 2Alexei B. Chukhlovin" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(64) "1Olga S. Pankratova, 2Alexei B. Chukhlovin" } ["SUMMARY_EN"]=> array(37) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8290" ["VALUE"]=> array(2) { ["TEXT"]=> string(2944) "<p> Total depletion of innate and adaptive immune cell populations occurs after intensive chemotherapy and hematopoietic stem cell transplantation (HSCT) then followed by gradual recovery of immune populations, due to progenitors derived from donor hematopoietic cells which differentiate to myeloid and lymphoid lineages. Time dynamics of immune reconstitution and differential maturation of distinct immune populations is only partially evaluated, especially, at early terms post-transplant. E.g., innate immunity is restored within 1st month after HSCT, due to rapid reconstitution of granulocytes, monocytes, and natural killer (NK) cells. Meanwhile, functional recovery of mature NK-cell subsets and blood monocytes may continue for up to 3 months. </p> <p> Both T- and B-lymphocyte pools are restored much slower than myelomonocytic populations. The available information on their post-HSCT immune recovery is limited, since most studies are performed at later terms (>1 month post-transplant). Absolute numbers of CD8+ T cells return to control values ca. 4 months post-HSCT, however, exhibiting a skewed repertoire of memory T lymphocytes. Recovery and maturation rates of CD4+ T cells largely depend on residual thymus function, especially, in young subjects. Hence, a naïve CD4+ T cell population in pediatric patients predominates over 6 months post-HSCT. In older persons with inactive thymopoiesis the total CD4 cell counts remain low for years after HSCT. Meanwhile, antiviral cellular immunity is active since early terms post-transplant. E.g., cytotoxic CD8+ cells specific for cytomegalovirus (CMV), or Epstein- Barr virus (EBV) rapidly expand in cases of CMV or EBV activation. </p> <p> Despite recovery of absolute B-cell counts by day 30 post-HSCT, their functions, i.e., antigen-specific antibody production, are reduced for months and years after HSCT, due to slow restoration of mature immune cell populations, thus resemling normal evolution of B cell hierarchy in human organism. </p> <p> Reactivation of herpesviruses (mostly, CMV, EBV and Herpes Simplex) is a known feature of immune deficiency. Timing of maximal herpesvirus incidence (2-3 months post-HSCT) corresponds to the period of CD8+ and CD4+ T cell functional deficiency and B cell immaturity, thus reflecting their suboptimal ability to eliminate herpesvirus-affected leukocytes. Individual terms of immune recovery after allo-HSCT depend on the patients’ age, source of donor cells, acute GvHD post-HSCT etc. Vaccination response, being a potent in vivo criterion of immune recovery in post-transplant patients, is also dependent on the subjects’ age and restored B cell functions. Time dynamics of specific antibody response shows that the patients with latent CMV reactivation may later exhibit a strong humoral immune response, thus making the infection self-limiting. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2892) "Total depletion of innate and adaptive immune cell populations occurs after intensive chemotherapy and hematopoietic stem cell transplantation (HSCT) then followed by gradual recovery of immune populations, due to progenitors derived from donor hematopoietic cells which differentiate to myeloid and lymphoid lineages. Time dynamics of immune reconstitution and differential maturation of distinct immune populations is only partially evaluated, especially, at early terms post-transplant. E.g., innate immunity is restored within 1st month after HSCT, due to rapid reconstitution of granulocytes, monocytes, and natural killer (NK) cells. Meanwhile, functional recovery of mature NK-cell subsets and blood monocytes may continue for up to 3 months.
Both T- and B-lymphocyte pools are restored much slower than myelomonocytic populations. The available information on their post-HSCT immune recovery is limited, since most studies are performed at later terms (>1 month post-transplant). Absolute numbers of CD8+ T cells return to control values ca. 4 months post-HSCT, however, exhibiting a skewed repertoire of memory T lymphocytes. Recovery and maturation rates of CD4+ T cells largely depend on residual thymus function, especially, in young subjects. Hence, a naïve CD4+ T cell population in pediatric patients predominates over 6 months post-HSCT. In older persons with inactive thymopoiesis the total CD4 cell counts remain low for years after HSCT. Meanwhile, antiviral cellular immunity is active since early terms post-transplant. E.g., cytotoxic CD8+ cells specific for cytomegalovirus (CMV), or Epstein- Barr virus (EBV) rapidly expand in cases of CMV or EBV activation.
Despite recovery of absolute B-cell counts by day 30 post-HSCT, their functions, i.e., antigen-specific antibody production, are reduced for months and years after HSCT, due to slow restoration of mature immune cell populations, thus resemling normal evolution of B cell hierarchy in human organism.
Reactivation of herpesviruses (mostly, CMV, EBV and Herpes Simplex) is a known feature of immune deficiency. Timing of maximal herpesvirus incidence (2-3 months post-HSCT) corresponds to the period of CD8+ and CD4+ T cell functional deficiency and B cell immaturity, thus reflecting their suboptimal ability to eliminate herpesvirus-affected leukocytes. Individual terms of immune recovery after allo-HSCT depend on the patients’ age, source of donor cells, acute GvHD post-HSCT etc. Vaccination response, being a potent in vivo criterion of immune recovery in post-transplant patients, is also dependent on the subjects’ age and restored B cell functions. Time dynamics of specific antibody response shows that the patients with latent CMV reactivation may later exhibit a strong humoral immune response, thus making the infection self-limiting.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(2892) "Total depletion of innate and adaptive immune cell populations occurs after intensive chemotherapy and hematopoietic stem cell transplantation (HSCT) then followed by gradual recovery of immune populations, due to progenitors derived from donor hematopoietic cells which differentiate to myeloid and lymphoid lineages. Time dynamics of immune reconstitution and differential maturation of distinct immune populations is only partially evaluated, especially, at early terms post-transplant. E.g., innate immunity is restored within 1st month after HSCT, due to rapid reconstitution of granulocytes, monocytes, and natural killer (NK) cells. Meanwhile, functional recovery of mature NK-cell subsets and blood monocytes may continue for up to 3 months.
Both T- and B-lymphocyte pools are restored much slower than myelomonocytic populations. The available information on their post-HSCT immune recovery is limited, since most studies are performed at later terms (>1 month post-transplant). Absolute numbers of CD8+ T cells return to control values ca. 4 months post-HSCT, however, exhibiting a skewed repertoire of memory T lymphocytes. Recovery and maturation rates of CD4+ T cells largely depend on residual thymus function, especially, in young subjects. Hence, a naïve CD4+ T cell population in pediatric patients predominates over 6 months post-HSCT. In older persons with inactive thymopoiesis the total CD4 cell counts remain low for years after HSCT. Meanwhile, antiviral cellular immunity is active since early terms post-transplant. E.g., cytotoxic CD8+ cells specific for cytomegalovirus (CMV), or Epstein- Barr virus (EBV) rapidly expand in cases of CMV or EBV activation.
Despite recovery of absolute B-cell counts by day 30 post-HSCT, their functions, i.e., antigen-specific antibody production, are reduced for months and years after HSCT, due to slow restoration of mature immune cell populations, thus resemling normal evolution of B cell hierarchy in human organism.
Reactivation of herpesviruses (mostly, CMV, EBV and Herpes Simplex) is a known feature of immune deficiency. Timing of maximal herpesvirus incidence (2-3 months post-HSCT) corresponds to the period of CD8+ and CD4+ T cell functional deficiency and B cell immaturity, thus reflecting their suboptimal ability to eliminate herpesvirus-affected leukocytes. Individual terms of immune recovery after allo-HSCT depend on the patients’ age, source of donor cells, acute GvHD post-HSCT etc. Vaccination response, being a potent in vivo criterion of immune recovery in post-transplant patients, is also dependent on the subjects’ age and restored B cell functions. Time dynamics of specific antibody response shows that the patients with latent CMV reactivation may later exhibit a strong humoral immune response, thus making the infection self-limiting.
" } ["DOI"]=> array(37) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8287" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-32-43" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-32-43" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-32-43" } ["NAME_EN"]=> array(37) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8291" ["VALUE"]=> string(103) "Time course of immune recovery and viral reactivation following hematopoietic stem cell transplantation" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(103) "Time course of immune recovery and viral reactivation following hematopoietic stem cell transplantation" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(103) "Time course of immune recovery and viral reactivation following hematopoietic stem cell transplantation" } ["ORGANIZATION_EN"]=> array(37) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8289" ["VALUE"]=> array(2) { ["TEXT"]=> string(256) "<sup>1</sup>Tampere University Hospital, Tampere, Finland <br> <sup>2</sup>R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(226) "1Tampere University Hospital, Tampere, Finland2R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(226) "1Tampere University Hospital, Tampere, Finland
2R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University" } ["AUTHOR_RU"]=> array(37) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8284" ["VALUE"]=> array(2) { ["TEXT"]=> string(120) "Ольга С. Панкратова<sup>1</sup>, Алексей Б. Чухловин<sup>2</sup>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(96) "Ольга С. Панкратова1, Алексей Б. Чухловин2" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(96) "Ольга С. Панкратова1, Алексей Б. Чухловин2" } ["SUBMITTED"]=> array(37) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8282" ["VALUE"]=> string(22) "11/02/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/02/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "11/02/2016 12:00:00 am" } ["ACCEPTED"]=> array(37) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8283" ["VALUE"]=> string(22) "12/09/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "12/09/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "12/09/2016 12:00:00 am" } ["KEYWORDS"]=> array(38) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(6) { [0]=> string(4) "8318" [1]=> string(4) "8319" [2]=> string(4) "8320" [3]=> string(4) "8321" [4]=> string(4) "8322" [5]=> string(4) "8323" } ["VALUE"]=> array(6) { [0]=> string(2) "15" [1]=> string(3) "674" [2]=> string(3) "675" [3]=> string(3) "676" [4]=> string(3) "677" [5]=> string(3) "647" } ["DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(6) { [0]=> string(2) "15" [1]=> string(3) "674" [2]=> string(3) "675" [3]=> string(3) "676" [4]=> string(3) "677" [5]=> string(3) "647" } ["~DESCRIPTION"]=> array(6) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" [5]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(6) { [0]=> string(134) "трансплантация гемопоэтических стволовых клеток" [1]=> string(93) "восстановление иммунитета" [2]=> string(84) "различия в созревании" [3]=> string(79) "временная динамика" [4]=> string(68) "герпесвирусы" [5]=> string(66) "реактивация" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["SUMMARY_RU"]=> array(37) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8286" ["VALUE"]=> array(2) { ["TEXT"]=> string(5526) "<p> Полное истощение клеточных популяций врожденного и адаптивного иммунитета возникает после интенсивной химиотерапии и трансплантации гемопоэтических стволовых клеток (ТГСК), за чем следует постепенное восстановление иммунной системы за счет предшественников, образующихся из донорских гемопоэтических клеток, которые дифференцируются в миелоидный и лимфоидные ростки. Временная динамика восстановления иммунной системы и, в частности, отдельных клеточных популяций изучена лишь отчасти, особенно в ранние сроки после ТГСК. Например, популяции клеток врожденного иммунитета восстанавливаются в течение 1-го месяца после ТГСК, в связи с быстрым обновлением популяций гранулоцитов, моноцитов и НК-клеток. В то же время функциональное созревание субпопуляций НК-клеток и моноцитов крови может длиться до 3 мес. </p> <p> Как Т-, так и В-лимфоциты восстанавливаются гораздо медленнее, чем миеломоноцитарные популяции. Есть лишь ограниченная информация об их посттрансплантационном обновлении, так как большинство исследований проводились в поздние сроки (>1 мес. после ТГСК). Так, абсолютное число CD8+ T-клеток нормализуется примерно через 4 мес., при суженном спектре Т-лимфоцитов памяти. Темпы восстановления и созревания CD4+ T во многом зависят от остаточной функции тимуса, особенно в младших возрастах. Поэтому у младших пациентов преобладает популяция «наивных» CD4+ T cell в сроки >6 мес. после ТГСК. У лиц старшего возраста с инактивацией функции тимуса общее число CD4 клеток остается низким годами после ТГСК. В то же время антивирусный клеточный иммунитет активен, начиная с ранних сроков после трансплантации. Так, цитотоксические CD8+ клетки, специфичные в отношении ЦМВ или ВЭБ, быстро размножаются в случаях реактивации этих вирусов после ТГСК. </p> <p> Несмотря на восстановление абсолютных количеств В-клеток к 30-му дню после ТГСК, их функции (в т.ч. - антиген-специфическая продукция антител) снижены многие месяцы и годы после ТГСК из-за медленного восстановления зрелых популяций иммунных клеток, что напоминает постепенную эволюцию в иерархии В-клеток в организме человека. </p> <p> Реактивация герпесвирусов (в основном –ЦМВ, ВЭБ и ВПГ) является известным признаком иммунодефицита. Периоды максимального риска реактивации герпесвирусов (2-3 мес. после ТГСК) соответствуют периодам функционального дефицита CD8+ и CD4+ T-клеток, а также незрелости В-клеток, что отражает их недостаточную способность к устранению лейкоцитов, пораженных герпесвирусами. Индивидуальные сроки иммунного восстановления после ТГСК зависят от возраста больного, источника донорских клеток, развития острой РТПХ после трансплантации и др. Ответ на вакцинирование – важный критерии состояния иммунитета после ТГСК – также зависит от возраста больного и сохранности функций В-клеток. Временная динамика специфического антительного ответа показывает, что больные с латентной реактивацией ЦМВ могут позже проявить сильный гуморальный иммунитет, что ведет к ограничению инфекционного процесса. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(5470) "
Полное истощение клеточных популяций врожденного и адаптивного иммунитета возникает после интенсивной химиотерапии и трансплантации гемопоэтических стволовых клеток (ТГСК), за чем следует постепенное восстановление иммунной системы за счет предшественников, образующихся из донорских гемопоэтических клеток, которые дифференцируются в миелоидный и лимфоидные ростки. Временная динамика восстановления иммунной системы и, в частности, отдельных клеточных популяций изучена лишь отчасти, особенно в ранние сроки после ТГСК. Например, популяции клеток врожденного иммунитета восстанавливаются в течение 1-го месяца после ТГСК, в связи с быстрым обновлением популяций гранулоцитов, моноцитов и НК-клеток. В то же время функциональное созревание субпопуляций НК-клеток и моноцитов крови может длиться до 3 мес.
Как Т-, так и В-лимфоциты восстанавливаются гораздо медленнее, чем миеломоноцитарные популяции. Есть лишь ограниченная информация об их посттрансплантационном обновлении, так как большинство исследований проводились в поздние сроки (>1 мес. после ТГСК). Так, абсолютное число CD8+ T-клеток нормализуется примерно через 4 мес., при суженном спектре Т-лимфоцитов памяти. Темпы восстановления и созревания CD4+ T во многом зависят от остаточной функции тимуса, особенно в младших возрастах. Поэтому у младших пациентов преобладает популяция «наивных» CD4+ T cell в сроки >6 мес. после ТГСК. У лиц старшего возраста с инактивацией функции тимуса общее число CD4 клеток остается низким годами после ТГСК. В то же время антивирусный клеточный иммунитет активен, начиная с ранних сроков после трансплантации. Так, цитотоксические CD8+ клетки, специфичные в отношении ЦМВ или ВЭБ, быстро размножаются в случаях реактивации этих вирусов после ТГСК.
Несмотря на восстановление абсолютных количеств В-клеток к 30-му дню после ТГСК, их функции (в т.ч. - антиген-специфическая продукция антител) снижены многие месяцы и годы после ТГСК из-за медленного восстановления зрелых популяций иммунных клеток, что напоминает постепенную эволюцию в иерархии В-клеток в организме человека.
Реактивация герпесвирусов (в основном –ЦМВ, ВЭБ и ВПГ) является известным признаком иммунодефицита. Периоды максимального риска реактивации герпесвирусов (2-3 мес. после ТГСК) соответствуют периодам функционального дефицита CD8+ и CD4+ T-клеток, а также незрелости В-клеток, что отражает их недостаточную способность к устранению лейкоцитов, пораженных герпесвирусами. Индивидуальные сроки иммунного восстановления после ТГСК зависят от возраста больного, источника донорских клеток, развития острой РТПХ после трансплантации и др. Ответ на вакцинирование – важный критерии состояния иммунитета после ТГСК – также зависит от возраста больного и сохранности функций В-клеток. Временная динамика специфического антительного ответа показывает, что больные с латентной реактивацией ЦМВ могут позже проявить сильный гуморальный иммунитет, что ведет к ограничению инфекционного процесса.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(5470) "Полное истощение клеточных популяций врожденного и адаптивного иммунитета возникает после интенсивной химиотерапии и трансплантации гемопоэтических стволовых клеток (ТГСК), за чем следует постепенное восстановление иммунной системы за счет предшественников, образующихся из донорских гемопоэтических клеток, которые дифференцируются в миелоидный и лимфоидные ростки. Временная динамика восстановления иммунной системы и, в частности, отдельных клеточных популяций изучена лишь отчасти, особенно в ранние сроки после ТГСК. Например, популяции клеток врожденного иммунитета восстанавливаются в течение 1-го месяца после ТГСК, в связи с быстрым обновлением популяций гранулоцитов, моноцитов и НК-клеток. В то же время функциональное созревание субпопуляций НК-клеток и моноцитов крови может длиться до 3 мес.
Как Т-, так и В-лимфоциты восстанавливаются гораздо медленнее, чем миеломоноцитарные популяции. Есть лишь ограниченная информация об их посттрансплантационном обновлении, так как большинство исследований проводились в поздние сроки (>1 мес. после ТГСК). Так, абсолютное число CD8+ T-клеток нормализуется примерно через 4 мес., при суженном спектре Т-лимфоцитов памяти. Темпы восстановления и созревания CD4+ T во многом зависят от остаточной функции тимуса, особенно в младших возрастах. Поэтому у младших пациентов преобладает популяция «наивных» CD4+ T cell в сроки >6 мес. после ТГСК. У лиц старшего возраста с инактивацией функции тимуса общее число CD4 клеток остается низким годами после ТГСК. В то же время антивирусный клеточный иммунитет активен, начиная с ранних сроков после трансплантации. Так, цитотоксические CD8+ клетки, специфичные в отношении ЦМВ или ВЭБ, быстро размножаются в случаях реактивации этих вирусов после ТГСК.
Несмотря на восстановление абсолютных количеств В-клеток к 30-му дню после ТГСК, их функции (в т.ч. - антиген-специфическая продукция антител) снижены многие месяцы и годы после ТГСК из-за медленного восстановления зрелых популяций иммунных клеток, что напоминает постепенную эволюцию в иерархии В-клеток в организме человека.
Реактивация герпесвирусов (в основном –ЦМВ, ВЭБ и ВПГ) является известным признаком иммунодефицита. Периоды максимального риска реактивации герпесвирусов (2-3 мес. после ТГСК) соответствуют периодам функционального дефицита CD8+ и CD4+ T-клеток, а также незрелости В-клеток, что отражает их недостаточную способность к устранению лейкоцитов, пораженных герпесвирусами. Индивидуальные сроки иммунного восстановления после ТГСК зависят от возраста больного, источника донорских клеток, развития острой РТПХ после трансплантации и др. Ответ на вакцинирование – важный критерии состояния иммунитета после ТГСК – также зависит от возраста больного и сохранности функций В-клеток. Временная динамика специфического антительного ответа показывает, что больные с латентной реактивацией ЦМВ могут позже проявить сильный гуморальный иммунитет, что ведет к ограничению инфекционного процесса.
" } ["ORGANIZATION_RU"]=> array(37) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8285" ["VALUE"]=> array(2) { ["TEXT"]=> string(502) "<sup>1</sup>Госпиталь Университета г. Тампере, Финляндия <br> <sup>2</sup>НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский<br> государственный медицинский университет им. акад. И. П.Павлова, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(466) "1Госпиталь Университета г. Тампере, Финляндия2НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. акад. И. П.Павлова, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(466) "1Госпиталь Университета г. Тампере, Финляндия
2НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. акад. И. П.Павлова, Санкт-Петербург, Россия" } } } [4]=> array(49) { ["IBLOCK_SECTION_ID"]=> string(2) "20" ["~IBLOCK_SECTION_ID"]=> string(2) "20" ["ID"]=> string(3) "684" ["~ID"]=> string(3) "684" ["IBLOCK_ID"]=> string(1) "2" ["~IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(209) "Потенциальные клинические приложения мезенхимных стволовых клеток костного мозга в области костной регенерации" ["~NAME"]=> string(209) "Потенциальные клинические приложения мезенхимных стволовых клеток костного мозга в области костной регенерации" ["ACTIVE_FROM"]=> NULL ["~ACTIVE_FROM"]=> NULL ["TIMESTAMP_X"]=> string(22) "03/30/2017 05:02:29 pm" ["~TIMESTAMP_X"]=> string(22) "03/30/2017 05:02:29 pm" ["DETAIL_PAGE_URL"]=> string(150) "/en/archive/vypusk-5-nomer-4/klinicheskie-raboty/potentsialnye-klinicheskie-prilozheniya-mezenkhimnykh-stvolovykh-kletok-kostnogo-mozga-v-oblasti-kos/" ["~DETAIL_PAGE_URL"]=> string(150) "/en/archive/vypusk-5-nomer-4/klinicheskie-raboty/potentsialnye-klinicheskie-prilozheniya-mezenkhimnykh-stvolovykh-kletok-kostnogo-mozga-v-oblasti-kos/" ["LIST_PAGE_URL"]=> string(12) "/en/archive/" ["~LIST_PAGE_URL"]=> string(12) "/en/archive/" ["DETAIL_TEXT"]=> string(59953) "
Introduction
In vitro expanded mesenchymal stem cells (MSCs) are currently tested as a promising tool for, e.g., prophylaxis of acute graft-versus-host disease (aGvHD) and organ rejection. More recent studies are performed, concerning feasibility of MSC-based therapies in heart insufficiency and acute renal failure [108]. Despite multiple studies, the main problem is to choose optimal and standard growth supplements in order to obtain reproducible results of the differentiation experiments. Meanwhile, much more expectations are connected with their abilities of in vitro differentiation into various specialized cell types. Two main pathways are easily detected under conventional culture conditions, i.e., adipogenic and osteogenic differentiation modes. Moreover, some specific growth factors may drive MSC to differentiate into the cells of other lineages, e.g., hepatocytes, brain, or renal cells. There are, however, some doubts on reproducibility of such trans-differentiation events for a sufficient fraction of proliferating MSCs.
Most clinical trials with MSC injections are aimed for treatment of autoimmune and chronic inflammatory disorders [19]. Moreover, these cell populations are regarded as a potential source for regeneration of hematopoietic and other tissues, due to a number of biologically active factors produced by MSCs, as shown by in vitro and in vivo studies [55].
Bone marrow-derived MSCs
Bone marrow (BM) represents a reservoir of different-type stem cells and precursor cells. Along with hematopoietic stem cells, the marrow contains a cell population which was previously referred to as ‘mesenchymal stromal cells’, ‘bone marrow stromal cells’, or ‘marrow multipotent mesenchymal stem cells’ [31]. All these denominations are covered with an acronym ‘BM MSC’. These cells comprise a small fraction (0.001 to 0.01%) of the entire BM nucleated population, but they could be isolated and subject to expansion on the basis of their ability to adhere to different surfaces [18]. BM MSC are multipotent and are able to differente into precursors of osteoblasts, adipocytes and chondrocytes [74]. This heterogenous cell population takes active part in hematopoietic regulation, by secreting growth factors, cytokines and other biologically active substances, as well as by intercellular and cell/matrix interactions. BM MSCs exhibit spindle-like morphology, CD73, CD90 and CD105 expression, along with negativity for hematopoietic cell markers (CD45, CD34, CD14 etc.). Numerous studies have shown that the ex vivo expanded MSCs, by systemic or local injection, are able to migrate into damaged tissues and organs and actively participate in tissue repair processes [11;77;93,98;107]. Moreover, BM MSC possess low immunogenic potential and suppress immune response, both in vitro and in vivo [2,45,47,94].
Historically, MSCs were for the first time isolated from bone marrow. Nevertheless, in further studies, MSC with similar characteristics were obtained from other organs and tissues, including subcutaneous fat, umbilical blood, placenta etc. [39,57]. Morphology, phenotype and functional properties of MSC from other sources are largely similar to BM MSC.
MSC abilities for a multi-lineage differentiation are actively studied since their discovery by A.Ya.Friedenstein in 60’s of XX century [1]. Numerous studies have demonstrated that the ex vivo cultured MSCs are able for in vitro and in vivo differentiation to the terminally differentiatiated cells of mesenchymal lineage, e,g, osteoblasts/osteocytes,chondrocytes, adipocytes, myocytes and stromal cells that may support hematopoiesis [9,10,14,20,21,37,76].
There are some problems with in vivo fate of the in vitro expanded MSCs isolated for subsequent therapeutic use. In fact, sufficient complement-mediated MSC cytotoxicity and lysis were revealed following their short-term in vitro treatment with fresh isologous serum [63]. This cytotoxic effect was abolished by the serum pretreatment with anti-C5 monoclonal antibody (Eculizumab), or divalent ion deprivation. Therefore, one should expect sufficient loss of survival for intravenously infused MSCs, as it was shown in previous studies, thus sufficiently changing their homing pattern and biological activity. Moreover, a big part of infused MSCs is entrapped in small vessels of lungs and, therefore, does no reach hematopoietic tissues or other target regions [43]. As a result, only small MSC fraction may reach bone marrow niches while homing in lungs spleen etc. Hence, MSC persistence in the body is rather short-timed, with only small amounts residing for months.
Animal models
Most studies concerning distribution of ex vivo cultured and post-labeled human MSCs after their intravenous administration to animals (i.e., mice with immune deficiency) have shown that a vast majority of the cells was captured and entrapped in lungs within 15 to 30 min. [48]. Meanwhile, halflife time of the cells in lungs was about 24 hours. Histological examination of the lung samples demonstrated MSC-associated embolism in small arteries, along with progressing apoptosis of the most MSCs. [49]. Only a small fraction of injected cells entered blood circulation, being distributed into different organs, e.g., liver, lungs, kidneys, bone marrow et al. At 48 and 96 hours post-injection, respectively, 0.04% and 0.01% of initially applied cells were detectable in these organs and tissues. Thereby, the MSC distribution patterns did not differ from those obtained upon injection of human mammary carcinoma cells [48].
The data concerning long-term MSC persistence showed sufficient inter-study differences, probably, due to the label type chosen. In some studies, human cells were revealed only in spleen by the day 7, but not after 3 months [44]. Meanwhile, other detection techniques have shown that small MSC amounts may persist in bones, cartilages, bone marrow, muscles and spleen for several months [3].
Human studies
By now, only few studies were dedicated to distribution of MSC following intravenous injection to humans. Appropriate results confirm a similar distribution pattern, i.e., cell entrapment in pulmonary microvascular network early after infusion, and probable differences in their subsequent recirculation, which may be connected with species-specific anatomical and physiological features, disease states in the persons under study, or alternative techniques of cell detection. E.g., radioindium-labeled MSCs were infused i/v to the patients with liver cirrhosis [22]. At early terms, the cells were accumulated in lungs, however, they were displaced to liver and spleen within hours and days. Radioactivity in lungs diminished, respectively, from 33.5% to 2% in lungs, while being increased in spleen from 2% to 42%.
Long-term persistence of allogeneic MSCs was studied in patients with acute graft-versus-host disease (GvHD), by means of donor DNA detection in biopsies form different tissues [100]. In 8 of 13 patients, minimal amounts of donor DNA were detected in one or more samples obtained, mostly, from lungs, spleen, lymph nodes and small intestine, within 50 days after last infusion. Quantitative analysis has shown that the donor cell numbers in these tissues did not typically exceed 0.001%.
Safety of therapy with ex vivo cultured MSC
Ex vivo grown MSC from bone marrow and other sources have been actively tested in clinical trials, from the 1990s. At the present time, more than 400 clinical trials are registered in different databases [51]. Analysis of short- and longterm effects revealed a high-level safety of this therapeutic approach. A special meta-analysis concerned adverse effects after MSC injections performed in 1012 patients enrolled into 26 clinical studes [42]. Allogeneic HLA-compatible, or HLA-mismatched MSC were infused in 56% of the studies included. No correlations were revealed between MSC infusions and acute posttransfusion reactions, organ-specific complications, infections, development or progression of malignancies and/or lethal outcomes. A transitory fever was the only significant adverse effect that could be ascribed to MSC injections. It was documented for 30 to 40% of the cases in randomized studies, when applying both autologous and allogeneic cells.
Underlying mechanisms of MSC therapeutic effects
One may discern two main mechanisms underlying the therapeutic effects observed after MSC injection. The first mechanism implies MSC proliferation and differentiation into various cell types which replenish and/or replace functional cells lost due to certain pathological process or medical influence. The second mechanism presumes trophic and immunomodulatory effects exerted by MSC upon surrounding and remote cells and tissues, due to wide-spectrum secretion of biologically active substances as well as microvesicles and apoptosis products released into extracellular space intercellular exchange. Immediate therapeutic effects observed upon BM MSC injection are mostly mediated by soluble factors (cytokines, growth factors, low-molecular compounds) produced by the BM MSC or other cells upon their interactions with BM MSC [2; 64] and microvesicules, and due to close contacts, e.g., with hematopoietic cells [72, 12, 102].
MSCs secrete a number of soluble substances (cytokines, growth factors, low-molecular compounds) which exert direct or indirect influence upon surrounding and distant cells and tissues. The MSC-mediated endocrine and paracrine effects may be divided into trophic and immunomodulatory ones. [11]. In turn, the MSC trophic effects are underlied by their ability to prevent apoptosis of neighbor cells, induce proliferation and differentiation of endogenous precursor cells, as well as to initiate angiogenesis. At the present time, some distinct factors mediating MSC effects are partially identified (Table 1).
Immunomodulatory activity of MSC is exerted via inhibition of CD4+and CD8+ Т cell, and NK cell proliferation, decreased Ig production by plasmocytes, inhibition of dendritic cell proliferation, and stimulation of regulatory T cell proliferation. These effects are performed by a number of enzymes and secreted factors: prostaglandin E2 (PGE-2), soluble leukocyte antrigen (HLA-G5), hepatocyte growth factor (HGF), inducible NO synthase (iNO), indole-2,3-deoxygenase (IDO), transforming growth factor beta (TGF-b), leukemia-inhibiting factor (LIF), and interleukin 10 (IL-10).
Multiple experiments concerning MSC effects in acute injury models of different organs, tissues and cells have shown that MSCs are able to prevent massive apoptotic cell death. MSC suppress apoptosis initiated by hypoxia, chemical factors/ acidity, mechanical damage, and ionizing irradiation [62]. This protective effect is mediated by some key secreted molecules. e.g., vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), insulin-like growth factor (IGF-I), stanniocalcin-1, transforming growth factor-beta (TGF-b), and granulocyte-macrophage colony-stimulating growth factor (GM-CSF).
Extracellular matrix molecules, (VEGF), (IGF-I), placental growth factor (PIGF), macrophage chemotaxis factor-1 (MCP- 1), fibroblast growth factor (bFGF), and interleukin 6 (IL-6), secreted MSC are initiating angiogenesis. In turn, restoration of blood circulation in the injured tissues maya represent a fundamental factor for their successful reconstitution.
Endogenous precursor cells activated and attracted to injured sites from the surrounding tissues are playing a major role in repair processes. They migrate to the damaged areas and are the main source of the newly formed differentiated cells replacing the lost ones. Nevertheless, the efficiency of this process is often insufficient, especially in cases of severe injuries, when an external stimulation is necessary. MSC-secreted factors, e.g., stem cell factor (SCF), leukemia-inhibiting factor (LIF), M-CSF, stroma-derived factor (SDF-1) and angiopoetin-1 favor cell survival, proliferation and differentiation of tissue-specific endogenous precursor cells.
Choice of MSC donor and immunogenicity of allogeneic MSC
MSCs may be harvested from the patient, then expanded and returned in autologous manner. However, MSCs for autotransplants should be sometimes isolated from senior people, diseased individuals, and females with suppressed osteogenic potential. Therefore, some benefits from usage of allogeneic MSCs are supposed in a review by Kovach et al. [41]. E.g., they suppose allogeneic MSCs isolated from young, healthy males to be optimal grafts for boosting bone repair in these populations at risk. However, one should account for non-predictable immune reactions, since some animal studies suggest that use of allogeneic MSCs is not feasible owing to immune response of the recipients to transplanted MSCs.
Ex vivo cultured MSC possess a pronounced immunomodulatory activity, express modest amounts of MHC I molecules, and do not express MHC II [5,45]. These properties allow to suggest a low immunogenic potential of allogeneic MSC, as confirmed by some experimental studies, both in vitro and in vivo [4,78,85]. On other hand, proinflammatory cytokines induce higher MHC I levels, and MHC II antigen expression. [71]. In rodent experiments, it was demonstrated that systemic infusion of allogeneic MSC is accompanied by alloimmune response [68,109]. However, if compared with other cell types, the alloimmune response to MSC is more delayed and less pronounced, and allogeneic MSC may persist in the organism of immunocompetent recipient for a much longer time. [109].
Some cautions concerning immunosuppressive effects of allogeneic MSC transplants arise from experimental study by Prigozhina et al. [75]. Immunosuppressive properties of MSCs in vivo were tested by a well-known model of ectopic bone formation in both syngeneic and allogeneic murine recipients. MSCs from different sources were implanted with neutral bone scaffold under the kidney capsule. Bone development was observed in only the syngeneic hosts, whereas the allogeneic hosts experienced transplant rejection. This data argue for perturbed in vivo immune interactions of MSCs in allogeneic recipients.
Meanwhile clinical studies in humans have shown that allogeneic MSC could be safely injected to immunocompetent recipient without development of clinically significant alloimmune reaction [6, 26, 27]. In spite of big number of trials, there are no convincing data which presume production of donor-specific antibodies (including HLA-specific) after systemic injections of allogeneic MSC. Moreover, no sufficient differences were detectable, when comparing therapeutic efficiency of auto- and allogeneic MSC [5,26]. Hence, the issue of MSC immunogenicity and its influence upon therapeutic efficiency with allogeneic MSC remains unsolved and needs further studies.
Directed MSC migration
It was shown in several experimental animal studies that, at least, a part of MSCs arriving from lungs may migrate to the foci of injury/inflammation [7, 35, 36] and, hence, accumulate at these sites in greater concentrations than in intact tissues. A key role in the directed migration of MSC belongs to chemokines and adhesion molecules. Their induced expression is initiated in the cells involved into inflammation at the injured sites. At least, a fraction of MSCs (3-4%) is expressing the CXCR4 receptor which provides cellular chemotaxis along SDF-1 concentration gradient [106], thus playing a key role in migration of other cell types, e.g., hematopoietic and endothelial populations [44, 90]. Along with CXCR4, the MSC express receptors for other chemokines. i.e., CCR1, CCR4, CCR7, CCR10, CCR9, CXCR5 и CXCR6 [29, 101]. Directed MSC migration is performed due to the expression of some adhesion molecules on their surface, i.e., integrin beta1, and integrin alpha4. Inhibition of these molecules blocks the migration process [33, 84]. Moreover, MSC may express a number of matrix metalloproteinases, thus allowing the cells to migrate in extracellular matrix [83].
Time course of osteogenic effects in regenerating bones and MSC-derived factors
Bone injury and its repair is a multistep process which exhibits different patterns of bioactive molecules released at the damaged site, especially, in the cartilage growth plate which is the bone growth area in children. Depending on the terms post-fracture, osteogenesis strongly depends on specific growth factors released by cellular microenvironment [13]. The authors discern four stages of repair mechanisms, i.e., inflammatory, fibrogenic, osteogenic and remodeling phases. Inflammatory cytokines, e.g., TNF alpha, IL- beta exert strong regulating effects at the initial post-injury phase, whereas chemokines, PDGF and FGF2 are active at the fibrogenic stage. Specifically, transforming growth factor β1 (TGFβ1) plays a critical role in bone reconstitution due to its potent chemotactic and proliferative effect on mesenchymal stem cells, therefore promoting accumulation of bone-forming tissues at the injured site. In addition, TGFβ1 induces production of ECM components, e.g., collagen, osteopontin, and alkaline phosphatase from MSCs and osteoblasts. Bone morphogenetic proteins (BMPs) also belong to the TGF-β superfamily and are additional growth factors for mesenchymal stem cells that induce both osteogenesis and angiogenesis. For more recent data see a review by Zigdon-Gilad et al. [110]. By these mechanisms, BMPs are involved at all phases of the cartilage and bone repair.
Likewise, several angiogenesis factors are important at the osteogenic response. MSCs may release angiogenic factors [angiopoietin (Ang)-1, -2, Anglike-1, -2, -3, -4, VEGF, and fibroblast growth factor-2] that attract resident MSCs and promote local angiogenesis, a conditio sine qua non for renewal of osteoid structures [73].
Potential MSC applications in osteogenesis imperfecta
Some studies concerned engraftment of ex vivo cultured syngeneic MSC in murine model of osteogenesis imperfecta (OI), an inherited collagen synthesis disorder. Upon postnatal intravenous injection of the MSCs, only negligible numbers of osteoblasts (< 2%) of donor origin were detected in recipient mice [17, 71], or their total absence [34]. Moreover, despite minimal amounts of donor osteoblasts (ca.1%) in homozygous oim/oim mice, no signs of α2 collagen type I chains were found in the bone tissue samples [71]. These data are indicative for a potentially low efficiency of MSC infusions, as a replacement cell therapy in (OI). Worth of note, despite lacking MSC engraftment, the oim/oim mice exhibited a notable increase in linear bone growth and total body mass as compared with mice from control group. The workers presumed this effect to be determined by chondrocyte proliferation in epiphyseal plates of the tubulous bones, due to some indirect paracrine effects of the MSCs injected.
Despite low efficiency of the MSC systemic infusions to intact bones, the MSC engraftment rates may be sufficiently higher if delivered to regenerating bone after its fracture. This depends on the well-known active migration of mesenchymal precursor cells from periost and other surrounding tissues, e.g., to blood circulation and repairing bone areas [87]. Several studies have shown that a small number of intravenously injected MSC could migrate to the fracture zones, later being differentiated to chondrocytes and osteoblasts which took active part in the tissue repair and bone callus formation [25, 69]. Thereby, the CXCR4-expressing MSC represented the only directly migrating population [25]. The MSC provided a significant improvement of the fracture healing, due to increased mass of regenerating chondrogenic and bone tissues, and development of harder bone calluses, when compared with control, non-MSC-treated, mice. Along with direct differentiation to chondroblasitc and osteoblastic cells, the MSCs exerted a favorable effect upon repair, by means of local and systemic immunomodulatory and trophic effects promoting further survival and proliferation of endogenous mesenchymal precursor cells, faster transition from inflammation to the callus formation, and, hence, accelerated consolidation of the bone fragments [25, 54, 69, 80].
Hence, therapeutic effects of systemic MSC infusions in the patients with OI after corrective osteotomy may proceed via several different mechanisms:
––The MSCs may exert their systemic trophic effect which causes enhanced proliferation of chondrocytes of the epiphyseal plates, and, subsequently, increased linear bone growth in pediatric patients.
––A minor MSC subpopulation may migrate to the damaged areas, differentiate to chondrocytes and osteoblasts, and, hence, participate in bone repair processes. Moreover, the de novo produced osteoblasts of donor origin in the bone callus may represent a significant part of total osteoblastic population in regenerating bone [25], and, therefore, produce sufficient amounts of type I collagen. The latter may contribute to mechanical strength of the tubulous bones subjected to osteotomy. Consequently, it may result in decreased frequency of fractures, and prevent recurrent bone deformities.
––Both systemic and local immunomodulatory and trophic effects exerted by the migrating cells promote reduction of inflammatory phase and more rapid development of the bone callus, as well as survival and proliferation of mesenchymal precursor cells, increased mass of chondrogenic and osteogenic tissues participating in the bone repair, thus, finally, leading to eventual decrease of the bone consolidation terms following osteotomy.
Pre-clinical studies of MSCs in bone regeneration models
A comprehensive systhematic review of 20 selected preclinical studies involving large animals (dogs, sheeps, rabbits) with bone defects was published by a group from China [52]. The experimental procedures included, mainly, implants of scaffolds seeded by fat- or bone marrow-derived MSCs, or direct injections of MSC into the injury site. Observation terms varied between 12 and 36 weeks. Forest plot data analysis showed a significant beneficial effect of stem cell therapy in increasing new bone formation tested by different visualization techniques (17.8%; 95% CI, 10.54; 25.03; P< 0.001), and confirmed by available data on increased bone mineral density following MSC treatment. Some differences depended on cellularity of the transplant. Usage of matrix scaffolds seemed to be more effective than direct cell injection. Noteworthy, the effects of MSC treatment proved to be diminished after 12 weeks post-transplant, thus presuming a need for repeated cell injections at longer terms.
Preliminary clinical data
At present time, only two study groups have published data on clinical application of systemic MSC infusions for treatment of OI patients. In the study by Horwitz et al. [30], six patients with type 3 OI, after previous allogeneic bone marrow transplantation, were subjected to double MSC infusions fron the same donors at a dose of 1-5×106 cells/kg body weight. In five patients of six, an accelerated growth dynamics was observed within 4 to 6 weeks after infusions. The improvement comprised 60% to 94% (a mean of 70%) of the expected median values for healthy sex- and agematched children. As compared with 0% to 40% (a mean of 20%), that were observed during 6 месяцев preceding the infusions. Horwitz and colleagues have also launched a more extensive study with 15 patients, who received regular infusions of allo- or syngeneic MSC once every 4 months over a total of 20 months. However, the results of this study are not published so far.
Another group of investigators has published two cases of allogeneic MSC infusions to 2 patients with OI (types III and IV), who were diagnosed prenatally [24, 46]. This study had some specific features:
––Allogeneic MSC derived from fetal liver were used as a grafting material due to their higher potential for proliferation and multilineage differentiation, as compared with MSC from adults [23].
––The first infusion was performed at the intrauterine stage (into the umbilical vein), hence, the infused cells got directly to systemic circulation, avoiding the pulmonary circuiut.
––At the moment of MSC infusions, both patients had multiple bone fractures, according to ultrasound data. Probably, these aspects of the study could predetermine high level of chimerism (up to 7-15%) in osteoblastic cellular lineage as seen from the bone sample testing. However, high chimerism levels proved to be transient and further dropped down to undetectable values. In both cases, the infusions were accompanied by total healing of the fractures and successful deliveries. Repeated infusions were performed post partum, at the age of 8 years and 19 months, primarily, due to stunting growth. MSC infusions were associated with resuming growth in both patients.
We have not find any works concerning systemic MSC infusions for immediate treatment of children with OI after corrective osteotomy of femoral and/or tibial bone.
Rationale and design of the ongoing study
Despite some favorable effects of the biphosphonate-type bone resorption inhibitors (e.g., increased bone mineral density and decreased fracture incidence), there is no effective treatment aimed for restoration of linear bone growth and prevention of the bone deformities in the patients with childhood OI. Moreover, the biphosphonate therapy is poorly compatible with corrective osteotomy, since these drugs suppress bone tissue remodeling, thus causing delayed healing of the osteotomy site [61]. Hence, a clear need for novel therapeutic approaches still exists for this group of patients.
On the basis of experimental and primary clinical data, we suggest that allogeneic MSC infusion should be a safe procedure, and, moreover, a stimulation of chondrocyte proliferation could be achieved in epiphyseal plates of the long bones, due to paracrine/endocrine effects of MSC infusions, thus leading to restoration of linear bone growth in pediatric patients with moderate-to-severe clinical forms of OI. Increase of the bone mass, improved quality of osteogenic tissue and its higher mineral density could be achieved via systemic and local immunomodulatory and trophic effects of MSCs, their differentiation to chondroblastic and osteoblastic lineages, and probably, due to sufficient synthesis of normal type I collagen. These effects will cause increased strength of the bones subjected to surgical interventions. In clinical aspects, these events will lead to decreased incidence of bone fractures, accelerated consolidation and lower recurrence of bone deformities.
Allogeneic MSC are planned to be used in the present study. This is due to ability of such donor cells to produce normal type I collagen, if differentiating to osteoblastic lineage. On the other side, it is well known that occurrence of alloimmune response may be a limiting factor of cell therapy efficiency, since its long-range goal is to replace and restore cells and tissues by the donor cells. Therefore, an alloreactive immune response may prevent a long-term persistence of the donor osteoblasts and normal collagen sysnthesis. Neverthless, MSCs are known to be only weakly immunogenic, and so far, according to numerous clinical trials with allogeneic cells, there are no sufficient clinical data indicative for a pronounced alloimmune response arising after systemic MSC injection (for details see under “Allogeneic MSC immunogenicity”). Moreover, some experimental results show that, in cases of alloimmune reactions towards MSC, such response is relatively delayed, and, hence, a period from cell injection to immune rejection signs is prolonged up to 20 days [109]. This time interval is sufficient for MSC migration to the osteotomy area and their differentiation to osteoblasts actively producing normal type I collagen. Besides that, we suppose that a therapeutic action of injected MSCs is determined, mainly, by systemic and local trophic/immunomodulatory effects that are produced by the donor cells within short terms post-infusion. Hence, the time intervals necessary for the basic therapeutic actions of the MSCs (hours to days post-injection), are not interfering with their probable rejection terms, due to allo-immune mechanisms (ca.20 days following the cell infusion).
To reduce probability of MSC rejection, we are planning to use the cells from HLA-matched bone marrow donors (either related, or unrelated persons from available marrow donor registries). In case of their lack, we are suggesting to employ partially compatible, and, at least, incompatible donors. For HLA-incompatible donor/recipient pairs, the patients will be monitored for specific anti-HLA antibodies.
So far, there are no recommendations concerning choice of optimal dosage and regimens for MSC injections. In vast majority of clinical studies with MSC, the doses of 1-2x106 cells/kg weight were used. Even higher cell doses were applied in some trials (8-10x106 cells/kg), without any adverse effects [38; 59, 104]. The data about dependence of treatment efficiency on the cell dosage are rather controversial. A series of pharmacodynamic studies has shown that a dose of 1x106 cells/kg exerted a weak, but statistically significant therapeutic effect in a model of myocardial infarction in hamsters [89]. The same study yielded maximal therapeutic effects when using much higher cell doses (40x106 cells/kg). Meanwhile, some clinical results suggest that higher MSC doses are not more effective, when treating acute graft-versus-host disease [53]. Moreover, MSCs are rather large-sized cells which are prone to aggregation, thus, being able of blocking blood flow in small pulmonary vessels upon intravenous infusion. Therefore, the i/v infusions at abundant MSC concentrations may be accompanied by a risk of clinically significant embolization of the small blood vessels in lungs.
Due to proven clinical safety of i.v. MSC infusions at the dose scale of 1 to 10x106 cells/kg, a therapeutic window between the cell infusions, and time required for potential immune rejection, as well as economic considerations, our present study provides for two infusions of allogeneic MSCs per patient. The allogeneic MSCs should be applied on days +1 and +10 after a corrective osteotomy at a dose of 5x106 cells per 1 kg body weight.
Primary goal of our study is to assess safety of cryopreserved, ex vivo cultured allogeneic MSCs after intravenous infusion of the cells to the patients with osteogenesis imperfecta, evaluating acute infusion toxicity and immunogenicity (anti- HLA antibody production). Secondary goal is to study potential therapeutic effects of allogeneic MSC infusions in the patients by clinical parameters, e.g., decreased incidence of bone fractures within 2 years after MSC infusions; acceleration of linear bone growth rates, accelerated bone fragment consolidation, and increased mineral density of bone tissue, as well as laboratory markers of osteogenesis. This will be a prospective open-label single-center trial, Phase 1-2. Allogeneic MSCs will isolated from the marrow of HLA-compatible related, or unrelated donors, in order to obtain ex vivo cultured MSCs. A total of 15 patients with osteogenesis imperfecta will be subjected to infusions of cryopreserved, ex vivo cultured allogeneic MSCs 24 h after a corrective osteotomy of femoral and/or tibial bone, at two doses of 5×106 cells/kg, with an interval of 10 days. Toxicity and potential efficiency of the treatment will be evaluated. A five-year observation period after MSC infusions is scheduled.
Conclusion
Multipotent mesenchymal stem cells (MSC) represent an adherent, easily cultured cell population from different sources which is recognized by specific markers and secretes a number of growth-promoting factors and cytokines.
Bone marrow-derived MSCs may differentiate to adipogenic or osteogenic direction. The fate and viability of infused MSCs is not studied in details, like as their in vivo differentiation abilities. The injected MSCs are shown to improve tissue repair processes and modulate adverse immune reactions, such as severe GVHD post-transplant.
Therefore, a novel protocol is proposed for treatment of osteogenesis imperfecta (OI), based on MSC injections performed during corrective bone plastics in the OI patients. A small group of cases should be observed for assessment of toxicity, immunogenicity and duration of potential therapeutic effects produced by MSC infusions.
Conflict of interests
No conflict of interests is declared.
References
- Afanasyev BV, Elstner EE, Zander AR, A.J.Friedenstein, founder of the mesenchymal stem cell concept. Cell Ther Transplant 2009; 1(3):35-38.
- Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005;105:1815-22.
- Allers C, Sierralta WD, Neubauer S, Rivera F, Minguell JJ, Conget PA. Dynamic of distribution of human bone marrowderived mesenchymal stem cells after transplantation into adult unconditioned mice.Transplantation. 2004;78(4):503-508.
- Amado LC, Saliaris AP, Schuleri KH, St John M, Xie JS, Cattaneo S, Durand DJ, Fitton T, Kuang JQ, Stewart G, Lehrke S, Baumgartner WW, Martin BJ, Heldman AW, Hare JM. Cardiac repair with intramyocardial injection of allogeneic mesenchymal stem cells after myocardial infarction. Proc Natl Acad Sci USA 2005;102(32):11474-11479.
- Ankrum JA, Ong JF, Karp JM. Mesenchymal stem cells: immune evasive, not immune privileged. Nat Biotechnol. 2014;32(3):252-260.
- Ascheim DD, Gelijns AC, Goldstein D, Moye LA, Smedira N, Lee S, Klodell CT, Szady A, Parides MK, Jeffries NO, Skerrett D, Taylor DA, Rame JE, Milano C, Rogers JG, Lynch J, Dewey T, Eichhorn E, Sun B, Feldman D, Simari R, O’Gara PT, Taddei-Peters WC, Miller MA, Naka Y, Bagiella E, Rose EA, Woo YJ. Mesenchymal precursor cells as adjunctive therapy in recipients of contemporary left ventricular assist devices.Circulation. 2014;129(22):2287-2296.
- Assis AC, Carvalho JL, Jacoby BA, Ferreira RL, Castanheira P, Diniz SO, Cardoso VN, Goes AM, Ferreira AJ. Time-dependent migration of systemically delivered bone marrow mesenchymal stem cells to the infarcted heart. Cell Transplant. 2010;19(2):219–230.
- Block G, Ohkouchi S, Fung F, Frenkel J, Gregory C, Pochampally R, DiMattia G, Sullivan DE, Prockop DJ. Multipotent stromal cells are activated to reduce apoptosis in part by upregulation and secretion of stanniocalcin-1. Stem Cells 2009;27(3):670–81.
- Bruder SP, Jaiswal N, Haynesworth SE, Growth kinetics, self-renewal, and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryopreservation. J Cell Biochem 1997;64:278-294.
- Bruder SP, Kurth AA, Shea M, Hayes WC, Jaiswal N, Kadiyala S. Bone regeneration by implantation of purified, culture-expanded human mesenchymal stem cells. J Orthop Res 1998; 16:155-162.
- Caplan A, Dennis J. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006 Aug 1;98(5):1076-1084.
- Chen J, An R, Liu Z, Wang JJ, Chen SZ, Hong MM, Liu JH, Xiao MY, Chen YF. Therapeutic effects of mesenchymal stem cell-derived microvesicles on pulmonary arterial hypertension in rats. Acta Pharmacol Sin. 2014; 35(9):1121-1128.
- Chung R, Xian CJ. Mechanisms for growth plate injury repair and potential cell-based therapies for regeneration. Journal of Molecular Endocrinology 2014;53:T45–T61.
- Dennis J.E., Merriam A., Awadallah A., Yoo J.U., Johnstone B., Caplan A.I., A quadripotential mesenchymal progenitor cell isolated from the marrow of an adult mouse. J. Bone Miner. Res. 1999;14:700-709.
- Di Ianni M, Del Papa B, De Ioanni M, Moretti L, Bonifacio E, Cecchini D, Sportoletti P, Falzetti F, Tabilio A. Mesenchymal cells recruit and regulate T regulatory cells. Exp Hematol 2008; 36(3): 309–318.
- Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P, Grisanti S, Gianni AM. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 2002;99(10):3838–3843.
- Dominici M, Pritchard C, Garlits JE, Hofmann TJ, Persons DA, Horwitz EM. Hematopoietic cells and osteoblasts are derived from a common marrow progenitor after bone marrow transplantation. Proc Natl Acad Sci US A. 2004;101(32):11761-11766.
- Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006;8(4):315-7.
- Dothel G, Raschi E, Rimondini R, De Ponti F. Mesenchymal stromal cell-based therapy: Regulatory and translational aspects in gastroenterology. World J Gastroenterol 2016;22(41):9057-9068.
- Ferrari G, Cusella-De Angelis G, Coletta M, Paolucci E, Stornaiuolo A, Cossu G, Mavilio F. Muscle regeneration by bone marrow-derived myogenic progenitors. Science. 1998;279:1528-1530.
- Galmiche M.C., Koteliansky V.E., Briere J., Herve P., Charbord P. Stromal cells from human long-term marrow cultures are mesenchymal cells that differentiate following a vascular smooth muscle differentiation pathway. Blood. 1993;82:66-76.
- Gholamrezanezhad A, Mirpour S, Bagheri M, Mohamadnejad M, Alimoghaddam K, Abdolahzadeh L, Saghari M, Malekzadeh R. In vivo tracking of 111In-oxine labeled mesenchymal stem cells following infusion in patients with advanced cirrhosis.Nucl Med Biol. 2011;38(7):961-967.
- Gotherstrom C, Ringden O, Tammik C, Zetterberg E, Westgren M, Le Blanc K. Immunologic properties of human fetal mesenchymal stem cells. Am J Obstet Gynecol 2004;190(1):239.
- Götherström C, Westgren M, Shaw SW, Aström E, Biswas A, Byers PH, Mattar CN, Graham GE, Taslimi J, Ewald U, Fisk NM, Yeoh AE, Lin JL, Cheng PJ, Choolani M, Le Blanc K, Chan JK. Pre- and postnatal transplantation of fetal mesenchymal stem cells in osteogenesis imperfecta: a two-center experience. Stem Cells Transl Med. 2014;3(2):255-264.
- Granero-Moltó F, Weis JA, Miga MI, Landis B, Myers TJ, O’Rear L, Longobardi L, Jansen ED, Mortlock DP, Spagnoli A. Regenerative effects of transplanted mesenchymal stem cells in fracture healing. Stem Cells 2009;27(8):1887-1898.
- Hare JM, Fishman JE, Gerstenblith G, DiFede Velazquez DL, Zambrano JP, Suncion VY, Tracy M, Ghersin E, Johnston PV, Brinker JA, Breton E, Davis-Sproul J, Schulman IH, Byrnes J, Mendizabal AM, Lowery MH, Rouy D, Altman P, Wong Po Foo C, Ruiz P, Amador A, Da Silva J, McNiece IK, Heldman AW, George R, Lardo A. Comparison of allogeneic vs autologous bone marrow–derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: the POSEIDON randomized trial. J Am Med Ass. 2012;308(22):2369-79.
- Hare JM, Traverse JH, Henry TD, Dib N, Strumpf RK, Schulman SP, Gerstenblith G, DeMaria AN, Denktas AE, Gammon RS, Hermiller JB Jr, Reisman MA, Schaer GL, Sherman W. A randomized, double-blind, placebo-controlled, dose-escalation study of intravenous adult human mesenchymal stem cells (prochymal) after acute myocardial infarction. J Am CollCardiol. 2009;54(24):2277-2286.
- Haynesworth SE, Baber MA, Caplan AI. Cytokine expression by human marrow- derived mesenchymal progenitor cells in vitro: effects of dexamethasone and IL-1 alpha. J Cell Physiol 1996;166(3):585–592.
- Honczarenko M, Le Y, Swierkowski M, Ghiran I, Glodek AM, Silberstein LE. Human bone marrow stromal cells express a distinct set of biologically functional chemokine receptors. Stem Cells. 2006;24(4):1030–1041.
- Horwitz EM, Gordon PL, Koo WK, Marx JC, Neel MD, McNall RY, Muul L, Hofmann T. Isolated allogeneic bone marrow-derived mesenchymal cells engraft and stimulate growth in children with osteogenesis imperfecta: Implications for cell therapy of bone. Proc Natl Acad Sci USA 2002;99(13):8932-8937.
- Horwitz EM, Le Blanc K, Dominici M, Mueller I, Slaper-Cortenbach I, Marini FC, Deans RJ, Krause DS, Keating A. Clarification of the nomenclature for MSC: the International Society for Cellular Therapy position statement. Cytotherapy 2005;7:393-395.
- Hung SC, Pochampally RR, Chen SC, Hsu SC, Prockop DJ. Angiogenic effects of human multipotent stromal cell conditioned medium activate the PI3K-Akt pathway in hypoxic endothelial cells to inhibit apoptosis, increase survival and stimulate angiogenesis. Stem Cells 2007;25(9):2363– 2370.
- Ip JE, Wu Y, Huang J, Zhang L, Pratt RE, Dzau VJ. Mesenchymal stem cells use integrin β1 not CXC chemokine receptor 4 for myocardial migration and engraftment. Mol Biol of Cell 2007;18(8):2873–2882.
- Ishihara A, Ohmine K, Weisbrode SE, Bertone AL. Effect of intra-medullar and intra-venous infusions of mesenchymal stem cells on cell engraftment by in-vivo cell tracking and osteoinductivity in rabbit long bones: a pilot study. Orthop Muscular Syst 2014;3(3).
- Jackson JS, Golding JP, Chapon C, Jones WA, Bhakoo KK. Homing of stem cells to sites of inflammatory brain injury after intracerebral and intravenous administration: a longitudinal imaging study. Stem Cell Res Ther 2010;1(2):17.
- Jin SZ, Liu BR, Xu J, Gao FL, Hu ZJ, Wang XH, Pei FH, Hong Y, Hu HY, Han MZ. Ex vivo-expanded bone marrow stem cells home to the liver and ameliorate functional recovery in a mouse model of acute hepatic injury. Hepatobiliary Pancreat Dis Int 2012;11(1):66–73.
- Kadiyala S, Young RG, Thiede MA, Bruder SP. Culture expanded canine mesenchymal stem cells possess osteochondrogenic potential in vivo and in vitro. Cell Transplant 1997;6:125-134.
- Kebriaei P, Isola L, Bahceci E, Holland K, Rowley S, McGuirk J, Devetten M, Jansen J, Herzig R, Schuster M, Monroy R, Uberti J. Adult human mesenchymal stem cells added to corticosteroid therapy for the treatment of acute graft-versus-host disease. Biol Blood Marrow Transplant 2009;15(7):804-811.
- Kern S, Eichler H, Stoeve J, Klüter H, Bieback K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 2006;24:1294–1301.
- Kinnaird T, Stabile E, Burnett MS, Shou M, Lee CW, Barr S, Fuchs S, Epstein SE. Local delivery of marrow-derived stromal cells augments collateral perfusion through paracrine mechanisms. Circulation 2004;109(12):1543–1549.
- Kovach TK, Dighe AS, Lobo PI, Cui QJ. Interactions between MSCs and Immune Cells: Implications for Bone Healing. J Immunol Res 2015, Article ID 752510.
- Lalu MM, McIntyre L, Pugliese C, Fergusson D, Winston BW, Marshall JC, Granton J, Stewart DJ. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic review and meta-analysis of clinical trials. PLoSOne 2012;7(10):e47559.
- Lange C, Reimer R, Zustin J, Brunswig-Spickenheier B. Mesenchymal stromal cells protect from consequences of HSCT-transplantation preparatory irradiation: insights into possible mechanisms. Cell Ther Transplant 2016; 5(2):50-58.
- Lapidot T. Mechanism of human stem cell migration and repopulation of NOD/SCID and B2mnull NOD/SCID mice. The role of SDF-1/CXCR4 interactions. Ann N Y Acad Sci 2001;938:83-95.
- Le Blanc K, Tammik C, Rosendahl K, Zetterberg E, Ringden O. HLA expression and immunologic properties of differentiated and undifferentiated mesenchymal stem cells. Exp Hematol 2003;31(10):890-896.
- Le Blanc K, Götherström C, Ringdén O, Hassan M, Mc- Mahon R, Horwitz E, Anneren G, Axelsson O, Nunn J, Ewald U, Nordén-Lindeberg S, Jansson M, Dalton A, Aström E, Westgren M. Fetal mesenchymal stem-cell engraftment in bone after in utero transplantation in a patient with severe osteogenesis imperfecta. Transplantation 2005;79(11):1607- 1614.
- Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I, Lanino E, Sundberg B, Bernardo ME, Remberger M, Dini G, Egeler RM, Bacigalupo A, Fibbe W, Ringdén O. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 2008;371:1579-1586.
- Lee RH, Pulin AA, Seo MJ, Kota DJ, Ylostalo J, Larson BL, Semprun-Prieto L, Delafontaine P, Prockop DJ. Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell 2009;5(1):54-63.
- Lee RH, Seo MJ, Pulin AA, Gregory CA, Ylostalo J, Prockop DJ. The CD34-like protein PODXL and alpha6-integrin (CD49f) identify early progenitor MSCs with increased clonogenicity and migration to infarcted heart in mice. Blood. 2009;113:816–826.
- Li L, Zhang S, Zhang Y, Yu B, Xu Y, Guan Z. Paracrine action mediate theantifibrotic effect of transplanted mesenchymal stem cells in a rat model of global heart failure. Mol Biol Rep 2009;36(4):725–731.
- Li MD, Atkins H, Bubela T. The global landscape of stem cell clinical trials. Regenerative Medicine 2014;9(1):27-39.
- Liao Y, Zhang XL, Li L, Shen FM, Zhong MK. Stem cell therapy for bone repair: a systematic review and meta-analysis of preclinical studies with large animal models. Br J Clin Pharmacol 2014;78(4):718–726.
- Lin Y, Hogan WJ. Clinical application of mesenchymal stem cells in the treatment and prevention of graft-versushost disease. Adv Hematol 2011;2011:427863.
- Liu Y, Yang R, Shi S. Systemic infusion of mesenchymal stem cells improves cell-based bone regeneration via upregulation of regulatory T cells. Tissue Eng Part A 2014. 21 (3-4):498-509.
- Madrigal M, Rao KS, Riordan NH. A review of therapeutic effects of mesenchymal stem cell secretions and induction of secretory modification by different culture methods. J Transl Med 2014;12:260.
- Majumdar MK, Thiede MA, Mosca JD, Moorman M, Gerson SL. Phenotypic and functional comparison of cultures of marrow-derived mesenchymal stem cells(MSCs) and stromal cells. J Cell Physiol 1998; 176(1):57–66.
- Makhoul G, Chiu R, Cecere R. Placental mesenchymal stem cells: a unique source for cellular cardiomyoplasty. Ann Thorac Surg. 2013; 95(5):1827-1833.
- Martinet L, Fleury-Cappellesso S, Gadelorge M, Dietrich G, Bourin P, Fournié JJ, Poupot R. A regulatory crosstalk between Vgamma9 Vdelta2 T lymphocytes and mesenchymal stem cells. Eur J Immunol 2009; 39(3):752–762.
- Maziarz RT, Devos T, Bachier CR, Goldstein SC, Leis JF, Devine SM, Meyers G, Gajewski JL, Maertens J, Deans RJ, Van’t Hof W, Lazarus HM. Single and multiple dose MultiStem® (Multi-Potent Adult Progenitor Cell) therapy prophylaxis of acute GVHD in myeloablative allogeneic hematopoietic cell transplantation: a phase I trial. Biol Blood Marrow Transplant 2014. pii: S1083-8791(14)01469-4.
- Meisel R, Zibert A, Laryea M, Göbel U, Däubener W, Dilloo D. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood 2004;103(12):4619–4621.
- Munns CF, Rauch F, Zeitlin L, Fassier F, Glorieux FH. Delayed osteotomy but not fracture healing in pediatric osteogenesisimperfecta patients receiving pamidronate. J Bone Miner Res 2004;19(11):1779-1786.
- Murphy MB, Moncivais K, Caplan A. Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine. Exp Mol Med. 2013;45:e54.
- Muslimov AR, Mikhelashvili RT, Volchkov EV, Lepik KV, Sergeev VS, Afanasyev BV. Instant lysis of therapeutic mesenchymal stem cells after intravenous infusion: role of complement system and in vitro prevention strategies. Cell Ther Transplant 2016;5(1):68-69.
- Nauta AJ, Westerhuis G, Kruisselbrink AB, Lurvink EG, Willemze R, Fibbe WE. Donor-derived mesenchymal stem cells are immunogenic in an allogeneic host and stimulate donor graft rejection in a nonmyeloablative setting. Blood 2006;108(6):2114-2120.
- Nasef A, Mathieu N, Chapel A Frick J, François S, Mazurier C, Boutarfa A, Bouchet S, Gorin NC, Thierry D, Fouillard L. Immunosuppressive effects of mesenchymal stem cells: involvement of HLA-G. Transplantation 2007;84(2):231–237.
- Nasef A, Mazurier C, Bouchet S, François S, Chapel A, Thierry D, Gorin NC, Fouillard L. Leukemia inhibitory factor: role in human mesenchymal stem cells mediated immunosuppression. Cell Immunol 2008;253(1–2):16–22.
- Nauta AJ, Fibbe WE Immunomodulatory properties of mesenchymal stromal cells. Blood 2007;110:3499-06.
- Nauta AJ, Westerhuis G, Kruisselbrink AB, Lurvink EG, Willemze R, Fibbe WE. Donor-derived mesenchymal stem cells are immunogenic in an allogeneic host and stimulate donor graft rejection in a nonmyeloablative setting. Blood. 2006;108(6):2114-2120.
- Obermeyer TS, Yonick D, Lauing K. Stock SR, Nauer R, Strotman P, Shankar R, Gamelli R, Stover M, Callaci JJ.. Mesenchymal stem cells facilitate fracture repair in an alcohol- induced impaired healing model. J Orthop Trauma. 2012;26(12):712-718.
- Ohab JJ, Fleming S, Blesch A, Carmichael ST. A neurovascular niche for neurogenesis after stroke. J Neurosci 2006;26(50):13007–13016.
- Otsuru S, Gordon PL, Shimono K, Jethva R, Marino R, Phillips CL, Hofmann TJ, Veronesi E, Dominici M, Iwamoto M, Horwitz EM. Transplanted bone marrow mononuclear cells and MSCs impart clinical benefit to children with osteogenesis imperfecta through different mechanisms. Blood. 2012;120(9):1933-1941.
- Pascucci L, Alessandri G, Dall’Aglio C. Mercati F, Coliolo P, Bazzucchi C, Dante S, Petrini S, Curina G, Ceccarelli P. Membrane vesicles mediate pro-angiogenic activity of equine adipose-derived mesenchymal stromal cells. Vet J 2014;202(2):361-366.
- Phinney DG. Biochemical heterogeneity of mesenchymal stem cell populations: clues to their therapeutic efficacy. Cell Cycle 2007; 6:2884-2889.
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science 1999;284(5411):143-147.
- Prigozhina TB, Khitrin S, Elkin G, Eizik O, Morecki S, Slavin S. Mesenchymal stromal cells lose their immunosuppressive potential after allotransplantation. Exp Hematol 2008;36(10):1370–1376.
- Prockop DJ. Marrow stromal cells as stem cells for non-hematopoietic tissues. Science 1997;276:71-74.
- Prockop DJ, Gregory CA, Spees JL. One strategy for cell and gene therapy: harnessing the power of adult stem cells to repair tissues. Proc Natl Acad Sci USA 2003;100 (Suppl 1):11917-11923.
- Quevedo HC, Hatzistergos KE, Oskouei BN, Feigenbaum GS, Rodriguez JE, Valdes D, Pattany PM, Zambrano JP, Hu Q, McNiece I, Heldman AW, Hare JM. Allogeneic mesenchymal stem cells restore cardiac function in chronic ischemic cardiomyopathy via trilineage differentiating capacity. Proc Natl Acad Sci USA 2009;106(33):14022-14027.
- Rafei M, Hsieh J, Fortier S, Li M, Yuan S, Birman E, Forner K, Boivin MN, Doody K, Tremblay M, Annabi B, Galipeau J. Mesenchymal stromal cell-derived CCL2 suppresses plasma cell immunoglobulin production via STAT3 inactivation and PAX5 induction. Blood 2008;112(13):4991– 4998.
- Rapp AE, Bindl R, Heilmann A, Erbacher A, Müller I, Brenner RE, Ignatius A. Systemic mesenchymal stem cell administration enhances bone formation in fracture repair but not load-induced bone formation. Eur Cell Mater 2015; 29:22–34.
- Rehman J, Traktuev D, Li J, Merfeld-Clauss S, Temm- Grove CJ, Bovenkerk JE, Pell CL, Johnstone BH, Considine RV, March KL. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 2004;109(10):1292–1298.
- Repeated infusions of mesenchymal stromal cells in children with osteogenesis imperfecta (STOD3). https://clinicaltrials. gov/ct2/show/NCT01061099
- Ries С, Egea V, Karow M, Kolb H, Jochum M, Neth P. MMP-2, MT1-MMP, and TIMP-2 are essential for the invasive capacity of human mesenchymal stem cells: differential regulation by inflammatory cytokines. Blood 2007;109(9):4055–4063.
- Rüster B, Göttig S, Ludwig RJ, Bistrian R, Müller S, Seifried E, Gille J, Henschler R. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood. 2006;108(12):3938–3944.
- Ryan JM, Barry FP, Murphy JM, Mahon BP. Mesenchymal stem cells avoid allogeneic rejection. J Inflamm (Lond) 2005;2:8.
- Sato K, Ozaki K, Oh I Meguro A, Hatanaka K, Nagai T, Muroi K, Ozawa K. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood 2007;109(1):228–234.
- Schindeler A, McDonald MM, Bokko P, Little DG. Bone remodeling during fracture repair: The cellular picture. Semin Cell Dev Biol. 2008;19(5):459-466.
- Sensebé L, Fleury-Cappellesso S. Biodistribution of mesenchymal stem/stromal cells in a preclinical setting. Stem Cells Int 2013;2013:678063.
- Shabbir A, Zisa D, Suzuki G, Lee T. Heart failure therapy mediated by the trophic activities of bone marrow mesenchymal stem cells: a noninvasive therapeutic regimen. Am J Physiol Heart Circ Physiol 2009;296(6):H1888-97.
- Shen L, Gao Y, Qian J, Sun A, Ge J. A novel mechanism for endothelial progenitor cells homing: The SDF-1/ CXCR4-Rac pathway may regulate endothelial progenitor cells homing through cellular polarization. Med Hypotheses 2011;76(2):256–258.
- Sorrell JM, Baber MA, Caplan AI. Influence of adult mesenchymal stem cells onin vitro vascular formation. Tissue Eng Part A. 2009;15(7):1751–1761
- Sotiropoulou PA, Perez SA, Gritzapis AD, Baxevanis CN, Papamichail M. Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells 2006;24(1):74–85.
- Spees JL, Olson SD, Ylostalo J et al. Differentiation, cell fusion, and nuclear fusion during ex vivo repair of epithelium by human adult stem cells from bone marrow stroma. Proc Natl Acad Sci USA 2003;100:2397–2402
- Stagg J. Immune regulation by mesenchymal stem cells: two sides to the coin. Tissue Antigens 2007;69:1–9.
- Suga H, Eto H, Shigeura T, Inoue K, Aoi N, Kato H, Nishimura S, Manabe I, Gonda K, Yoshimura K. IFATS collection: Fibroblast growth factor-2-induced hepatocyte growth factor secretion by adipose-derived stromal cells inhibits postinjury fibrogenesis through a c-Jun N-terminal kinase-dependent mechanism. Stem Cells. 2009;27(1):238-249.
- Sugiyama T, Kohara H, Noda M, Nagasawa T. Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity 2006;25(6):977–988.
- Tang KC, Trzaska KA, Smirnov SV, Kotenko SV, Schwander SK, Ellner JJ, Rameshwar P. Down-regulation of MHC II in mesenchymal stem cells at high IFN-gamma can be partly explained by cytoplasmic retention of CIITA. J Immunol 2008;180(3):1826-1833.
- Terada N, Hamazaki T, Oka M, Hoki M, Mastalerz DM, Nakano Y, Meyer EM, Morel L, Petersen BE, Scott EW. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature 2002;416:542-545.
- Toegel F, Weiss K, Yang Y, Hu Z, Zhang P, Westenfelder C. Vasculotropic, paracrine actions of infused mesenchymal stem cells are important to the recovery from acute kidney injury. Am J Physiol Renal Physiol 2007;292(5):F1626–1635.
- von Bahr L, Batsis I, Moll G Hägg M, Szakos A, Sundberg B, Uzunel M, Ringden O, Le Blanc K. Analysis of tissues following mesenchymal stromal cell therapy in humans indicates limited long-term engraftment and no ectopic tissue formation. Stem Cells 2012;30(7):1575-1578.
- von Lüttichau I, Notohamiprodjo M, Wechselberger A, Peters C, Henger A, Seliger C, Djafarzadeh R, Huss R, Nelson PJ. Human adult CD34− progenitor cells functionally express the chemokine receptors CCR1, CCR4, CCR7, CXCR5, and CCR10 but not CXCR4. Stem Cells Devel 2005;14(3):329–336.
- Wang C, Chen C, Lin W. Direct intramyocardial injection of mesenchymal stem cell sheet fragments improves cardiac functions after infarction. Cardiovasc Res 2008;77(3):515-524.
- Wang LT, Ting CH, Yen ML, Liu KJ, Sytwu HK, Wu K, Yen BL. Human mesenchymal stem cells (MSCs) for treatment towards immune- and inflammation-mediated diseases: review of current clinical trials. Journal of Biomedical Science 2016;23:76. DOI 10.1186/s12929-016-0289-5.
- Wilson JG, Liu KD, Zhuo H, Caballero L, McMillan M, Fang X, Cosgrove K, Vojnik R, Calfee CS, Lee JW, Rogers AJ, Levitt J, Wiener-Kronish J, Bajwa EK, Leavitt A, McKenna D, Thompson BT, Matthay MA. Mesenchymal stem (stromal) cells for treatment of ARDS: a phase 1 clinical trial. Lancet Respir Med. 2015; 3(1): 24-32.
- WMA Declaration of Helsinki - Ethical Principles for Medical Research Involving Human Subjects. Adopted by the 18th WMA General Assembly, Helsinki, Finland, June 1964 and amended by the: 29th WMA General Assembly, Tokyo, Japan, October 1975, 35th WMA General Assembly, Venice, Italy, October 1983, 41st WMA General Assembly, Hong Kong, September 1989, 48th WMA General Assembly, Somerset West, Republic of South Africa, October 1996, 52nd WMA General Assembly, Edinburgh, Scotland, October 2000, 53rd WMA General Assembly, Washington DC, USA, October 2002 (Note of Clarification added), 55th WMA General Assembly, Tokyo, Japan, October 2004 (Note of Clarification added), 59th WMA General Assembly, Seoul, Republic of Korea, October 2008, 64th WMA General Assembly, Fortaleza, Brazil, October 2013. http://www.wma. net/en/30publications/10policies/b3/
- Wynn RF, Hart CA, Corradi-Perini C, O’Neill L, Evans CA, Wraith JE, Fairbairn LJ, Bellantuono I. A small proportion of mesenchymal stem cells strongly expresses functionally active CXCR4 receptor capable of promoting migration to bone marrow. Blood 2004;104(9):2643–2645.
- Yagi H, Soto-Gutierrez A, Parekkadan B, Kitagawa Y, Tompkins RG, Kobayashi N, Yarmush ML. Mesenchymal stem cells: mechanisms of immunomodulation and homing. Cell Transplant 2010;19:667-679.
- Zander AR, Lange C, Westenfelder C. Mesenchymal stromal cells: main factor or helper in regenerative medicine? Kidney Int Suppl 2011; 1(3):74-76.
- Zangi L, Margalit R, Reich-Zeliger S, Bachar-Lustig E, Beilhack A, Negrin R, Reisner Y. Direct imaging of immune rejection and memory induction by allogeneic mesenchymal stromal cells. Stem Cells 2009;27(11):2865-74.
- Zigdon-Giladi H, Rudich U, Geller GM, Evron A. Recent advances in bone regeneration using adult stem cells. World J Stem Cells 2015; 7(3): 630-640.
Introduction
In vitro expanded mesenchymal stem cells (MSCs) are currently tested as a promising tool for, e.g., prophylaxis of acute graft-versus-host disease (aGvHD) and organ rejection. More recent studies are performed, concerning feasibility of MSC-based therapies in heart insufficiency and acute renal failure [108]. Despite multiple studies, the main problem is to choose optimal and standard growth supplements in order to obtain reproducible results of the differentiation experiments. Meanwhile, much more expectations are connected with their abilities of in vitro differentiation into various specialized cell types. Two main pathways are easily detected under conventional culture conditions, i.e., adipogenic and osteogenic differentiation modes. Moreover, some specific growth factors may drive MSC to differentiate into the cells of other lineages, e.g., hepatocytes, brain, or renal cells. There are, however, some doubts on reproducibility of such trans-differentiation events for a sufficient fraction of proliferating MSCs.
Most clinical trials with MSC injections are aimed for treatment of autoimmune and chronic inflammatory disorders [19]. Moreover, these cell populations are regarded as a potential source for regeneration of hematopoietic and other tissues, due to a number of biologically active factors produced by MSCs, as shown by in vitro and in vivo studies [55].
Bone marrow-derived MSCs
Bone marrow (BM) represents a reservoir of different-type stem cells and precursor cells. Along with hematopoietic stem cells, the marrow contains a cell population which was previously referred to as ‘mesenchymal stromal cells’, ‘bone marrow stromal cells’, or ‘marrow multipotent mesenchymal stem cells’ [31]. All these denominations are covered with an acronym ‘BM MSC’. These cells comprise a small fraction (0.001 to 0.01%) of the entire BM nucleated population, but they could be isolated and subject to expansion on the basis of their ability to adhere to different surfaces [18]. BM MSC are multipotent and are able to differente into precursors of osteoblasts, adipocytes and chondrocytes [74]. This heterogenous cell population takes active part in hematopoietic regulation, by secreting growth factors, cytokines and other biologically active substances, as well as by intercellular and cell/matrix interactions. BM MSCs exhibit spindle-like morphology, CD73, CD90 and CD105 expression, along with negativity for hematopoietic cell markers (CD45, CD34, CD14 etc.). Numerous studies have shown that the ex vivo expanded MSCs, by systemic or local injection, are able to migrate into damaged tissues and organs and actively participate in tissue repair processes [11;77;93,98;107]. Moreover, BM MSC possess low immunogenic potential and suppress immune response, both in vitro and in vivo [2,45,47,94].
Historically, MSCs were for the first time isolated from bone marrow. Nevertheless, in further studies, MSC with similar characteristics were obtained from other organs and tissues, including subcutaneous fat, umbilical blood, placenta etc. [39,57]. Morphology, phenotype and functional properties of MSC from other sources are largely similar to BM MSC.
MSC abilities for a multi-lineage differentiation are actively studied since their discovery by A.Ya.Friedenstein in 60’s of XX century [1]. Numerous studies have demonstrated that the ex vivo cultured MSCs are able for in vitro and in vivo differentiation to the terminally differentiatiated cells of mesenchymal lineage, e,g, osteoblasts/osteocytes,chondrocytes, adipocytes, myocytes and stromal cells that may support hematopoiesis [9,10,14,20,21,37,76].
There are some problems with in vivo fate of the in vitro expanded MSCs isolated for subsequent therapeutic use. In fact, sufficient complement-mediated MSC cytotoxicity and lysis were revealed following their short-term in vitro treatment with fresh isologous serum [63]. This cytotoxic effect was abolished by the serum pretreatment with anti-C5 monoclonal antibody (Eculizumab), or divalent ion deprivation. Therefore, one should expect sufficient loss of survival for intravenously infused MSCs, as it was shown in previous studies, thus sufficiently changing their homing pattern and biological activity. Moreover, a big part of infused MSCs is entrapped in small vessels of lungs and, therefore, does no reach hematopoietic tissues or other target regions [43]. As a result, only small MSC fraction may reach bone marrow niches while homing in lungs spleen etc. Hence, MSC persistence in the body is rather short-timed, with only small amounts residing for months.
Animal models
Most studies concerning distribution of ex vivo cultured and post-labeled human MSCs after their intravenous administration to animals (i.e., mice with immune deficiency) have shown that a vast majority of the cells was captured and entrapped in lungs within 15 to 30 min. [48]. Meanwhile, halflife time of the cells in lungs was about 24 hours. Histological examination of the lung samples demonstrated MSC-associated embolism in small arteries, along with progressing apoptosis of the most MSCs. [49]. Only a small fraction of injected cells entered blood circulation, being distributed into different organs, e.g., liver, lungs, kidneys, bone marrow et al. At 48 and 96 hours post-injection, respectively, 0.04% and 0.01% of initially applied cells were detectable in these organs and tissues. Thereby, the MSC distribution patterns did not differ from those obtained upon injection of human mammary carcinoma cells [48].
The data concerning long-term MSC persistence showed sufficient inter-study differences, probably, due to the label type chosen. In some studies, human cells were revealed only in spleen by the day 7, but not after 3 months [44]. Meanwhile, other detection techniques have shown that small MSC amounts may persist in bones, cartilages, bone marrow, muscles and spleen for several months [3].
Human studies
By now, only few studies were dedicated to distribution of MSC following intravenous injection to humans. Appropriate results confirm a similar distribution pattern, i.e., cell entrapment in pulmonary microvascular network early after infusion, and probable differences in their subsequent recirculation, which may be connected with species-specific anatomical and physiological features, disease states in the persons under study, or alternative techniques of cell detection. E.g., radioindium-labeled MSCs were infused i/v to the patients with liver cirrhosis [22]. At early terms, the cells were accumulated in lungs, however, they were displaced to liver and spleen within hours and days. Radioactivity in lungs diminished, respectively, from 33.5% to 2% in lungs, while being increased in spleen from 2% to 42%.
Long-term persistence of allogeneic MSCs was studied in patients with acute graft-versus-host disease (GvHD), by means of donor DNA detection in biopsies form different tissues [100]. In 8 of 13 patients, minimal amounts of donor DNA were detected in one or more samples obtained, mostly, from lungs, spleen, lymph nodes and small intestine, within 50 days after last infusion. Quantitative analysis has shown that the donor cell numbers in these tissues did not typically exceed 0.001%.
Safety of therapy with ex vivo cultured MSC
Ex vivo grown MSC from bone marrow and other sources have been actively tested in clinical trials, from the 1990s. At the present time, more than 400 clinical trials are registered in different databases [51]. Analysis of short- and longterm effects revealed a high-level safety of this therapeutic approach. A special meta-analysis concerned adverse effects after MSC injections performed in 1012 patients enrolled into 26 clinical studes [42]. Allogeneic HLA-compatible, or HLA-mismatched MSC were infused in 56% of the studies included. No correlations were revealed between MSC infusions and acute posttransfusion reactions, organ-specific complications, infections, development or progression of malignancies and/or lethal outcomes. A transitory fever was the only significant adverse effect that could be ascribed to MSC injections. It was documented for 30 to 40% of the cases in randomized studies, when applying both autologous and allogeneic cells.
Underlying mechanisms of MSC therapeutic effects
One may discern two main mechanisms underlying the therapeutic effects observed after MSC injection. The first mechanism implies MSC proliferation and differentiation into various cell types which replenish and/or replace functional cells lost due to certain pathological process or medical influence. The second mechanism presumes trophic and immunomodulatory effects exerted by MSC upon surrounding and remote cells and tissues, due to wide-spectrum secretion of biologically active substances as well as microvesicles and apoptosis products released into extracellular space intercellular exchange. Immediate therapeutic effects observed upon BM MSC injection are mostly mediated by soluble factors (cytokines, growth factors, low-molecular compounds) produced by the BM MSC or other cells upon their interactions with BM MSC [2; 64] and microvesicules, and due to close contacts, e.g., with hematopoietic cells [72, 12, 102].
MSCs secrete a number of soluble substances (cytokines, growth factors, low-molecular compounds) which exert direct or indirect influence upon surrounding and distant cells and tissues. The MSC-mediated endocrine and paracrine effects may be divided into trophic and immunomodulatory ones. [11]. In turn, the MSC trophic effects are underlied by their ability to prevent apoptosis of neighbor cells, induce proliferation and differentiation of endogenous precursor cells, as well as to initiate angiogenesis. At the present time, some distinct factors mediating MSC effects are partially identified (Table 1).
Immunomodulatory activity of MSC is exerted via inhibition of CD4+and CD8+ Т cell, and NK cell proliferation, decreased Ig production by plasmocytes, inhibition of dendritic cell proliferation, and stimulation of regulatory T cell proliferation. These effects are performed by a number of enzymes and secreted factors: prostaglandin E2 (PGE-2), soluble leukocyte antrigen (HLA-G5), hepatocyte growth factor (HGF), inducible NO synthase (iNO), indole-2,3-deoxygenase (IDO), transforming growth factor beta (TGF-b), leukemia-inhibiting factor (LIF), and interleukin 10 (IL-10).
Multiple experiments concerning MSC effects in acute injury models of different organs, tissues and cells have shown that MSCs are able to prevent massive apoptotic cell death. MSC suppress apoptosis initiated by hypoxia, chemical factors/ acidity, mechanical damage, and ionizing irradiation [62]. This protective effect is mediated by some key secreted molecules. e.g., vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), insulin-like growth factor (IGF-I), stanniocalcin-1, transforming growth factor-beta (TGF-b), and granulocyte-macrophage colony-stimulating growth factor (GM-CSF).
Extracellular matrix molecules, (VEGF), (IGF-I), placental growth factor (PIGF), macrophage chemotaxis factor-1 (MCP- 1), fibroblast growth factor (bFGF), and interleukin 6 (IL-6), secreted MSC are initiating angiogenesis. In turn, restoration of blood circulation in the injured tissues maya represent a fundamental factor for their successful reconstitution.
Endogenous precursor cells activated and attracted to injured sites from the surrounding tissues are playing a major role in repair processes. They migrate to the damaged areas and are the main source of the newly formed differentiated cells replacing the lost ones. Nevertheless, the efficiency of this process is often insufficient, especially in cases of severe injuries, when an external stimulation is necessary. MSC-secreted factors, e.g., stem cell factor (SCF), leukemia-inhibiting factor (LIF), M-CSF, stroma-derived factor (SDF-1) and angiopoetin-1 favor cell survival, proliferation and differentiation of tissue-specific endogenous precursor cells.

Choice of MSC donor and immunogenicity of allogeneic MSC
MSCs may be harvested from the patient, then expanded and returned in autologous manner. However, MSCs for autotransplants should be sometimes isolated from senior people, diseased individuals, and females with suppressed osteogenic potential. Therefore, some benefits from usage of allogeneic MSCs are supposed in a review by Kovach et al. [41]. E.g., they suppose allogeneic MSCs isolated from young, healthy males to be optimal grafts for boosting bone repair in these populations at risk. However, one should account for non-predictable immune reactions, since some animal studies suggest that use of allogeneic MSCs is not feasible owing to immune response of the recipients to transplanted MSCs.
Ex vivo cultured MSC possess a pronounced immunomodulatory activity, express modest amounts of MHC I molecules, and do not express MHC II [5,45]. These properties allow to suggest a low immunogenic potential of allogeneic MSC, as confirmed by some experimental studies, both in vitro and in vivo [4,78,85]. On other hand, proinflammatory cytokines induce higher MHC I levels, and MHC II antigen expression. [71]. In rodent experiments, it was demonstrated that systemic infusion of allogeneic MSC is accompanied by alloimmune response [68,109]. However, if compared with other cell types, the alloimmune response to MSC is more delayed and less pronounced, and allogeneic MSC may persist in the organism of immunocompetent recipient for a much longer time. [109].
Some cautions concerning immunosuppressive effects of allogeneic MSC transplants arise from experimental study by Prigozhina et al. [75]. Immunosuppressive properties of MSCs in vivo were tested by a well-known model of ectopic bone formation in both syngeneic and allogeneic murine recipients. MSCs from different sources were implanted with neutral bone scaffold under the kidney capsule. Bone development was observed in only the syngeneic hosts, whereas the allogeneic hosts experienced transplant rejection. This data argue for perturbed in vivo immune interactions of MSCs in allogeneic recipients.
Meanwhile clinical studies in humans have shown that allogeneic MSC could be safely injected to immunocompetent recipient without development of clinically significant alloimmune reaction [6, 26, 27]. In spite of big number of trials, there are no convincing data which presume production of donor-specific antibodies (including HLA-specific) after systemic injections of allogeneic MSC. Moreover, no sufficient differences were detectable, when comparing therapeutic efficiency of auto- and allogeneic MSC [5,26]. Hence, the issue of MSC immunogenicity and its influence upon therapeutic efficiency with allogeneic MSC remains unsolved and needs further studies.
Directed MSC migration
It was shown in several experimental animal studies that, at least, a part of MSCs arriving from lungs may migrate to the foci of injury/inflammation [7, 35, 36] and, hence, accumulate at these sites in greater concentrations than in intact tissues. A key role in the directed migration of MSC belongs to chemokines and adhesion molecules. Their induced expression is initiated in the cells involved into inflammation at the injured sites. At least, a fraction of MSCs (3-4%) is expressing the CXCR4 receptor which provides cellular chemotaxis along SDF-1 concentration gradient [106], thus playing a key role in migration of other cell types, e.g., hematopoietic and endothelial populations [44, 90]. Along with CXCR4, the MSC express receptors for other chemokines. i.e., CCR1, CCR4, CCR7, CCR10, CCR9, CXCR5 и CXCR6 [29, 101]. Directed MSC migration is performed due to the expression of some adhesion molecules on their surface, i.e., integrin beta1, and integrin alpha4. Inhibition of these molecules blocks the migration process [33, 84]. Moreover, MSC may express a number of matrix metalloproteinases, thus allowing the cells to migrate in extracellular matrix [83].
Time course of osteogenic effects in regenerating bones and MSC-derived factors
Bone injury and its repair is a multistep process which exhibits different patterns of bioactive molecules released at the damaged site, especially, in the cartilage growth plate which is the bone growth area in children. Depending on the terms post-fracture, osteogenesis strongly depends on specific growth factors released by cellular microenvironment [13]. The authors discern four stages of repair mechanisms, i.e., inflammatory, fibrogenic, osteogenic and remodeling phases. Inflammatory cytokines, e.g., TNF alpha, IL- beta exert strong regulating effects at the initial post-injury phase, whereas chemokines, PDGF and FGF2 are active at the fibrogenic stage. Specifically, transforming growth factor β1 (TGFβ1) plays a critical role in bone reconstitution due to its potent chemotactic and proliferative effect on mesenchymal stem cells, therefore promoting accumulation of bone-forming tissues at the injured site. In addition, TGFβ1 induces production of ECM components, e.g., collagen, osteopontin, and alkaline phosphatase from MSCs and osteoblasts. Bone morphogenetic proteins (BMPs) also belong to the TGF-β superfamily and are additional growth factors for mesenchymal stem cells that induce both osteogenesis and angiogenesis. For more recent data see a review by Zigdon-Gilad et al. [110]. By these mechanisms, BMPs are involved at all phases of the cartilage and bone repair.
Likewise, several angiogenesis factors are important at the osteogenic response. MSCs may release angiogenic factors [angiopoietin (Ang)-1, -2, Anglike-1, -2, -3, -4, VEGF, and fibroblast growth factor-2] that attract resident MSCs and promote local angiogenesis, a conditio sine qua non for renewal of osteoid structures [73].
Potential MSC applications in osteogenesis imperfecta
Some studies concerned engraftment of ex vivo cultured syngeneic MSC in murine model of osteogenesis imperfecta (OI), an inherited collagen synthesis disorder. Upon postnatal intravenous injection of the MSCs, only negligible numbers of osteoblasts (< 2%) of donor origin were detected in recipient mice [17, 71], or their total absence [34]. Moreover, despite minimal amounts of donor osteoblasts (ca.1%) in homozygous oim/oim mice, no signs of α2 collagen type I chains were found in the bone tissue samples [71]. These data are indicative for a potentially low efficiency of MSC infusions, as a replacement cell therapy in (OI). Worth of note, despite lacking MSC engraftment, the oim/oim mice exhibited a notable increase in linear bone growth and total body mass as compared with mice from control group. The workers presumed this effect to be determined by chondrocyte proliferation in epiphyseal plates of the tubulous bones, due to some indirect paracrine effects of the MSCs injected.
Despite low efficiency of the MSC systemic infusions to intact bones, the MSC engraftment rates may be sufficiently higher if delivered to regenerating bone after its fracture. This depends on the well-known active migration of mesenchymal precursor cells from periost and other surrounding tissues, e.g., to blood circulation and repairing bone areas [87]. Several studies have shown that a small number of intravenously injected MSC could migrate to the fracture zones, later being differentiated to chondrocytes and osteoblasts which took active part in the tissue repair and bone callus formation [25, 69]. Thereby, the CXCR4-expressing MSC represented the only directly migrating population [25]. The MSC provided a significant improvement of the fracture healing, due to increased mass of regenerating chondrogenic and bone tissues, and development of harder bone calluses, when compared with control, non-MSC-treated, mice. Along with direct differentiation to chondroblasitc and osteoblastic cells, the MSCs exerted a favorable effect upon repair, by means of local and systemic immunomodulatory and trophic effects promoting further survival and proliferation of endogenous mesenchymal precursor cells, faster transition from inflammation to the callus formation, and, hence, accelerated consolidation of the bone fragments [25, 54, 69, 80].
Hence, therapeutic effects of systemic MSC infusions in the patients with OI after corrective osteotomy may proceed via several different mechanisms:
––The MSCs may exert their systemic trophic effect which causes enhanced proliferation of chondrocytes of the epiphyseal plates, and, subsequently, increased linear bone growth in pediatric patients.
––A minor MSC subpopulation may migrate to the damaged areas, differentiate to chondrocytes and osteoblasts, and, hence, participate in bone repair processes. Moreover, the de novo produced osteoblasts of donor origin in the bone callus may represent a significant part of total osteoblastic population in regenerating bone [25], and, therefore, produce sufficient amounts of type I collagen. The latter may contribute to mechanical strength of the tubulous bones subjected to osteotomy. Consequently, it may result in decreased frequency of fractures, and prevent recurrent bone deformities.
––Both systemic and local immunomodulatory and trophic effects exerted by the migrating cells promote reduction of inflammatory phase and more rapid development of the bone callus, as well as survival and proliferation of mesenchymal precursor cells, increased mass of chondrogenic and osteogenic tissues participating in the bone repair, thus, finally, leading to eventual decrease of the bone consolidation terms following osteotomy.
Pre-clinical studies of MSCs in bone regeneration models
A comprehensive systhematic review of 20 selected preclinical studies involving large animals (dogs, sheeps, rabbits) with bone defects was published by a group from China [52]. The experimental procedures included, mainly, implants of scaffolds seeded by fat- or bone marrow-derived MSCs, or direct injections of MSC into the injury site. Observation terms varied between 12 and 36 weeks. Forest plot data analysis showed a significant beneficial effect of stem cell therapy in increasing new bone formation tested by different visualization techniques (17.8%; 95% CI, 10.54; 25.03; P< 0.001), and confirmed by available data on increased bone mineral density following MSC treatment. Some differences depended on cellularity of the transplant. Usage of matrix scaffolds seemed to be more effective than direct cell injection. Noteworthy, the effects of MSC treatment proved to be diminished after 12 weeks post-transplant, thus presuming a need for repeated cell injections at longer terms.
Preliminary clinical data
At present time, only two study groups have published data on clinical application of systemic MSC infusions for treatment of OI patients. In the study by Horwitz et al. [30], six patients with type 3 OI, after previous allogeneic bone marrow transplantation, were subjected to double MSC infusions fron the same donors at a dose of 1-5×106 cells/kg body weight. In five patients of six, an accelerated growth dynamics was observed within 4 to 6 weeks after infusions. The improvement comprised 60% to 94% (a mean of 70%) of the expected median values for healthy sex- and agematched children. As compared with 0% to 40% (a mean of 20%), that were observed during 6 месяцев preceding the infusions. Horwitz and colleagues have also launched a more extensive study with 15 patients, who received regular infusions of allo- or syngeneic MSC once every 4 months over a total of 20 months. However, the results of this study are not published so far.
Another group of investigators has published two cases of allogeneic MSC infusions to 2 patients with OI (types III and IV), who were diagnosed prenatally [24, 46]. This study had some specific features:
––Allogeneic MSC derived from fetal liver were used as a grafting material due to their higher potential for proliferation and multilineage differentiation, as compared with MSC from adults [23].
––The first infusion was performed at the intrauterine stage (into the umbilical vein), hence, the infused cells got directly to systemic circulation, avoiding the pulmonary circuiut.
––At the moment of MSC infusions, both patients had multiple bone fractures, according to ultrasound data. Probably, these aspects of the study could predetermine high level of chimerism (up to 7-15%) in osteoblastic cellular lineage as seen from the bone sample testing. However, high chimerism levels proved to be transient and further dropped down to undetectable values. In both cases, the infusions were accompanied by total healing of the fractures and successful deliveries. Repeated infusions were performed post partum, at the age of 8 years and 19 months, primarily, due to stunting growth. MSC infusions were associated with resuming growth in both patients.
We have not find any works concerning systemic MSC infusions for immediate treatment of children with OI after corrective osteotomy of femoral and/or tibial bone.
Rationale and design of the ongoing study
Despite some favorable effects of the biphosphonate-type bone resorption inhibitors (e.g., increased bone mineral density and decreased fracture incidence), there is no effective treatment aimed for restoration of linear bone growth and prevention of the bone deformities in the patients with childhood OI. Moreover, the biphosphonate therapy is poorly compatible with corrective osteotomy, since these drugs suppress bone tissue remodeling, thus causing delayed healing of the osteotomy site [61]. Hence, a clear need for novel therapeutic approaches still exists for this group of patients.
On the basis of experimental and primary clinical data, we suggest that allogeneic MSC infusion should be a safe procedure, and, moreover, a stimulation of chondrocyte proliferation could be achieved in epiphyseal plates of the long bones, due to paracrine/endocrine effects of MSC infusions, thus leading to restoration of linear bone growth in pediatric patients with moderate-to-severe clinical forms of OI. Increase of the bone mass, improved quality of osteogenic tissue and its higher mineral density could be achieved via systemic and local immunomodulatory and trophic effects of MSCs, their differentiation to chondroblastic and osteoblastic lineages, and probably, due to sufficient synthesis of normal type I collagen. These effects will cause increased strength of the bones subjected to surgical interventions. In clinical aspects, these events will lead to decreased incidence of bone fractures, accelerated consolidation and lower recurrence of bone deformities.
Allogeneic MSC are planned to be used in the present study. This is due to ability of such donor cells to produce normal type I collagen, if differentiating to osteoblastic lineage. On the other side, it is well known that occurrence of alloimmune response may be a limiting factor of cell therapy efficiency, since its long-range goal is to replace and restore cells and tissues by the donor cells. Therefore, an alloreactive immune response may prevent a long-term persistence of the donor osteoblasts and normal collagen sysnthesis. Neverthless, MSCs are known to be only weakly immunogenic, and so far, according to numerous clinical trials with allogeneic cells, there are no sufficient clinical data indicative for a pronounced alloimmune response arising after systemic MSC injection (for details see under “Allogeneic MSC immunogenicity”). Moreover, some experimental results show that, in cases of alloimmune reactions towards MSC, such response is relatively delayed, and, hence, a period from cell injection to immune rejection signs is prolonged up to 20 days [109]. This time interval is sufficient for MSC migration to the osteotomy area and their differentiation to osteoblasts actively producing normal type I collagen. Besides that, we suppose that a therapeutic action of injected MSCs is determined, mainly, by systemic and local trophic/immunomodulatory effects that are produced by the donor cells within short terms post-infusion. Hence, the time intervals necessary for the basic therapeutic actions of the MSCs (hours to days post-injection), are not interfering with their probable rejection terms, due to allo-immune mechanisms (ca.20 days following the cell infusion).
To reduce probability of MSC rejection, we are planning to use the cells from HLA-matched bone marrow donors (either related, or unrelated persons from available marrow donor registries). In case of their lack, we are suggesting to employ partially compatible, and, at least, incompatible donors. For HLA-incompatible donor/recipient pairs, the patients will be monitored for specific anti-HLA antibodies.
So far, there are no recommendations concerning choice of optimal dosage and regimens for MSC injections. In vast majority of clinical studies with MSC, the doses of 1-2x106 cells/kg weight were used. Even higher cell doses were applied in some trials (8-10x106 cells/kg), without any adverse effects [38; 59, 104]. The data about dependence of treatment efficiency on the cell dosage are rather controversial. A series of pharmacodynamic studies has shown that a dose of 1x106 cells/kg exerted a weak, but statistically significant therapeutic effect in a model of myocardial infarction in hamsters [89]. The same study yielded maximal therapeutic effects when using much higher cell doses (40x106 cells/kg). Meanwhile, some clinical results suggest that higher MSC doses are not more effective, when treating acute graft-versus-host disease [53]. Moreover, MSCs are rather large-sized cells which are prone to aggregation, thus, being able of blocking blood flow in small pulmonary vessels upon intravenous infusion. Therefore, the i/v infusions at abundant MSC concentrations may be accompanied by a risk of clinically significant embolization of the small blood vessels in lungs.
Due to proven clinical safety of i.v. MSC infusions at the dose scale of 1 to 10x106 cells/kg, a therapeutic window between the cell infusions, and time required for potential immune rejection, as well as economic considerations, our present study provides for two infusions of allogeneic MSCs per patient. The allogeneic MSCs should be applied on days +1 and +10 after a corrective osteotomy at a dose of 5x106 cells per 1 kg body weight.
Primary goal of our study is to assess safety of cryopreserved, ex vivo cultured allogeneic MSCs after intravenous infusion of the cells to the patients with osteogenesis imperfecta, evaluating acute infusion toxicity and immunogenicity (anti- HLA antibody production). Secondary goal is to study potential therapeutic effects of allogeneic MSC infusions in the patients by clinical parameters, e.g., decreased incidence of bone fractures within 2 years after MSC infusions; acceleration of linear bone growth rates, accelerated bone fragment consolidation, and increased mineral density of bone tissue, as well as laboratory markers of osteogenesis. This will be a prospective open-label single-center trial, Phase 1-2. Allogeneic MSCs will isolated from the marrow of HLA-compatible related, or unrelated donors, in order to obtain ex vivo cultured MSCs. A total of 15 patients with osteogenesis imperfecta will be subjected to infusions of cryopreserved, ex vivo cultured allogeneic MSCs 24 h after a corrective osteotomy of femoral and/or tibial bone, at two doses of 5×106 cells/kg, with an interval of 10 days. Toxicity and potential efficiency of the treatment will be evaluated. A five-year observation period after MSC infusions is scheduled.
Conclusion
Multipotent mesenchymal stem cells (MSC) represent an adherent, easily cultured cell population from different sources which is recognized by specific markers and secretes a number of growth-promoting factors and cytokines.
Bone marrow-derived MSCs may differentiate to adipogenic or osteogenic direction. The fate and viability of infused MSCs is not studied in details, like as their in vivo differentiation abilities. The injected MSCs are shown to improve tissue repair processes and modulate adverse immune reactions, such as severe GVHD post-transplant.
Therefore, a novel protocol is proposed for treatment of osteogenesis imperfecta (OI), based on MSC injections performed during corrective bone plastics in the OI patients. A small group of cases should be observed for assessment of toxicity, immunogenicity and duration of potential therapeutic effects produced by MSC infusions.
Conflict of interests
No conflict of interests is declared.
References
- Afanasyev BV, Elstner EE, Zander AR, A.J.Friedenstein, founder of the mesenchymal stem cell concept. Cell Ther Transplant 2009; 1(3):35-38.
- Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 2005;105:1815-22.
- Allers C, Sierralta WD, Neubauer S, Rivera F, Minguell JJ, Conget PA. Dynamic of distribution of human bone marrowderived mesenchymal stem cells after transplantation into adult unconditioned mice.Transplantation. 2004;78(4):503-508.
- Amado LC, Saliaris AP, Schuleri KH, St John M, Xie JS, Cattaneo S, Durand DJ, Fitton T, Kuang JQ, Stewart G, Lehrke S, Baumgartner WW, Martin BJ, Heldman AW, Hare JM. Cardiac repair with intramyocardial injection of allogeneic mesenchymal stem cells after myocardial infarction. Proc Natl Acad Sci USA 2005;102(32):11474-11479.
- Ankrum JA, Ong JF, Karp JM. Mesenchymal stem cells: immune evasive, not immune privileged. Nat Biotechnol. 2014;32(3):252-260.
- Ascheim DD, Gelijns AC, Goldstein D, Moye LA, Smedira N, Lee S, Klodell CT, Szady A, Parides MK, Jeffries NO, Skerrett D, Taylor DA, Rame JE, Milano C, Rogers JG, Lynch J, Dewey T, Eichhorn E, Sun B, Feldman D, Simari R, O’Gara PT, Taddei-Peters WC, Miller MA, Naka Y, Bagiella E, Rose EA, Woo YJ. Mesenchymal precursor cells as adjunctive therapy in recipients of contemporary left ventricular assist devices.Circulation. 2014;129(22):2287-2296.
- Assis AC, Carvalho JL, Jacoby BA, Ferreira RL, Castanheira P, Diniz SO, Cardoso VN, Goes AM, Ferreira AJ. Time-dependent migration of systemically delivered bone marrow mesenchymal stem cells to the infarcted heart. Cell Transplant. 2010;19(2):219–230.
- Block G, Ohkouchi S, Fung F, Frenkel J, Gregory C, Pochampally R, DiMattia G, Sullivan DE, Prockop DJ. Multipotent stromal cells are activated to reduce apoptosis in part by upregulation and secretion of stanniocalcin-1. Stem Cells 2009;27(3):670–81.
- Bruder SP, Jaiswal N, Haynesworth SE, Growth kinetics, self-renewal, and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryopreservation. J Cell Biochem 1997;64:278-294.
- Bruder SP, Kurth AA, Shea M, Hayes WC, Jaiswal N, Kadiyala S. Bone regeneration by implantation of purified, culture-expanded human mesenchymal stem cells. J Orthop Res 1998; 16:155-162.
- Caplan A, Dennis J. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006 Aug 1;98(5):1076-1084.
- Chen J, An R, Liu Z, Wang JJ, Chen SZ, Hong MM, Liu JH, Xiao MY, Chen YF. Therapeutic effects of mesenchymal stem cell-derived microvesicles on pulmonary arterial hypertension in rats. Acta Pharmacol Sin. 2014; 35(9):1121-1128.
- Chung R, Xian CJ. Mechanisms for growth plate injury repair and potential cell-based therapies for regeneration. Journal of Molecular Endocrinology 2014;53:T45–T61.
- Dennis J.E., Merriam A., Awadallah A., Yoo J.U., Johnstone B., Caplan A.I., A quadripotential mesenchymal progenitor cell isolated from the marrow of an adult mouse. J. Bone Miner. Res. 1999;14:700-709.
- Di Ianni M, Del Papa B, De Ioanni M, Moretti L, Bonifacio E, Cecchini D, Sportoletti P, Falzetti F, Tabilio A. Mesenchymal cells recruit and regulate T regulatory cells. Exp Hematol 2008; 36(3): 309–318.
- Di Nicola M, Carlo-Stella C, Magni M, Milanesi M, Longoni PD, Matteucci P, Grisanti S, Gianni AM. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 2002;99(10):3838–3843.
- Dominici M, Pritchard C, Garlits JE, Hofmann TJ, Persons DA, Horwitz EM. Hematopoietic cells and osteoblasts are derived from a common marrow progenitor after bone marrow transplantation. Proc Natl Acad Sci US A. 2004;101(32):11761-11766.
- Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop Dj, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006;8(4):315-7.
- Dothel G, Raschi E, Rimondini R, De Ponti F. Mesenchymal stromal cell-based therapy: Regulatory and translational aspects in gastroenterology. World J Gastroenterol 2016;22(41):9057-9068.
- Ferrari G, Cusella-De Angelis G, Coletta M, Paolucci E, Stornaiuolo A, Cossu G, Mavilio F. Muscle regeneration by bone marrow-derived myogenic progenitors. Science. 1998;279:1528-1530.
- Galmiche M.C., Koteliansky V.E., Briere J., Herve P., Charbord P. Stromal cells from human long-term marrow cultures are mesenchymal cells that differentiate following a vascular smooth muscle differentiation pathway. Blood. 1993;82:66-76.
- Gholamrezanezhad A, Mirpour S, Bagheri M, Mohamadnejad M, Alimoghaddam K, Abdolahzadeh L, Saghari M, Malekzadeh R. In vivo tracking of 111In-oxine labeled mesenchymal stem cells following infusion in patients with advanced cirrhosis.Nucl Med Biol. 2011;38(7):961-967.
- Gotherstrom C, Ringden O, Tammik C, Zetterberg E, Westgren M, Le Blanc K. Immunologic properties of human fetal mesenchymal stem cells. Am J Obstet Gynecol 2004;190(1):239.
- Götherström C, Westgren M, Shaw SW, Aström E, Biswas A, Byers PH, Mattar CN, Graham GE, Taslimi J, Ewald U, Fisk NM, Yeoh AE, Lin JL, Cheng PJ, Choolani M, Le Blanc K, Chan JK. Pre- and postnatal transplantation of fetal mesenchymal stem cells in osteogenesis imperfecta: a two-center experience. Stem Cells Transl Med. 2014;3(2):255-264.
- Granero-Moltó F, Weis JA, Miga MI, Landis B, Myers TJ, O’Rear L, Longobardi L, Jansen ED, Mortlock DP, Spagnoli A. Regenerative effects of transplanted mesenchymal stem cells in fracture healing. Stem Cells 2009;27(8):1887-1898.
- Hare JM, Fishman JE, Gerstenblith G, DiFede Velazquez DL, Zambrano JP, Suncion VY, Tracy M, Ghersin E, Johnston PV, Brinker JA, Breton E, Davis-Sproul J, Schulman IH, Byrnes J, Mendizabal AM, Lowery MH, Rouy D, Altman P, Wong Po Foo C, Ruiz P, Amador A, Da Silva J, McNiece IK, Heldman AW, George R, Lardo A. Comparison of allogeneic vs autologous bone marrow–derived mesenchymal stem cells delivered by transendocardial injection in patients with ischemic cardiomyopathy: the POSEIDON randomized trial. J Am Med Ass. 2012;308(22):2369-79.
- Hare JM, Traverse JH, Henry TD, Dib N, Strumpf RK, Schulman SP, Gerstenblith G, DeMaria AN, Denktas AE, Gammon RS, Hermiller JB Jr, Reisman MA, Schaer GL, Sherman W. A randomized, double-blind, placebo-controlled, dose-escalation study of intravenous adult human mesenchymal stem cells (prochymal) after acute myocardial infarction. J Am CollCardiol. 2009;54(24):2277-2286.
- Haynesworth SE, Baber MA, Caplan AI. Cytokine expression by human marrow- derived mesenchymal progenitor cells in vitro: effects of dexamethasone and IL-1 alpha. J Cell Physiol 1996;166(3):585–592.
- Honczarenko M, Le Y, Swierkowski M, Ghiran I, Glodek AM, Silberstein LE. Human bone marrow stromal cells express a distinct set of biologically functional chemokine receptors. Stem Cells. 2006;24(4):1030–1041.
- Horwitz EM, Gordon PL, Koo WK, Marx JC, Neel MD, McNall RY, Muul L, Hofmann T. Isolated allogeneic bone marrow-derived mesenchymal cells engraft and stimulate growth in children with osteogenesis imperfecta: Implications for cell therapy of bone. Proc Natl Acad Sci USA 2002;99(13):8932-8937.
- Horwitz EM, Le Blanc K, Dominici M, Mueller I, Slaper-Cortenbach I, Marini FC, Deans RJ, Krause DS, Keating A. Clarification of the nomenclature for MSC: the International Society for Cellular Therapy position statement. Cytotherapy 2005;7:393-395.
- Hung SC, Pochampally RR, Chen SC, Hsu SC, Prockop DJ. Angiogenic effects of human multipotent stromal cell conditioned medium activate the PI3K-Akt pathway in hypoxic endothelial cells to inhibit apoptosis, increase survival and stimulate angiogenesis. Stem Cells 2007;25(9):2363– 2370.
- Ip JE, Wu Y, Huang J, Zhang L, Pratt RE, Dzau VJ. Mesenchymal stem cells use integrin β1 not CXC chemokine receptor 4 for myocardial migration and engraftment. Mol Biol of Cell 2007;18(8):2873–2882.
- Ishihara A, Ohmine K, Weisbrode SE, Bertone AL. Effect of intra-medullar and intra-venous infusions of mesenchymal stem cells on cell engraftment by in-vivo cell tracking and osteoinductivity in rabbit long bones: a pilot study. Orthop Muscular Syst 2014;3(3).
- Jackson JS, Golding JP, Chapon C, Jones WA, Bhakoo KK. Homing of stem cells to sites of inflammatory brain injury after intracerebral and intravenous administration: a longitudinal imaging study. Stem Cell Res Ther 2010;1(2):17.
- Jin SZ, Liu BR, Xu J, Gao FL, Hu ZJ, Wang XH, Pei FH, Hong Y, Hu HY, Han MZ. Ex vivo-expanded bone marrow stem cells home to the liver and ameliorate functional recovery in a mouse model of acute hepatic injury. Hepatobiliary Pancreat Dis Int 2012;11(1):66–73.
- Kadiyala S, Young RG, Thiede MA, Bruder SP. Culture expanded canine mesenchymal stem cells possess osteochondrogenic potential in vivo and in vitro. Cell Transplant 1997;6:125-134.
- Kebriaei P, Isola L, Bahceci E, Holland K, Rowley S, McGuirk J, Devetten M, Jansen J, Herzig R, Schuster M, Monroy R, Uberti J. Adult human mesenchymal stem cells added to corticosteroid therapy for the treatment of acute graft-versus-host disease. Biol Blood Marrow Transplant 2009;15(7):804-811.
- Kern S, Eichler H, Stoeve J, Klüter H, Bieback K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 2006;24:1294–1301.
- Kinnaird T, Stabile E, Burnett MS, Shou M, Lee CW, Barr S, Fuchs S, Epstein SE. Local delivery of marrow-derived stromal cells augments collateral perfusion through paracrine mechanisms. Circulation 2004;109(12):1543–1549.
- Kovach TK, Dighe AS, Lobo PI, Cui QJ. Interactions between MSCs and Immune Cells: Implications for Bone Healing. J Immunol Res 2015, Article ID 752510.
- Lalu MM, McIntyre L, Pugliese C, Fergusson D, Winston BW, Marshall JC, Granton J, Stewart DJ. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic review and meta-analysis of clinical trials. PLoSOne 2012;7(10):e47559.
- Lange C, Reimer R, Zustin J, Brunswig-Spickenheier B. Mesenchymal stromal cells protect from consequences of HSCT-transplantation preparatory irradiation: insights into possible mechanisms. Cell Ther Transplant 2016; 5(2):50-58.
- Lapidot T. Mechanism of human stem cell migration and repopulation of NOD/SCID and B2mnull NOD/SCID mice. The role of SDF-1/CXCR4 interactions. Ann N Y Acad Sci 2001;938:83-95.
- Le Blanc K, Tammik C, Rosendahl K, Zetterberg E, Ringden O. HLA expression and immunologic properties of differentiated and undifferentiated mesenchymal stem cells. Exp Hematol 2003;31(10):890-896.
- Le Blanc K, Götherström C, Ringdén O, Hassan M, Mc- Mahon R, Horwitz E, Anneren G, Axelsson O, Nunn J, Ewald U, Nordén-Lindeberg S, Jansson M, Dalton A, Aström E, Westgren M. Fetal mesenchymal stem-cell engraftment in bone after in utero transplantation in a patient with severe osteogenesis imperfecta. Transplantation 2005;79(11):1607- 1614.
- Le Blanc K, Frassoni F, Ball L, Locatelli F, Roelofs H, Lewis I, Lanino E, Sundberg B, Bernardo ME, Remberger M, Dini G, Egeler RM, Bacigalupo A, Fibbe W, Ringdén O. Mesenchymal stem cells for treatment of steroid-resistant, severe, acute graft-versus-host disease: a phase II study. Lancet 2008;371:1579-1586.
- Lee RH, Pulin AA, Seo MJ, Kota DJ, Ylostalo J, Larson BL, Semprun-Prieto L, Delafontaine P, Prockop DJ. Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell 2009;5(1):54-63.
- Lee RH, Seo MJ, Pulin AA, Gregory CA, Ylostalo J, Prockop DJ. The CD34-like protein PODXL and alpha6-integrin (CD49f) identify early progenitor MSCs with increased clonogenicity and migration to infarcted heart in mice. Blood. 2009;113:816–826.
- Li L, Zhang S, Zhang Y, Yu B, Xu Y, Guan Z. Paracrine action mediate theantifibrotic effect of transplanted mesenchymal stem cells in a rat model of global heart failure. Mol Biol Rep 2009;36(4):725–731.
- Li MD, Atkins H, Bubela T. The global landscape of stem cell clinical trials. Regenerative Medicine 2014;9(1):27-39.
- Liao Y, Zhang XL, Li L, Shen FM, Zhong MK. Stem cell therapy for bone repair: a systematic review and meta-analysis of preclinical studies with large animal models. Br J Clin Pharmacol 2014;78(4):718–726.
- Lin Y, Hogan WJ. Clinical application of mesenchymal stem cells in the treatment and prevention of graft-versushost disease. Adv Hematol 2011;2011:427863.
- Liu Y, Yang R, Shi S. Systemic infusion of mesenchymal stem cells improves cell-based bone regeneration via upregulation of regulatory T cells. Tissue Eng Part A 2014. 21 (3-4):498-509.
- Madrigal M, Rao KS, Riordan NH. A review of therapeutic effects of mesenchymal stem cell secretions and induction of secretory modification by different culture methods. J Transl Med 2014;12:260.
- Majumdar MK, Thiede MA, Mosca JD, Moorman M, Gerson SL. Phenotypic and functional comparison of cultures of marrow-derived mesenchymal stem cells(MSCs) and stromal cells. J Cell Physiol 1998; 176(1):57–66.
- Makhoul G, Chiu R, Cecere R. Placental mesenchymal stem cells: a unique source for cellular cardiomyoplasty. Ann Thorac Surg. 2013; 95(5):1827-1833.
- Martinet L, Fleury-Cappellesso S, Gadelorge M, Dietrich G, Bourin P, Fournié JJ, Poupot R. A regulatory crosstalk between Vgamma9 Vdelta2 T lymphocytes and mesenchymal stem cells. Eur J Immunol 2009; 39(3):752–762.
- Maziarz RT, Devos T, Bachier CR, Goldstein SC, Leis JF, Devine SM, Meyers G, Gajewski JL, Maertens J, Deans RJ, Van’t Hof W, Lazarus HM. Single and multiple dose MultiStem® (Multi-Potent Adult Progenitor Cell) therapy prophylaxis of acute GVHD in myeloablative allogeneic hematopoietic cell transplantation: a phase I trial. Biol Blood Marrow Transplant 2014. pii: S1083-8791(14)01469-4.
- Meisel R, Zibert A, Laryea M, Göbel U, Däubener W, Dilloo D. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase-mediated tryptophan degradation. Blood 2004;103(12):4619–4621.
- Munns CF, Rauch F, Zeitlin L, Fassier F, Glorieux FH. Delayed osteotomy but not fracture healing in pediatric osteogenesisimperfecta patients receiving pamidronate. J Bone Miner Res 2004;19(11):1779-1786.
- Murphy MB, Moncivais K, Caplan A. Mesenchymal stem cells: environmentally responsive therapeutics for regenerative medicine. Exp Mol Med. 2013;45:e54.
- Muslimov AR, Mikhelashvili RT, Volchkov EV, Lepik KV, Sergeev VS, Afanasyev BV. Instant lysis of therapeutic mesenchymal stem cells after intravenous infusion: role of complement system and in vitro prevention strategies. Cell Ther Transplant 2016;5(1):68-69.
- Nauta AJ, Westerhuis G, Kruisselbrink AB, Lurvink EG, Willemze R, Fibbe WE. Donor-derived mesenchymal stem cells are immunogenic in an allogeneic host and stimulate donor graft rejection in a nonmyeloablative setting. Blood 2006;108(6):2114-2120.
- Nasef A, Mathieu N, Chapel A Frick J, François S, Mazurier C, Boutarfa A, Bouchet S, Gorin NC, Thierry D, Fouillard L. Immunosuppressive effects of mesenchymal stem cells: involvement of HLA-G. Transplantation 2007;84(2):231–237.
- Nasef A, Mazurier C, Bouchet S, François S, Chapel A, Thierry D, Gorin NC, Fouillard L. Leukemia inhibitory factor: role in human mesenchymal stem cells mediated immunosuppression. Cell Immunol 2008;253(1–2):16–22.
- Nauta AJ, Fibbe WE Immunomodulatory properties of mesenchymal stromal cells. Blood 2007;110:3499-06.
- Nauta AJ, Westerhuis G, Kruisselbrink AB, Lurvink EG, Willemze R, Fibbe WE. Donor-derived mesenchymal stem cells are immunogenic in an allogeneic host and stimulate donor graft rejection in a nonmyeloablative setting. Blood. 2006;108(6):2114-2120.
- Obermeyer TS, Yonick D, Lauing K. Stock SR, Nauer R, Strotman P, Shankar R, Gamelli R, Stover M, Callaci JJ.. Mesenchymal stem cells facilitate fracture repair in an alcohol- induced impaired healing model. J Orthop Trauma. 2012;26(12):712-718.
- Ohab JJ, Fleming S, Blesch A, Carmichael ST. A neurovascular niche for neurogenesis after stroke. J Neurosci 2006;26(50):13007–13016.
- Otsuru S, Gordon PL, Shimono K, Jethva R, Marino R, Phillips CL, Hofmann TJ, Veronesi E, Dominici M, Iwamoto M, Horwitz EM. Transplanted bone marrow mononuclear cells and MSCs impart clinical benefit to children with osteogenesis imperfecta through different mechanisms. Blood. 2012;120(9):1933-1941.
- Pascucci L, Alessandri G, Dall’Aglio C. Mercati F, Coliolo P, Bazzucchi C, Dante S, Petrini S, Curina G, Ceccarelli P. Membrane vesicles mediate pro-angiogenic activity of equine adipose-derived mesenchymal stromal cells. Vet J 2014;202(2):361-366.
- Phinney DG. Biochemical heterogeneity of mesenchymal stem cell populations: clues to their therapeutic efficacy. Cell Cycle 2007; 6:2884-2889.
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science 1999;284(5411):143-147.
- Prigozhina TB, Khitrin S, Elkin G, Eizik O, Morecki S, Slavin S. Mesenchymal stromal cells lose their immunosuppressive potential after allotransplantation. Exp Hematol 2008;36(10):1370–1376.
- Prockop DJ. Marrow stromal cells as stem cells for non-hematopoietic tissues. Science 1997;276:71-74.
- Prockop DJ, Gregory CA, Spees JL. One strategy for cell and gene therapy: harnessing the power of adult stem cells to repair tissues. Proc Natl Acad Sci USA 2003;100 (Suppl 1):11917-11923.
- Quevedo HC, Hatzistergos KE, Oskouei BN, Feigenbaum GS, Rodriguez JE, Valdes D, Pattany PM, Zambrano JP, Hu Q, McNiece I, Heldman AW, Hare JM. Allogeneic mesenchymal stem cells restore cardiac function in chronic ischemic cardiomyopathy via trilineage differentiating capacity. Proc Natl Acad Sci USA 2009;106(33):14022-14027.
- Rafei M, Hsieh J, Fortier S, Li M, Yuan S, Birman E, Forner K, Boivin MN, Doody K, Tremblay M, Annabi B, Galipeau J. Mesenchymal stromal cell-derived CCL2 suppresses plasma cell immunoglobulin production via STAT3 inactivation and PAX5 induction. Blood 2008;112(13):4991– 4998.
- Rapp AE, Bindl R, Heilmann A, Erbacher A, Müller I, Brenner RE, Ignatius A. Systemic mesenchymal stem cell administration enhances bone formation in fracture repair but not load-induced bone formation. Eur Cell Mater 2015; 29:22–34.
- Rehman J, Traktuev D, Li J, Merfeld-Clauss S, Temm- Grove CJ, Bovenkerk JE, Pell CL, Johnstone BH, Considine RV, March KL. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation 2004;109(10):1292–1298.
- Repeated infusions of mesenchymal stromal cells in children with osteogenesis imperfecta (STOD3). https://clinicaltrials. gov/ct2/show/NCT01061099
- Ries С, Egea V, Karow M, Kolb H, Jochum M, Neth P. MMP-2, MT1-MMP, and TIMP-2 are essential for the invasive capacity of human mesenchymal stem cells: differential regulation by inflammatory cytokines. Blood 2007;109(9):4055–4063.
- Rüster B, Göttig S, Ludwig RJ, Bistrian R, Müller S, Seifried E, Gille J, Henschler R. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood. 2006;108(12):3938–3944.
- Ryan JM, Barry FP, Murphy JM, Mahon BP. Mesenchymal stem cells avoid allogeneic rejection. J Inflamm (Lond) 2005;2:8.
- Sato K, Ozaki K, Oh I Meguro A, Hatanaka K, Nagai T, Muroi K, Ozawa K. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood 2007;109(1):228–234.
- Schindeler A, McDonald MM, Bokko P, Little DG. Bone remodeling during fracture repair: The cellular picture. Semin Cell Dev Biol. 2008;19(5):459-466.
- Sensebé L, Fleury-Cappellesso S. Biodistribution of mesenchymal stem/stromal cells in a preclinical setting. Stem Cells Int 2013;2013:678063.
- Shabbir A, Zisa D, Suzuki G, Lee T. Heart failure therapy mediated by the trophic activities of bone marrow mesenchymal stem cells: a noninvasive therapeutic regimen. Am J Physiol Heart Circ Physiol 2009;296(6):H1888-97.
- Shen L, Gao Y, Qian J, Sun A, Ge J. A novel mechanism for endothelial progenitor cells homing: The SDF-1/ CXCR4-Rac pathway may regulate endothelial progenitor cells homing through cellular polarization. Med Hypotheses 2011;76(2):256–258.
- Sorrell JM, Baber MA, Caplan AI. Influence of adult mesenchymal stem cells onin vitro vascular formation. Tissue Eng Part A. 2009;15(7):1751–1761
- Sotiropoulou PA, Perez SA, Gritzapis AD, Baxevanis CN, Papamichail M. Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells 2006;24(1):74–85.
- Spees JL, Olson SD, Ylostalo J et al. Differentiation, cell fusion, and nuclear fusion during ex vivo repair of epithelium by human adult stem cells from bone marrow stroma. Proc Natl Acad Sci USA 2003;100:2397–2402
- Stagg J. Immune regulation by mesenchymal stem cells: two sides to the coin. Tissue Antigens 2007;69:1–9.
- Suga H, Eto H, Shigeura T, Inoue K, Aoi N, Kato H, Nishimura S, Manabe I, Gonda K, Yoshimura K. IFATS collection: Fibroblast growth factor-2-induced hepatocyte growth factor secretion by adipose-derived stromal cells inhibits postinjury fibrogenesis through a c-Jun N-terminal kinase-dependent mechanism. Stem Cells. 2009;27(1):238-249.
- Sugiyama T, Kohara H, Noda M, Nagasawa T. Maintenance of the hematopoietic stem cell pool by CXCL12-CXCR4 chemokine signaling in bone marrow stromal cell niches. Immunity 2006;25(6):977–988.
- Tang KC, Trzaska KA, Smirnov SV, Kotenko SV, Schwander SK, Ellner JJ, Rameshwar P. Down-regulation of MHC II in mesenchymal stem cells at high IFN-gamma can be partly explained by cytoplasmic retention of CIITA. J Immunol 2008;180(3):1826-1833.
- Terada N, Hamazaki T, Oka M, Hoki M, Mastalerz DM, Nakano Y, Meyer EM, Morel L, Petersen BE, Scott EW. Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature 2002;416:542-545.
- Toegel F, Weiss K, Yang Y, Hu Z, Zhang P, Westenfelder C. Vasculotropic, paracrine actions of infused mesenchymal stem cells are important to the recovery from acute kidney injury. Am J Physiol Renal Physiol 2007;292(5):F1626–1635.
- von Bahr L, Batsis I, Moll G Hägg M, Szakos A, Sundberg B, Uzunel M, Ringden O, Le Blanc K. Analysis of tissues following mesenchymal stromal cell therapy in humans indicates limited long-term engraftment and no ectopic tissue formation. Stem Cells 2012;30(7):1575-1578.
- von Lüttichau I, Notohamiprodjo M, Wechselberger A, Peters C, Henger A, Seliger C, Djafarzadeh R, Huss R, Nelson PJ. Human adult CD34− progenitor cells functionally express the chemokine receptors CCR1, CCR4, CCR7, CXCR5, and CCR10 but not CXCR4. Stem Cells Devel 2005;14(3):329–336.
- Wang C, Chen C, Lin W. Direct intramyocardial injection of mesenchymal stem cell sheet fragments improves cardiac functions after infarction. Cardiovasc Res 2008;77(3):515-524.
- Wang LT, Ting CH, Yen ML, Liu KJ, Sytwu HK, Wu K, Yen BL. Human mesenchymal stem cells (MSCs) for treatment towards immune- and inflammation-mediated diseases: review of current clinical trials. Journal of Biomedical Science 2016;23:76. DOI 10.1186/s12929-016-0289-5.
- Wilson JG, Liu KD, Zhuo H, Caballero L, McMillan M, Fang X, Cosgrove K, Vojnik R, Calfee CS, Lee JW, Rogers AJ, Levitt J, Wiener-Kronish J, Bajwa EK, Leavitt A, McKenna D, Thompson BT, Matthay MA. Mesenchymal stem (stromal) cells for treatment of ARDS: a phase 1 clinical trial. Lancet Respir Med. 2015; 3(1): 24-32.
- WMA Declaration of Helsinki - Ethical Principles for Medical Research Involving Human Subjects. Adopted by the 18th WMA General Assembly, Helsinki, Finland, June 1964 and amended by the: 29th WMA General Assembly, Tokyo, Japan, October 1975, 35th WMA General Assembly, Venice, Italy, October 1983, 41st WMA General Assembly, Hong Kong, September 1989, 48th WMA General Assembly, Somerset West, Republic of South Africa, October 1996, 52nd WMA General Assembly, Edinburgh, Scotland, October 2000, 53rd WMA General Assembly, Washington DC, USA, October 2002 (Note of Clarification added), 55th WMA General Assembly, Tokyo, Japan, October 2004 (Note of Clarification added), 59th WMA General Assembly, Seoul, Republic of Korea, October 2008, 64th WMA General Assembly, Fortaleza, Brazil, October 2013. http://www.wma. net/en/30publications/10policies/b3/
- Wynn RF, Hart CA, Corradi-Perini C, O’Neill L, Evans CA, Wraith JE, Fairbairn LJ, Bellantuono I. A small proportion of mesenchymal stem cells strongly expresses functionally active CXCR4 receptor capable of promoting migration to bone marrow. Blood 2004;104(9):2643–2645.
- Yagi H, Soto-Gutierrez A, Parekkadan B, Kitagawa Y, Tompkins RG, Kobayashi N, Yarmush ML. Mesenchymal stem cells: mechanisms of immunomodulation and homing. Cell Transplant 2010;19:667-679.
- Zander AR, Lange C, Westenfelder C. Mesenchymal stromal cells: main factor or helper in regenerative medicine? Kidney Int Suppl 2011; 1(3):74-76.
- Zangi L, Margalit R, Reich-Zeliger S, Bachar-Lustig E, Beilhack A, Negrin R, Reisner Y. Direct imaging of immune rejection and memory induction by allogeneic mesenchymal stromal cells. Stem Cells 2009;27(11):2865-74.
- Zigdon-Giladi H, Rudich U, Geller GM, Evron A. Recent advances in bone regeneration using adult stem cells. World J Stem Cells 2015; 7(3): 630-640.
государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_RU"]=> array(36) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8345" ["VALUE"]=> array(2) { ["TEXT"]=> string(4401) "<p> Костный мозг (КМ) содержит популяцию резидентных клеток, именуемую «мультипотентными мезенхимными стволовыми клетками» (МСК). Эти адгезивные клетки могут быть выделены и культивированы в простых питательных средах, и способны к дифференцировке в жировые или костные клетки. До сих пор не доказана возможность массовой дифференцировки МСК в гепатоциты, клетки почек или головного мозга. В то же время рассматриваются два возможных варианта их клинического применения: (1) в качестве средства иммунной модуляции при реакции «трансплантат против хозяина» (РТПХ) и аутоиммунных болезнях или (2) как потенциального источника рост-стимулирующих факторов в специализированных тканях. Эта гетерогенная популяция может поддерживать гемопоэз путем секреции факторов роста, цитокинов и других биологически активных веществ. После введения МСК способны мигрировать в поврежденные ткани, тем самым способствуя их регенерации. Однако только малая часть МСК может достичь костномозговых ниш после внутривенной инфузии. Многочисленные эксперименты с МСК на различных моделях тканевых повреждений показали их способность подавлять апоптоз, вызванный гипоксией, химическими агентами и другими разрушительными факторами. Этот защитный эффект опосредован рядом секретируемых факторов роста, как, например, ГМ-КСФ. В большом числе клинических исследований показана безопасность терапии МСК. Как клинические, так и экспериментальные исследования доказали слабую иммуногенность аллогенных МСК при их введении иммунокомпетентным реципиентам. К настоящему времени проводились инъекции культивированных МСК больным с острой РТПХ после трансплантации гемопоэтических клеток (ТГСК) и при некоторых аутоиммунных заболеваниях. </p> <p> За последнее десятилетие несколько работ кались потенциальной эффективности МСК при введении в пораженные участки костей у пациентов с несовершенным остеогенезом (ОИ) – тяжелым наследственным заболеванием с нарушением структуры коллагена, приводящим к повышенной ломкости костей. Здесь представлены сведения о клиническом протоколе для оценки безопасности, иммуногенности и клиническ их эффектов МСК, вводимых больным ОИ в ходе корригирующей остеотомии. Можно допустить, что небольшая субпопуляция МСК сможет мигрировать в поврежденные участки, дифференцироваться в хондроциты и остеобласты и, тем самым, участвовать в процессах регенерации кости. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(4377) "
Костный мозг (КМ) содержит популяцию резидентных клеток, именуемую «мультипотентными мезенхимными стволовыми клетками» (МСК). Эти адгезивные клетки могут быть выделены и культивированы в простых питательных средах, и способны к дифференцировке в жировые или костные клетки. До сих пор не доказана возможность массовой дифференцировки МСК в гепатоциты, клетки почек или головного мозга. В то же время рассматриваются два возможных варианта их клинического применения: (1) в качестве средства иммунной модуляции при реакции «трансплантат против хозяина» (РТПХ) и аутоиммунных болезнях или (2) как потенциального источника рост-стимулирующих факторов в специализированных тканях. Эта гетерогенная популяция может поддерживать гемопоэз путем секреции факторов роста, цитокинов и других биологически активных веществ. После введения МСК способны мигрировать в поврежденные ткани, тем самым способствуя их регенерации. Однако только малая часть МСК может достичь костномозговых ниш после внутривенной инфузии. Многочисленные эксперименты с МСК на различных моделях тканевых повреждений показали их способность подавлять апоптоз, вызванный гипоксией, химическими агентами и другими разрушительными факторами. Этот защитный эффект опосредован рядом секретируемых факторов роста, как, например, ГМ-КСФ. В большом числе клинических исследований показана безопасность терапии МСК. Как клинические, так и экспериментальные исследования доказали слабую иммуногенность аллогенных МСК при их введении иммунокомпетентным реципиентам. К настоящему времени проводились инъекции культивированных МСК больным с острой РТПХ после трансплантации гемопоэтических клеток (ТГСК) и при некоторых аутоиммунных заболеваниях.
За последнее десятилетие несколько работ кались потенциальной эффективности МСК при введении в пораженные участки костей у пациентов с несовершенным остеогенезом (ОИ) – тяжелым наследственным заболеванием с нарушением структуры коллагена, приводящим к повышенной ломкости костей. Здесь представлены сведения о клиническом протоколе для оценки безопасности, иммуногенности и клиническ их эффектов МСК, вводимых больным ОИ в ходе корригирующей остеотомии. Можно допустить, что небольшая субпопуляция МСК сможет мигрировать в поврежденные участки, дифференцироваться в хондроциты и остеобласты и, тем самым, участвовать в процессах регенерации кости.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["DOI"]=> array(36) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8346" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-44-55" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-44-55" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_EN"]=> array(36) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8347" ["VALUE"]=> array(2) { ["TEXT"]=> string(20) "Vladislav S. Sergeev" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(20) "Vladislav S. Sergeev" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_EN"]=> array(36) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8348" ["VALUE"]=> array(2) { ["TEXT"]=> string(503) "R. Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical<br> University, St. Petersburg, Russia<br> <br> Dr. V. S. Sergeev, St. Petersburg State I. Pavlov Medical University,<br> L. Tolstoy St. 6-8, 197022, St. Petersburg, Russia<br> <br> Phone: +7 (911) 129 7580<br> E-mail: <a href="mailto:sergeev.vlad@gmail.com">sergeev.vlad@gmail.com</a><br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(433) "R. Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov MedicalUniversity, St. Petersburg, Russia
Dr. V. S. Sergeev, St. Petersburg State I. Pavlov Medical University,
L. Tolstoy St. 6-8, 197022, St. Petersburg, Russia
Phone: +7 (911) 129 7580
E-mail: sergeev.vlad@gmail.com
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_EN"]=> array(36) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8349" ["VALUE"]=> array(2) { ["TEXT"]=> string(2348) "<p> Bone marrow (BM) contains a small resident cell population referred to as ‘multipotent mesenchymal stem cells’ (MSC). These adherent cells could be isolated and expanded in simple culture media and may differentiate in adipogenic or osteogenic pathway. So far an opportunity of MSC differentiation to hepatocytes, brain, or renal cells is not proven yet. Meanwhile, two potential clinical applications are considered for MSC: (1) as a tool for immune modulation in graft-versus-host disease (GVHD) and autoimmune diseases, or, (2) as a potential source of growth-promoting factors in specialized tissues. This heterogenous population may support hematopoiesis by secreting growth factors, cytokines and other biologically active substances. Upon injection, MSCs are able to migrate into damaged tissues, thus promoting their repair. However, only small MSC fraction may reach bone marrow niches following intravenous infusion. Multiple experiments with MSCs in different injury models show their ability to suppress apoptosis initiated by hypoxia, chemical agents/acidity and other deteriorating factors. This protective effect is mediated by a number of secreted growth factors, e.g., granulocyte-macrophage colony- stimulating growth factor (GM-CSF). A big number of clinical trials show high level of safety for the MSC therapy. Both clinical and experimental studies demonstrated only weak immunogenic effects of allogeneic MSC upon injection into immunocompetent recipients. At the present time, injections of in vitro expanded MSCs were performed in the patients developing acute GVHD after hematopoietic stem cell transplantation (HSCT), and in some autoimmune disorders. </p> <p> Over last decade, several studies concerned potentially curative effects of MSCs injected into affected bone areas in the patients with osteogenesis imperfecta (OI), a severe inherited disease with altered collagen structure resulting into increased bone fragility. Here we present a synopsis of clinical protocol aimed for assessing safety, immunogenicity, and clinical effects of MSC injected to the OI patients during corrective osteotomy. One may suggest that a minor MSC subpopulation may migrate to the damaged areas differentiating to chondrocytes and osteoblasts, and, hence, contributing to the bone repair. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2324) "
Bone marrow (BM) contains a small resident cell population referred to as ‘multipotent mesenchymal stem cells’ (MSC). These adherent cells could be isolated and expanded in simple culture media and may differentiate in adipogenic or osteogenic pathway. So far an opportunity of MSC differentiation to hepatocytes, brain, or renal cells is not proven yet. Meanwhile, two potential clinical applications are considered for MSC: (1) as a tool for immune modulation in graft-versus-host disease (GVHD) and autoimmune diseases, or, (2) as a potential source of growth-promoting factors in specialized tissues. This heterogenous population may support hematopoiesis by secreting growth factors, cytokines and other biologically active substances. Upon injection, MSCs are able to migrate into damaged tissues, thus promoting their repair. However, only small MSC fraction may reach bone marrow niches following intravenous infusion. Multiple experiments with MSCs in different injury models show their ability to suppress apoptosis initiated by hypoxia, chemical agents/acidity and other deteriorating factors. This protective effect is mediated by a number of secreted growth factors, e.g., granulocyte-macrophage colony- stimulating growth factor (GM-CSF). A big number of clinical trials show high level of safety for the MSC therapy. Both clinical and experimental studies demonstrated only weak immunogenic effects of allogeneic MSC upon injection into immunocompetent recipients. At the present time, injections of in vitro expanded MSCs were performed in the patients developing acute GVHD after hematopoietic stem cell transplantation (HSCT), and in some autoimmune disorders.
Over last decade, several studies concerned potentially curative effects of MSCs injected into affected bone areas in the patients with osteogenesis imperfecta (OI), a severe inherited disease with altered collagen structure resulting into increased bone fragility. Here we present a synopsis of clinical protocol aimed for assessing safety, immunogenicity, and clinical effects of MSC injected to the OI patients during corrective osteotomy. One may suggest that a minor MSC subpopulation may migrate to the damaged areas differentiating to chondrocytes and osteoblasts, and, hence, contributing to the bone repair.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["NAME_EN"]=> array(36) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8350" ["VALUE"]=> string(98) "Potential clinical applications of bone marrow-derived mesenchymal stem cells in bone regeneration" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(98) "Potential clinical applications of bone marrow-derived mesenchymal stem cells in bone regeneration" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" } ["FULL_TEXT_RU"]=> array(36) { ["ID"]=> string(2) "42" ["TIMESTAMP_X"]=> string(19) "2015-09-07 20:29:18" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(23) "Полный текст" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(12) "FULL_TEXT_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "42" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(23) "Полный текст" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["PDF_RU"]=> array(36) { ["ID"]=> string(2) "43" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF RUS" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_RU" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "43" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8357" ["VALUE"]=> string(3) "263" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "263" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF RUS" ["~DEFAULT_VALUE"]=> string(0) "" } ["PDF_EN"]=> array(36) { ["ID"]=> string(2) "44" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF ENG" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "44" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8358" ["VALUE"]=> string(3) "264" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "264" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF ENG" ["~DEFAULT_VALUE"]=> string(0) "" } ["NAME_LONG"]=> array(36) { ["ID"]=> string(2) "45" ["TIMESTAMP_X"]=> string(19) "2023-04-13 00:55:00" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(72) "Название (для очень длинных заголовков)" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "NAME_LONG" ["DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "45" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(80) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(72) "Название (для очень длинных заголовков)" ["~DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } } } ["DISPLAY_PROPERTIES"]=> array(13) { ["AUTHOR_EN"]=> array(37) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8347" ["VALUE"]=> array(2) { ["TEXT"]=> string(20) "Vladislav S. Sergeev" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(20) "Vladislav S. Sergeev" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(20) "Vladislav S. Sergeev" } ["SUMMARY_EN"]=> array(37) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8349" ["VALUE"]=> array(2) { ["TEXT"]=> string(2348) "<p> Bone marrow (BM) contains a small resident cell population referred to as ‘multipotent mesenchymal stem cells’ (MSC). These adherent cells could be isolated and expanded in simple culture media and may differentiate in adipogenic or osteogenic pathway. So far an opportunity of MSC differentiation to hepatocytes, brain, or renal cells is not proven yet. Meanwhile, two potential clinical applications are considered for MSC: (1) as a tool for immune modulation in graft-versus-host disease (GVHD) and autoimmune diseases, or, (2) as a potential source of growth-promoting factors in specialized tissues. This heterogenous population may support hematopoiesis by secreting growth factors, cytokines and other biologically active substances. Upon injection, MSCs are able to migrate into damaged tissues, thus promoting their repair. However, only small MSC fraction may reach bone marrow niches following intravenous infusion. Multiple experiments with MSCs in different injury models show their ability to suppress apoptosis initiated by hypoxia, chemical agents/acidity and other deteriorating factors. This protective effect is mediated by a number of secreted growth factors, e.g., granulocyte-macrophage colony- stimulating growth factor (GM-CSF). A big number of clinical trials show high level of safety for the MSC therapy. Both clinical and experimental studies demonstrated only weak immunogenic effects of allogeneic MSC upon injection into immunocompetent recipients. At the present time, injections of in vitro expanded MSCs were performed in the patients developing acute GVHD after hematopoietic stem cell transplantation (HSCT), and in some autoimmune disorders. </p> <p> Over last decade, several studies concerned potentially curative effects of MSCs injected into affected bone areas in the patients with osteogenesis imperfecta (OI), a severe inherited disease with altered collagen structure resulting into increased bone fragility. Here we present a synopsis of clinical protocol aimed for assessing safety, immunogenicity, and clinical effects of MSC injected to the OI patients during corrective osteotomy. One may suggest that a minor MSC subpopulation may migrate to the damaged areas differentiating to chondrocytes and osteoblasts, and, hence, contributing to the bone repair. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2324) "Bone marrow (BM) contains a small resident cell population referred to as ‘multipotent mesenchymal stem cells’ (MSC). These adherent cells could be isolated and expanded in simple culture media and may differentiate in adipogenic or osteogenic pathway. So far an opportunity of MSC differentiation to hepatocytes, brain, or renal cells is not proven yet. Meanwhile, two potential clinical applications are considered for MSC: (1) as a tool for immune modulation in graft-versus-host disease (GVHD) and autoimmune diseases, or, (2) as a potential source of growth-promoting factors in specialized tissues. This heterogenous population may support hematopoiesis by secreting growth factors, cytokines and other biologically active substances. Upon injection, MSCs are able to migrate into damaged tissues, thus promoting their repair. However, only small MSC fraction may reach bone marrow niches following intravenous infusion. Multiple experiments with MSCs in different injury models show their ability to suppress apoptosis initiated by hypoxia, chemical agents/acidity and other deteriorating factors. This protective effect is mediated by a number of secreted growth factors, e.g., granulocyte-macrophage colony- stimulating growth factor (GM-CSF). A big number of clinical trials show high level of safety for the MSC therapy. Both clinical and experimental studies demonstrated only weak immunogenic effects of allogeneic MSC upon injection into immunocompetent recipients. At the present time, injections of in vitro expanded MSCs were performed in the patients developing acute GVHD after hematopoietic stem cell transplantation (HSCT), and in some autoimmune disorders.
Over last decade, several studies concerned potentially curative effects of MSCs injected into affected bone areas in the patients with osteogenesis imperfecta (OI), a severe inherited disease with altered collagen structure resulting into increased bone fragility. Here we present a synopsis of clinical protocol aimed for assessing safety, immunogenicity, and clinical effects of MSC injected to the OI patients during corrective osteotomy. One may suggest that a minor MSC subpopulation may migrate to the damaged areas differentiating to chondrocytes and osteoblasts, and, hence, contributing to the bone repair.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(2324) "Bone marrow (BM) contains a small resident cell population referred to as ‘multipotent mesenchymal stem cells’ (MSC). These adherent cells could be isolated and expanded in simple culture media and may differentiate in adipogenic or osteogenic pathway. So far an opportunity of MSC differentiation to hepatocytes, brain, or renal cells is not proven yet. Meanwhile, two potential clinical applications are considered for MSC: (1) as a tool for immune modulation in graft-versus-host disease (GVHD) and autoimmune diseases, or, (2) as a potential source of growth-promoting factors in specialized tissues. This heterogenous population may support hematopoiesis by secreting growth factors, cytokines and other biologically active substances. Upon injection, MSCs are able to migrate into damaged tissues, thus promoting their repair. However, only small MSC fraction may reach bone marrow niches following intravenous infusion. Multiple experiments with MSCs in different injury models show their ability to suppress apoptosis initiated by hypoxia, chemical agents/acidity and other deteriorating factors. This protective effect is mediated by a number of secreted growth factors, e.g., granulocyte-macrophage colony- stimulating growth factor (GM-CSF). A big number of clinical trials show high level of safety for the MSC therapy. Both clinical and experimental studies demonstrated only weak immunogenic effects of allogeneic MSC upon injection into immunocompetent recipients. At the present time, injections of in vitro expanded MSCs were performed in the patients developing acute GVHD after hematopoietic stem cell transplantation (HSCT), and in some autoimmune disorders.
Over last decade, several studies concerned potentially curative effects of MSCs injected into affected bone areas in the patients with osteogenesis imperfecta (OI), a severe inherited disease with altered collagen structure resulting into increased bone fragility. Here we present a synopsis of clinical protocol aimed for assessing safety, immunogenicity, and clinical effects of MSC injected to the OI patients during corrective osteotomy. One may suggest that a minor MSC subpopulation may migrate to the damaged areas differentiating to chondrocytes and osteoblasts, and, hence, contributing to the bone repair.
" } ["DOI"]=> array(37) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8346" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-44-55" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-44-55" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-44-55" } ["NAME_EN"]=> array(37) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8350" ["VALUE"]=> string(98) "Potential clinical applications of bone marrow-derived mesenchymal stem cells in bone regeneration" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(98) "Potential clinical applications of bone marrow-derived mesenchymal stem cells in bone regeneration" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(98) "Potential clinical applications of bone marrow-derived mesenchymal stem cells in bone regeneration" } ["ORGANIZATION_EN"]=> array(37) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8348" ["VALUE"]=> array(2) { ["TEXT"]=> string(503) "R. Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical<br> University, St. Petersburg, Russia<br> <br> Dr. V. S. Sergeev, St. Petersburg State I. Pavlov Medical University,<br> L. Tolstoy St. 6-8, 197022, St. Petersburg, Russia<br> <br> Phone: +7 (911) 129 7580<br> E-mail: <a href="mailto:sergeev.vlad@gmail.com">sergeev.vlad@gmail.com</a><br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(433) "R. Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov MedicalUniversity, St. Petersburg, Russia
Dr. V. S. Sergeev, St. Petersburg State I. Pavlov Medical University,
L. Tolstoy St. 6-8, 197022, St. Petersburg, Russia
Phone: +7 (911) 129 7580
E-mail: sergeev.vlad@gmail.com
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(433) "R. Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical
University, St. Petersburg, Russia
Dr. V. S. Sergeev, St. Petersburg State I. Pavlov Medical University,
L. Tolstoy St. 6-8, 197022, St. Petersburg, Russia
Phone: +7 (911) 129 7580
E-mail: sergeev.vlad@gmail.com
" } ["AUTHORS"]=> array(38) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(1) { [0]=> string(4) "8370" } ["VALUE"]=> array(1) { [0]=> string(3) "679" } ["DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(1) { [0]=> string(3) "679" } ["~DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(63) "Vladislav S. Sergeev" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["AUTHOR_RU"]=> array(37) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8343" ["VALUE"]=> array(2) { ["TEXT"]=> string(37) "Владислав С. Сергеев" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(37) "Владислав С. Сергеев" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(37) "Владислав С. Сергеев" } ["SUBMITTED"]=> array(37) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8339" ["VALUE"]=> string(22) "10/18/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "10/18/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "10/18/2016 12:00:00 am" } ["ACCEPTED"]=> array(37) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8340" ["VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "11/25/2016 12:00:00 am" } ["KEYWORDS"]=> array(38) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(5) { [0]=> string(4) "8365" [1]=> string(4) "8366" [2]=> string(4) "8367" [3]=> string(4) "8368" [4]=> string(4) "8369" } ["VALUE"]=> array(5) { [0]=> string(2) "83" [1]=> string(3) "680" [2]=> string(3) "681" [3]=> string(3) "682" [4]=> string(3) "683" } ["DESCRIPTION"]=> array(5) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(5) { [0]=> string(2) "83" [1]=> string(3) "680" [2]=> string(3) "681" [3]=> string(3) "682" [4]=> string(3) "683" } ["~DESCRIPTION"]=> array(5) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(5) { [0]=> string(97) "мезенхимные стволовые клетки" [1]=> string(85) "секретируемые факторы" [2]=> string(86) "размножение в культуре" [3]=> string(87) "клиническое применение" [4]=> string(91) "несовершенный остеогенез" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["CONTACT"]=> array(38) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8341" ["VALUE"]=> string(3) "679" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "679" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(63) "Vladislav S. Sergeev" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["SUMMARY_RU"]=> array(37) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8345" ["VALUE"]=> array(2) { ["TEXT"]=> string(4401) "<p> Костный мозг (КМ) содержит популяцию резидентных клеток, именуемую «мультипотентными мезенхимными стволовыми клетками» (МСК). Эти адгезивные клетки могут быть выделены и культивированы в простых питательных средах, и способны к дифференцировке в жировые или костные клетки. До сих пор не доказана возможность массовой дифференцировки МСК в гепатоциты, клетки почек или головного мозга. В то же время рассматриваются два возможных варианта их клинического применения: (1) в качестве средства иммунной модуляции при реакции «трансплантат против хозяина» (РТПХ) и аутоиммунных болезнях или (2) как потенциального источника рост-стимулирующих факторов в специализированных тканях. Эта гетерогенная популяция может поддерживать гемопоэз путем секреции факторов роста, цитокинов и других биологически активных веществ. После введения МСК способны мигрировать в поврежденные ткани, тем самым способствуя их регенерации. Однако только малая часть МСК может достичь костномозговых ниш после внутривенной инфузии. Многочисленные эксперименты с МСК на различных моделях тканевых повреждений показали их способность подавлять апоптоз, вызванный гипоксией, химическими агентами и другими разрушительными факторами. Этот защитный эффект опосредован рядом секретируемых факторов роста, как, например, ГМ-КСФ. В большом числе клинических исследований показана безопасность терапии МСК. Как клинические, так и экспериментальные исследования доказали слабую иммуногенность аллогенных МСК при их введении иммунокомпетентным реципиентам. К настоящему времени проводились инъекции культивированных МСК больным с острой РТПХ после трансплантации гемопоэтических клеток (ТГСК) и при некоторых аутоиммунных заболеваниях. </p> <p> За последнее десятилетие несколько работ кались потенциальной эффективности МСК при введении в пораженные участки костей у пациентов с несовершенным остеогенезом (ОИ) – тяжелым наследственным заболеванием с нарушением структуры коллагена, приводящим к повышенной ломкости костей. Здесь представлены сведения о клиническом протоколе для оценки безопасности, иммуногенности и клиническ их эффектов МСК, вводимых больным ОИ в ходе корригирующей остеотомии. Можно допустить, что небольшая субпопуляция МСК сможет мигрировать в поврежденные участки, дифференцироваться в хондроциты и остеобласты и, тем самым, участвовать в процессах регенерации кости. </p>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(4377) "
Костный мозг (КМ) содержит популяцию резидентных клеток, именуемую «мультипотентными мезенхимными стволовыми клетками» (МСК). Эти адгезивные клетки могут быть выделены и культивированы в простых питательных средах, и способны к дифференцировке в жировые или костные клетки. До сих пор не доказана возможность массовой дифференцировки МСК в гепатоциты, клетки почек или головного мозга. В то же время рассматриваются два возможных варианта их клинического применения: (1) в качестве средства иммунной модуляции при реакции «трансплантат против хозяина» (РТПХ) и аутоиммунных болезнях или (2) как потенциального источника рост-стимулирующих факторов в специализированных тканях. Эта гетерогенная популяция может поддерживать гемопоэз путем секреции факторов роста, цитокинов и других биологически активных веществ. После введения МСК способны мигрировать в поврежденные ткани, тем самым способствуя их регенерации. Однако только малая часть МСК может достичь костномозговых ниш после внутривенной инфузии. Многочисленные эксперименты с МСК на различных моделях тканевых повреждений показали их способность подавлять апоптоз, вызванный гипоксией, химическими агентами и другими разрушительными факторами. Этот защитный эффект опосредован рядом секретируемых факторов роста, как, например, ГМ-КСФ. В большом числе клинических исследований показана безопасность терапии МСК. Как клинические, так и экспериментальные исследования доказали слабую иммуногенность аллогенных МСК при их введении иммунокомпетентным реципиентам. К настоящему времени проводились инъекции культивированных МСК больным с острой РТПХ после трансплантации гемопоэтических клеток (ТГСК) и при некоторых аутоиммунных заболеваниях.
За последнее десятилетие несколько работ кались потенциальной эффективности МСК при введении в пораженные участки костей у пациентов с несовершенным остеогенезом (ОИ) – тяжелым наследственным заболеванием с нарушением структуры коллагена, приводящим к повышенной ломкости костей. Здесь представлены сведения о клиническом протоколе для оценки безопасности, иммуногенности и клиническ их эффектов МСК, вводимых больным ОИ в ходе корригирующей остеотомии. Можно допустить, что небольшая субпопуляция МСК сможет мигрировать в поврежденные участки, дифференцироваться в хондроциты и остеобласты и, тем самым, участвовать в процессах регенерации кости.
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(4377) "Костный мозг (КМ) содержит популяцию резидентных клеток, именуемую «мультипотентными мезенхимными стволовыми клетками» (МСК). Эти адгезивные клетки могут быть выделены и культивированы в простых питательных средах, и способны к дифференцировке в жировые или костные клетки. До сих пор не доказана возможность массовой дифференцировки МСК в гепатоциты, клетки почек или головного мозга. В то же время рассматриваются два возможных варианта их клинического применения: (1) в качестве средства иммунной модуляции при реакции «трансплантат против хозяина» (РТПХ) и аутоиммунных болезнях или (2) как потенциального источника рост-стимулирующих факторов в специализированных тканях. Эта гетерогенная популяция может поддерживать гемопоэз путем секреции факторов роста, цитокинов и других биологически активных веществ. После введения МСК способны мигрировать в поврежденные ткани, тем самым способствуя их регенерации. Однако только малая часть МСК может достичь костномозговых ниш после внутривенной инфузии. Многочисленные эксперименты с МСК на различных моделях тканевых повреждений показали их способность подавлять апоптоз, вызванный гипоксией, химическими агентами и другими разрушительными факторами. Этот защитный эффект опосредован рядом секретируемых факторов роста, как, например, ГМ-КСФ. В большом числе клинических исследований показана безопасность терапии МСК. Как клинические, так и экспериментальные исследования доказали слабую иммуногенность аллогенных МСК при их введении иммунокомпетентным реципиентам. К настоящему времени проводились инъекции культивированных МСК больным с острой РТПХ после трансплантации гемопоэтических клеток (ТГСК) и при некоторых аутоиммунных заболеваниях.
За последнее десятилетие несколько работ кались потенциальной эффективности МСК при введении в пораженные участки костей у пациентов с несовершенным остеогенезом (ОИ) – тяжелым наследственным заболеванием с нарушением структуры коллагена, приводящим к повышенной ломкости костей. Здесь представлены сведения о клиническом протоколе для оценки безопасности, иммуногенности и клиническ их эффектов МСК, вводимых больным ОИ в ходе корригирующей остеотомии. Можно допустить, что небольшая субпопуляция МСК сможет мигрировать в поврежденные участки, дифференцироваться в хондроциты и остеобласты и, тем самым, участвовать в процессах регенерации кости.
" } ["ORGANIZATION_RU"]=> array(37) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8344" ["VALUE"]=> array(2) { ["TEXT"]=> string(349) "НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский<br> государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(343) "НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургскийгосударственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(343) "НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия" } } } [5]=> array(49) { ["IBLOCK_SECTION_ID"]=> string(2) "21" ["~IBLOCK_SECTION_ID"]=> string(2) "21" ["ID"]=> string(3) "694" ["~ID"]=> string(3) "694" ["IBLOCK_ID"]=> string(1) "2" ["~IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["~NAME"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["ACTIVE_FROM"]=> NULL ["~ACTIVE_FROM"]=> NULL ["TIMESTAMP_X"]=> string(22) "05/03/2017 09:37:32 am" ["~TIMESTAMP_X"]=> string(22) "05/03/2017 09:37:32 am" ["DETAIL_PAGE_URL"]=> string(161) "/en/archive/vypusk-5-nomer-4/eksperimentalnye-issledovaniya/sovremennye-metody-i-vozmozhnosti-sekvenirovaniya-sleduyushchego-pokoleniya-ngs-dlya-tipirovaniya-si/" ["~DETAIL_PAGE_URL"]=> string(161) "/en/archive/vypusk-5-nomer-4/eksperimentalnye-issledovaniya/sovremennye-metody-i-vozmozhnosti-sekvenirovaniya-sleduyushchego-pokoleniya-ngs-dlya-tipirovaniya-si/" ["LIST_PAGE_URL"]=> string(12) "/en/archive/" ["~LIST_PAGE_URL"]=> string(12) "/en/archive/" ["DETAIL_TEXT"]=> string(29128) "
Introduction
Allogeneic hematopoietic stem cell transplantation (allo- HSCT) is a proven option for treatment of hematological disorders, e.g., lymphoma, leukemia, and aplastic anemias. Among other factors, the HSCT outcomes strongly depend on aggressive reactions of donor lymphocytes against the patient’s cells. This complication is known as a graft-versushost disease (GVHD) and represents a serious threat to the patients, along with the risk of relapse and concomitant infections [1]. Over last decades, a progress in the donor-recipient matching lead to an imminent increase of patients’ survival post transplant, due to decreased rates of immune complications [3,20]. These advances are made, mostly, due to an improved technique of HLA gene typing [24]. E.g., sufficiently increased survival rates and better outcomes were shown for the patient-donor pairs typed by means of high-resolution allele typing for HLA-A, -B, -C and -DRB1 [27]. Analysis of HLA-DQB1 and -DPB1 loci is still under development [10].
Hence, implementation of high-resolution techniques for the classic HLA typing brings about higher clinical efficiency in allo-HSCT from relatives and unrelated donors. The aspects of optimal donor matching are connected with a sufficient allelic diversity (polymorphism) of the gene loci, mainly, due to gene recombination of the chromosome 6 regions, thus producing repeats at distinct HLA gene segments and causing some difficulties with HLA typing, even at four-digit resolution level. Therefore, novel technological achievements in immunogenetics are of special importance, and, first of all, new approaches to DNA typing [6].
Milestones of molecular HLA typing
Since the very beginning of molecular biology, individual HLA matching was oriented for gene polymorphism analysis. Forty years ago, Fred Sanger has proposed a method of DNA sequencing, by the generation of distinct DNA strands based on a complementary matrix, using DNA polymerase I Klenoff fragment, and then followed by analysis of radioactively labeled DNA fragments in polyacrylamide gel. A breakthrough in this technology and development of routine HLA typing methods occurred with the application of fluorescent dideoxynucleotides in the course of the cyclic process followed by microcapillary electrophoresis. Direct sequencing of heterozygous DNA samples has its limitations since, in the case of allelic polymorphism, the nucleic bases may be located at the same or homologous chromosome, thus precluding detection of subtle differences in the given HLA gene sequence.
Over the last 20 years, the automated Sanger technique is a prevalent approach to genome sequencing in humans, animals, bacteria, and viruses. However, a need for more rapid routine genome screening stimulated novel technologies of multiplex DNA sequencing. These modern methods are depicted as the second-generation approaches (Next-Generation Sequencing, NGS). These technological platforms are based on different strategies, with respect to unique preparations of DNA templates, their sequencing, registration, retrieval and evaluation of the nucleotide sequences by means of novel bioinformatic approaches [13]. A principal benefit of the new-generation sequencing is an opportunity of getting large databases of multiple defined gene sequences within a short time period and at relatively low cost.
A known polymorphism of HLA genes presents a special problem in search of optimal donors for hematopoietic stem cell transplantation (HSCT). E.g., as for September 2016, the International Nomenclature (HLA International ImMunoGeneTics (IMGT) [26, 29] included 15,813 HLA and related alleles, registered in the numerical nomenclature used worldwide (Fig. 1).
imgIn addition to the unique allele number, there are additional optional suffixes that may be added to an allele to indicate its expression status. Alleles that have been shown not to be expressed – “Null” alleles – have been given the suffix ‘N’. Alleles that have been shown to be alternatively expressed may have the suffix “L”, “S”, “C”, “A” or “Q”. The suffix “L” is used to indicate an allele which has been shown to have “Low” cell surface expression when compared to normal levels. The “S” suffix is used to denote an allele specifying a protein which is expressed as a soluble, “Secreted” molecule but is not present on the cell surface. The “C” suffix is assigned to alleles that produce proteins that are present in the “Cytoplasm” and not on the cell surface. An “A” suffix indicates an “Aberrant” expression where there is some doubt as to whether a protein is actually expressed. A “Q” suffix is used when the expression of an allele is “Questionable”, given that the mutation seen in the allele has been shown to affect normal expression levels in other alleles.
No wonder that the general NGS approach adapted for HLA typing proved to be a breakthrough in molecular biology applications being quite promising to the transplantation clinics and bone marrow donor registries. However, to promote the NGS implementation, we need specialized typing strategies and digital program algorithms. The sequencing costs per single run are sharply decreased with NGS approach which may be quite accessible to the tissue typing laboratories in sooner time.
Multiple NGS technological platforms are offered on the market. At the present time, four types of NGS systems are implemented in research and clinics. E.g., the 454 Junior (Roche) was the first system equipped with commercial kits for HLA-typing. Illumina platform is most widely applied in fundamental and clinical research, however, not yet registered for HLA-typing. The Ion Torrent system was offered something later, being, however, a growing point in the field. Lately, the fourth generation of NGS appears – Single Molecule Sequencing. They can do long reads with very high throughput. Advantages: Low Cost, Simultaneous sequencing of several samples (barcoding). Currently manufactured by 2 companies, PacBio and Nanopore MiniON. They are amplicon-based and “true” single molecule sequencing. It has a very small footprint and even fits in the pocket.
At the present time, a four-digit resolution approach is used for optimal matching in the donor-recipient pairs, i.e., testing of HLA-A, -B, -C, (exons 2, 3); -DRB1 (exon 2), and – DQB1 (exons 2, 3) by means of conventional PCR technique.
Pitfalls in NGS-based HLA typing
To perform high-resolution typing, one should exclude non-coding alleles from HLA-A (exons 1, 4 and introns 2, 4), HLA-B (exons 1, 4 and intron 1), and HLA-C (exons 1, 4, 7).
Phase determination of the sequence motifs becomes an increasingly difficult task, due to new HLA polymorphisms reported, thus causing an exponential increase in genotyping ambiguity with every ongoing release of the HLA database release [10]. Therefore, novel typing strategies have been arranged, e.g., group-specific PCR [15], or allelic discrimination with distinct group-specific primers [18] before subsequent sequencing. These discrimination techniques are used at HLA laboratories for a long time. However, they are labor-consuming, and appropriate commercial kits are largely lacking.
The novel NGS-based HLA typing was developed over 2000’s, having been characterized by increased resolution ability and high throughput [22, 30]. In 2009, Gabriel et al. [11] and Erlich et al. [1] have independently shown an opportunity of HLA typing by means of a 454 technological platform. A double-blinded multicentric study based on exon amplification by primers’ annealing was performed and reported in 2011 [13]. This cooperative work has shown that HLA sequencing with 454 system allows reliable identification of the HLA genotypes. However, the analytic approaches proved to be quite diligent, labor-intensive and require automation. Moreover, a systemic algorithm for the Class I HLA typing was performed with different PCR-based barcoding methods [9]. Later on, Lank et al. [16, 19] proposed a DNA processing protocol for HLA typing with a less complicated DNA library preparation. Finally, it was shown that, as unlike with amplicon sequencing, the shotgun sequencing approach using long PCR products, fragmenting and ligation of the multiplex identifiers (MID) is a realistic way for the entire HLA gene sequencing. [20, 27, 32].
Meanwhile, the existing protocols provide additional proofs of NGS complexity. This technology is still difficult to introduce in laboratories working in the area of histocompatibility and immunogenetics. There were some unresolved technical issues concerning labor intensity of a gene library preparation, and too long time required for processing of the databases produced. Noteworthy, the bioinformatic tools in NGS rarely generated a readable report of HLA typing.
Taking this into account, an automated approach to the high-resolutionHLA typing was developed, being adapted for the 454 GS Junior, covering 17 exons of the following genes: HLA-A, -B, -C, -DQB1, -DPB1, -DRB1, -DRB3, -DRB4 и -DRB5 (DRB3 / 4/5). However, exact data on 5”- and 3”- untranslated sequences (UTR) are not available for all the alleles. Therefore, binding of primers in these regions cannot be definitely predicted, thus missing some alleles from analysis or nucleotide shifts due to suboptimal amplification primers. Another potential problem may be due to simultaneous amplification of pseudogenes since their sequences are quite similar to those of target HLA genes.
Despite these issues, NGS has a great future in the area. An opportunity of the whole-gene analysis should ultimately lead to better understanding of HLA role for HSCT outcomes and the course of the primary disease. Moreover, the zero alleles could be better defined. So far, we cannot exploit this benefit. However, some questions will be answered, e.g., assessing a “complete” HLA allele database and overcoming existing hurdles when assembling a single complete allelic sequence.
At the initial step, a maximal length of sequences readable with Myseq was limited of 2×150 bp. However, the manufacturer (Illumina) improves its software, as well as chemical equipment. Hence, their last version (version 3) allows sequencing of ca.300 bp, in order to cover one exon within each sequenced amplicon, thus diminishing potential error rates and simplifying the work process.
Main steps of the NGS as performed with Illumina platform
- Template generation: the complete sequences of HLA genes are amplified with complementary primers in a single reaction, using Long-Range DNA polymerase.
- Double-stranded DNA fragmentation by means of specific fragmentase optimized by its size for the given HLA locus
- Preparation of gene libraries: Appropriate reagents are used for the end repair, 5’ phosphorylation of poly-A and poly-T ends for subsequent adaptors’ ligation.
- Adapters’ ligation and their indexing.
- Mixing of the samples and their loading to the NGS sequencer.
- Analysis of results (Fig.1)
Following generation of a gene library, the DNA molecules are bound to the solid phase carrier, the s.c. flow well, by means of direct and reverse oligonucleotide adapters. During generation of the gene library, the complementary (adapter) sequences are fixed to the templates at the 5”- and 3”-ends, thus allowing them to bind the flow well surface covered with adapter molecules. To enhance the sequencing signal, molecular clusters are generated from one DNA molecule by means of solid-phase amplification bridge, resulting in up to 1000 tightly packed copies of the initial template in the flow well. Hence, each cluster corresponds to a single read by the end of sequencing. The adapter oligonucleotides are bound to the surface of flow well, both for the capture of sequenced templates molecules, and primers for bridge amplification. Sequencing is then carried out by means of chemical reaction “reversible terminator’” All 4 fluorescently labeled oligonucleotides are simultaneously driven through the flow well. Their labels are intended for termination of the chain synthesis. Therefore, only one nucleotide type is added for each polymerization cycle. The nonbound nucleotides are washed off. The labels and terminator are removed following CCD-assisted conversion of the pictures, in order to allow further elongation of the chain at the next sequencing cycle. To facilitate the multiplexing procedure, the adapter sequences are followed by the s.c. index sequences (DNA barcoding). These DNA barcodes are attached to the target fragments during the library design, with a unique code for given sample. Double indexing is possible by differential DNA barcoding from 5” to 3” end. In the course of four sequencing rounds, two indexing reads and two reads of target sequences are initiated. The data are accumulated for each single cluster and are used for generation of the output files in two directions of sequencing.
HLA data analysis with MiSeq platform
Miseq generates a working file in FASTQ format with two corresponding files (resp., direct and reverse reads). All the files contain equal numbers of the read sequences presented in the same order. Moreover, all the sequences read are of equal length according to the chosen specifications. Homogeneous structure of initial data provides some benefits for the pre-analytic data handling. Upon filtration and cutting, some algorithms are required here which are more applicable than with platforms generating variable reading length (e.g., Roche 454, or Ion Torrent systems). Data quality obtained with Miseq is high enough to perform HLA typing.
To assign the HLA alleles, software is required which allows communicating with updated HLA databases (IMGT last version, renewed 4 times a year). A number of such commercial packages are currently available. To date, the solutions with an open initial code are also offered, as follows:
HLAminer: http://genomemedicine.com/content/4/12/95
seq2HLA: http://genomemedicine.com/content/4/12/102
Athlates: http://nar.oxfordjournals.org/content/41/14/e142. long
HLAforest: http://www.plosone.org/article/info%3Adoi%2F10. 1371%2Fjournal.pone.0067885
PHLAT: http://www.biomedcentral.com/1471-2164/15/325
According to the guidelines of clinical trials and in order to observe acting regulations (European Federation for Immunogenetics (EFI), American Society for Histocompatibility and Immunogenetics (ASHI) standards), extensive studies of QC and QA are required for development of appropriate in-house reagents, and their testing for HLA typing bases on the Miseq platform. They should include both developments of the tests, and methods of automated data mining, and their presentation. Sufficient time is needed for design, feasibility evaluation, and implementation of NGS working protocols. The optimal choice of NGS platform and proper protocol is a difficult task for any individual setting in this rapidly developing technology [14].
Modern protocols of sequencing and data retrieval/handling differ, generally, in the aims of analysis, i.e., whole-genome evaluation (from 5’UTR to 3’UTR), or exome studies. So far, a limit of 400 bp exists for the length of sequencing, independently of the working protocol applied. This length of DNA fragment is sufficient to reading of whole exons and providing unequivocal clonal information for these exons. To sequence longer PCR products, a series of overlapping fragments is required, followed by their subsequent assembly (shotgun sequencing). The working processes under these two protocols significantly differ in labor intensity and complexity.
The output data are collected for each molecular cluster and used for generation of the output files containing results of direct and reverse reads as displayed on Fig. 2 [5]
imgThe detailed information concerns HLA-A genotype assignment, as show in the master layer. Direct and reverse sequence reads are aligned to exons 2, 3, and 4, with partial coverage of intronic regions [33]. Read numbers and sequence direction are shown in white cells. For example, exon 4 is represented by two (alleles 1 and 2) direct and reverse sequences, with 90 coverages, and, respectively, 81 nucleotides read, and two reverse sequences with 82 and 86 nucleotides read. Low case: exon map, consensus sequence, and genotype assigned. Stepwise distribution of the sequences at exon2/intron 2 border is also shown, and an HLA-A * 01: 01: 01: 01, 24: 02: 01: 01 combination is proposed with zero mismatch conditions (MM master layer) as compared with the database. A non-expressed (А* 01: 01: 01: 02 N), and low-expressed (А * 24: 02: 01: 02 L) variants are discriminated by means of exclusion of intron 2 variants, as shown in the noncoding column (N-C). Discordances in phase layers 3 and 4 (ММ3, MM4) demonstrate potential ambiguities which could occur if the SNP phase in exons 3 and 2 could not be revealed (with conventional Sanger sequencing). Other detectable alleles, A * 24: 02: 01: 03 (change in intron 3), and A * 24: 02: 40 (synonymous substitution in exon 5) cannot be excluded, and therefore, are displayed in a similar way with zero mismatches. [5]
Factors to consider in cost analysis of in-depth sequencing
Reagents for the entire process include those used for conventional pre-analytic methods (e.g., DNA extraction, quality assessment, and initial low-resolution typing step. Additional expenditures are a subject to some ambiguities, due to different prices reagents and equipment offered by distinct manufacturers. Moreover, it should be stressed that all commercial NGS systems are of closed type, thus causing broad variations in prices for the entire NGS procedure per single DNA sample, and depending on the per year capacity of the given HLA typing laboratory.
However, even taking into account maintenance costs (estimated 10% equipment cost), usage of core facilities or shared equipment, the Sanger sequencing (220 K) proves to be twice more expensive than NGS (variable, all less than Sanger), as shown in Fig.3. I.e., the sample preparation costs remain the same however the sequencing cost decreases (Fig. 4).
img img imgCurrent progress and future directions
In 2005, Rochе produced the first NGS instrument, the Genome Sequencer 20. By means of this device, 100-bp reads are possible, and in future, up to 20 Mbp could be sequenced. Over last decade, rapid progress in NGS technologies has led to revolutionary changes in general genomics and its applications for clinical sequencing or medical exomics (RNA expression analysis). However, PCR-SSO and PCR-sequencing still remain the first-line methods in HLA typing. Meanhwile, commercial kits for NGS-assisted HLA typing have been recently appeared [8]. Hence, elucidation of the whole HLA gene sequence will bring a knowledge which will be of value for medical science in sooner time. However, this knowledge will be insufficient to the total understanding of MHS and HLA interactions. One should assess the sequences and haplotypes of other HLA segments, including regulatory (non-coding) regions [12]. Further analysis should determine transcriptional factors and events regulating the entire HLA unit, including HLA genes and relevant gene structures. They all should be carefully studied, in order to get complete HLA description, i.e., HLA-omics [4]. These efforts will bring real clinical benefits for the patients. Novel methods of HLA typing will be helpful to more accurate medical examination, assignment of biologically diverse clinical groups and individualized treatment of the patients.
Conclusions
A routine tissue-typing laboratory should be flexible, providing valid and reproducible HLA data within short time period. NGS-based HLA analysis is performed with 100% reliability, and well fits the tasks of HLA typing in unrelated donors, in concordance with EFI and ASHI policies. This work process well corresponds to the working schedules for medium- and high-capacity laboratories, thus being potentially attractive to the donor registries.
Recently introduced next-generation sequencing techniques have a facilitating potential for the high-resolution genotyping via decrease of general uncertainty, like as due to extended sequencing regions. The entire set of MHS genes could be sequenced by means of these technologies, in order to uncover the latent immune determinants relevant to autoaggresive reactions [7, 25]. In near future, the whole exome sequencing may be considered a method of decoding some genetic variants, influencing HSCT outcomes. The NGS approaches will be a quite effective and cost-effective technology when evaluating histocompatibility parameters and immunogenetic interactions.
Conflict of interests
No conflict of interests is declared.
References
- Bentley G, Higuchi R, Hoglund B, Goodridge D, Sayer D, Trachtenberg EA, Erlich HA: High-resolution, high-throughput HLA genotyping by nextgeneration sequencing. Tissue Antigens 2009; 74: 393–403.
- Blazar BR, Murphy WJ, Abedi M: Advances in graftversus- host disease biology and therapy. Nat Rev Immunol 2012; 12: 443–458.
- Brunstein CG, Eapen M, Ahn KW, Appelbaum FR, Ballen KK, Champlin RE, Cutler C, Kan F, Laughlin MJ, Soiffer RJ, Weisdorf DJ, Woolfrey A, Wagner JE: Reduced-intensity conditioning transplantation in acute leukemia: the effect of source of unrelated donor stem cells on outcomes. Blood 2012; 119: 5591–5598.
- Danzer M, Polin H, Proll J, Hofer K, Fae I, Fischer GF, Gabriel C: Highthroughput sequence-based typing strategy for HLA-DRB1 based on real-time polymerase chain reaction. Hum Immunol 2007; 68: 915–917.
- Danzer M, Niklas N, Stabentheiner S, Hofer K, Pröll J, Stückler C, Raml E, Polinand H, Gabriel C. Rapid, scalable and highly automated HLA genotyping using next-generation sequencing: a transition from research to diagnostics. BMC Genomics 2013, 14: 221.
- Dunn PP: Human leucocyte antigen typing: techniques and technology, a critical appraisal. Int J Immunogenet 2011; 38: 463–473.
- Elsner HA, Blasczyk R: Immunogenetics of HLA null alleles: implications for blood stem cell transplantation. Tissue Antigens 2004; 64: 687–695.
- Erlich H: HLA DNA typing: past, present, and future. Tissue Antigens 2012; 80: 1–11.
- Erlich RL, Jia X, Anderson S, Banks E, Gao X, Carrington M, Gupta N, DePristo MA, Henn MR, Lennon NJ, de Bakker PI: Next-generation sequencing for HLA typing of class I loci. BMC Genomics 2011; 12: 42.
- Fleischhauer K, Shaw BE, Gooley T, Malkki M, Bardy P, Bignon JD, Dubois V, Horowitz MM, Madrigal JA, Morishima Y, Oudshoorn M, Ringden O, Spellman S, Velardi A, Zino E, Petersdorf EW: Effect of T-cell-epitope matching at HLA-DPB1 in recipients of unrelated-donor haemopoieticcell transplantation: a retrospective study. Lancet Oncol 2012; 13: 366–374.
- Gabriel C, Danzer M, Hackl C, Kopal G, Hufnagl P, Hofer K, Polin H, Stabentheiner S, Proll J: Rapid high-throughput human leukocyte antigen typing by massively parallel pyrosequencing for high-resolution allele identification. Hum Immunol; 2009, 70: 960–964.
- Harismendy O, Frazer K: Method for improving sequence coverage uniformity of targeted genomic intervals amplified by LR-PCR using Illumina GA sequencing-by-synthesis technology. Biotechniques 2009;46:229–231.
- Holcomb CL, Hoglund B, Anderson MW, Blake LA, Bohme I, Egholm M, Ferriola D, Gabriel C, Gelber SE, Goodridge D, Hawbecker S, Klein R, Ladner M, Lind C, Monos D, Pando MJ, Proll J, Sayer DC, Schmitz-Agheguian G, Simen BB, Thiele B, Trachtenberg EA, Tyan DB, Wassmuth R, White S, Erlich HA: A multi-site study using high-resolution HLA genotyping by next generation sequencing. Tissue Antigens 2011, 77: 206–217.
- Huse SM, Huber JA, Morrison HG, Sogin ML, Welch DM: Accuracy and quality of massively parallel DNA pyrosequencing. Genome Biol 2007; 8: R143.
- Kotsch K, Wehling J, Blasczyk R: Sequencing of HLA class II genes based on the conserved diversity of the non-coding regions: sequencing based typing of HLA-DRB genes. Tissue Antigens 1999; 53: 486–497.
- Lank SM, Golbach BA, Creager HM, Wiseman RW, Keskin DB, Reinherz EL, Brusic V, O’Connor DH: Ultra-high resolution HLA genotyping and allele discovery by highly multiplexed cDNA amplicon pyrosequencing. BMC Genomics 2012; 13: 378.
- Lank SM, Wiseman RW, Dudley DM, O’Connor DH: A novel single cDNA amplicon pyrosequencing method for high-throughput, cost-effective sequence-based HLA class I genotyping. Hum Immunol 2010; 71: 1011–1017.
- Lebedeva TV, Mastromarino SA, Lee E, Ohashi M, Alosco SM, Yu N: Resolution of HLA class I sequence-based typing ambiguities by groupspecific sequencing primers. Tissue Antigens 2011, 77: 247–250.
- Baxter-Lowe LA. Tailoring NGS for smaller volume labs. 42nd ASHI Annual Meeting. St. Louis, MO, Poster report (Sept 28, 2016).
- Li HW, Sykes M: Emerging concepts in haematopoietic cell transplantation. Nat Rev Immunol 2012; 12: 403–416.
- Lind C, Ferriola D, Mackiewicz K, Heron S, Rogers M, Slavich L, Walker R, Hsiao T, McLaughlin L, D’Arcy M, Gai X, Goodridge D, Sayer D, Monos D: Next-generation sequencing: the solution for high-resolution, unambiguous human leukocyte antigen typing. Hum Immunol 2010; 71: 1033–1042.
- McDevitt SL, Bredeson JV, Roy SW, Lane JA, Noble JA. HAPCAD: An open-source tool to detect PCR crossovers in next-generation sequencing generated HLA data. Hum Immunol. 2016; 77 (3): 257-63. doi: 10.1016/j.humimm. 2016.01.013.
- Metzker ML: Sequencing technologies – the next generation. Nat Rev Genet 2010; 11: 31–46.
- Petersdorf EW: Optimal HLA matching in hematopoietic cell transplantation. Curr Opin Immunol 2008; 20: 588–593.
- Proll J, Danzer M, Stabentheiner S, Niklas N, Hackl C, Hofer K, Atzmuller S, Hufnagl P, Gully C, Hauser H, Krieger O, Gabriel C: Sequence capture and next generation resequencing of the MHC region highlights potential transplantation determinants in HLA identical haematopoietic stem cell transplantation. DNA Res 2011; 18: 201–210.
- Robinson J, Halliwell JA, Hayhurst JD, Flicek P, Parham P, Marsh SG. The IPD and IMGT/HLA database: allele variant databases. Nucleic Acids Res 2015; 43(Database issue): D423-31. doi: 10.1093/nar/gku1161.
- Shaw BE, Arguello R, Garcia-Sepulveda CA, Madrigal JA: The impact of HLA genotyping on survival following unrelated donor haematopoietic stem cell transplantation. Br J Haematol 2010; 150: 251–258.
- Shiina T, Suzuki S, Ozaki Y, Taira H, Kikkawa E, Shigenari A, Oka A, UmemuraT, Joshita S, Takahashi O, Hayashi Y, Paumen M, Katsuyama Y, Mitsunaga S, Ota M, Kulski JK, Inoko H: Super high resolution for single molecule sequence-based typing of classical HLA loci at the 8-digit level using next generation sequencers. Tissue Antigens 2012; 80: 305–316.
- Marsh SG, WHO Nomenclature Committee for Factors of the HLA System. Nomenclature for factors of the HLA system, update March 2016. Human Immunology 2016; 77 (8): 706-709.
- Voorter CE, Mulkers E, Liebelt P, Sleyster E, van den Berg-Loonen EM: Reanalysis of sequence-based HLA-A, -B and -Cw typings: how ambiguous is today’s SBT typing tomorrow. Tissue Antigens 2007; 70: 383–389.
- Voelkerding KV, Dames SA, Durtschi JD. Next-generation sequencing: from basic research to diagnostics. Clin Chem 2009; 55: 641–658.
- Wang C, Krishnakumar S, Wilhelmy J, Babrzadeh F, Stepanyan L, Su LF, Levinson D, Fernandez-Vina MA, Davis RW, Davis MM, Mindrinos M: Highthroughput, high-fidelity HLA genotyping with deep sequencing. Proc Natl Acad Sci USA 2012; 109: 8676–8681.
- Weimer ET, Montgomery M, Petraroia R, Crawford J, Schmitz JL. Performance characteristics and validation of next-generation sequencing for human leucocyte antigen typing. J Mol Diagnostics 2016; 18 (5): 668-675.
" ["~DETAIL_TEXT"]=> string(29128) "
Introduction
Allogeneic hematopoietic stem cell transplantation (allo- HSCT) is a proven option for treatment of hematological disorders, e.g., lymphoma, leukemia, and aplastic anemias. Among other factors, the HSCT outcomes strongly depend on aggressive reactions of donor lymphocytes against the patient’s cells. This complication is known as a graft-versushost disease (GVHD) and represents a serious threat to the patients, along with the risk of relapse and concomitant infections [1]. Over last decades, a progress in the donor-recipient matching lead to an imminent increase of patients’ survival post transplant, due to decreased rates of immune complications [3,20]. These advances are made, mostly, due to an improved technique of HLA gene typing [24]. E.g., sufficiently increased survival rates and better outcomes were shown for the patient-donor pairs typed by means of high-resolution allele typing for HLA-A, -B, -C and -DRB1 [27]. Analysis of HLA-DQB1 and -DPB1 loci is still under development [10].
Hence, implementation of high-resolution techniques for the classic HLA typing brings about higher clinical efficiency in allo-HSCT from relatives and unrelated donors. The aspects of optimal donor matching are connected with a sufficient allelic diversity (polymorphism) of the gene loci, mainly, due to gene recombination of the chromosome 6 regions, thus producing repeats at distinct HLA gene segments and causing some difficulties with HLA typing, even at four-digit resolution level. Therefore, novel technological achievements in immunogenetics are of special importance, and, first of all, new approaches to DNA typing [6].
Milestones of molecular HLA typing
Since the very beginning of molecular biology, individual HLA matching was oriented for gene polymorphism analysis. Forty years ago, Fred Sanger has proposed a method of DNA sequencing, by the generation of distinct DNA strands based on a complementary matrix, using DNA polymerase I Klenoff fragment, and then followed by analysis of radioactively labeled DNA fragments in polyacrylamide gel. A breakthrough in this technology and development of routine HLA typing methods occurred with the application of fluorescent dideoxynucleotides in the course of the cyclic process followed by microcapillary electrophoresis. Direct sequencing of heterozygous DNA samples has its limitations since, in the case of allelic polymorphism, the nucleic bases may be located at the same or homologous chromosome, thus precluding detection of subtle differences in the given HLA gene sequence.
Over the last 20 years, the automated Sanger technique is a prevalent approach to genome sequencing in humans, animals, bacteria, and viruses. However, a need for more rapid routine genome screening stimulated novel technologies of multiplex DNA sequencing. These modern methods are depicted as the second-generation approaches (Next-Generation Sequencing, NGS). These technological platforms are based on different strategies, with respect to unique preparations of DNA templates, their sequencing, registration, retrieval and evaluation of the nucleotide sequences by means of novel bioinformatic approaches [13]. A principal benefit of the new-generation sequencing is an opportunity of getting large databases of multiple defined gene sequences within a short time period and at relatively low cost.
A known polymorphism of HLA genes presents a special problem in search of optimal donors for hematopoietic stem cell transplantation (HSCT). E.g., as for September 2016, the International Nomenclature (HLA International ImMunoGeneTics (IMGT) [26, 29] included 15,813 HLA and related alleles, registered in the numerical nomenclature used worldwide (Fig. 1).
imgIn addition to the unique allele number, there are additional optional suffixes that may be added to an allele to indicate its expression status. Alleles that have been shown not to be expressed – “Null” alleles – have been given the suffix ‘N’. Alleles that have been shown to be alternatively expressed may have the suffix “L”, “S”, “C”, “A” or “Q”. The suffix “L” is used to indicate an allele which has been shown to have “Low” cell surface expression when compared to normal levels. The “S” suffix is used to denote an allele specifying a protein which is expressed as a soluble, “Secreted” molecule but is not present on the cell surface. The “C” suffix is assigned to alleles that produce proteins that are present in the “Cytoplasm” and not on the cell surface. An “A” suffix indicates an “Aberrant” expression where there is some doubt as to whether a protein is actually expressed. A “Q” suffix is used when the expression of an allele is “Questionable”, given that the mutation seen in the allele has been shown to affect normal expression levels in other alleles.
No wonder that the general NGS approach adapted for HLA typing proved to be a breakthrough in molecular biology applications being quite promising to the transplantation clinics and bone marrow donor registries. However, to promote the NGS implementation, we need specialized typing strategies and digital program algorithms. The sequencing costs per single run are sharply decreased with NGS approach which may be quite accessible to the tissue typing laboratories in sooner time.
Multiple NGS technological platforms are offered on the market. At the present time, four types of NGS systems are implemented in research and clinics. E.g., the 454 Junior (Roche) was the first system equipped with commercial kits for HLA-typing. Illumina platform is most widely applied in fundamental and clinical research, however, not yet registered for HLA-typing. The Ion Torrent system was offered something later, being, however, a growing point in the field. Lately, the fourth generation of NGS appears – Single Molecule Sequencing. They can do long reads with very high throughput. Advantages: Low Cost, Simultaneous sequencing of several samples (barcoding). Currently manufactured by 2 companies, PacBio and Nanopore MiniON. They are amplicon-based and “true” single molecule sequencing. It has a very small footprint and even fits in the pocket.
At the present time, a four-digit resolution approach is used for optimal matching in the donor-recipient pairs, i.e., testing of HLA-A, -B, -C, (exons 2, 3); -DRB1 (exon 2), and – DQB1 (exons 2, 3) by means of conventional PCR technique.
Pitfalls in NGS-based HLA typing
To perform high-resolution typing, one should exclude non-coding alleles from HLA-A (exons 1, 4 and introns 2, 4), HLA-B (exons 1, 4 and intron 1), and HLA-C (exons 1, 4, 7).
Phase determination of the sequence motifs becomes an increasingly difficult task, due to new HLA polymorphisms reported, thus causing an exponential increase in genotyping ambiguity with every ongoing release of the HLA database release [10]. Therefore, novel typing strategies have been arranged, e.g., group-specific PCR [15], or allelic discrimination with distinct group-specific primers [18] before subsequent sequencing. These discrimination techniques are used at HLA laboratories for a long time. However, they are labor-consuming, and appropriate commercial kits are largely lacking.
The novel NGS-based HLA typing was developed over 2000’s, having been characterized by increased resolution ability and high throughput [22, 30]. In 2009, Gabriel et al. [11] and Erlich et al. [1] have independently shown an opportunity of HLA typing by means of a 454 technological platform. A double-blinded multicentric study based on exon amplification by primers’ annealing was performed and reported in 2011 [13]. This cooperative work has shown that HLA sequencing with 454 system allows reliable identification of the HLA genotypes. However, the analytic approaches proved to be quite diligent, labor-intensive and require automation. Moreover, a systemic algorithm for the Class I HLA typing was performed with different PCR-based barcoding methods [9]. Later on, Lank et al. [16, 19] proposed a DNA processing protocol for HLA typing with a less complicated DNA library preparation. Finally, it was shown that, as unlike with amplicon sequencing, the shotgun sequencing approach using long PCR products, fragmenting and ligation of the multiplex identifiers (MID) is a realistic way for the entire HLA gene sequencing. [20, 27, 32].
Meanwhile, the existing protocols provide additional proofs of NGS complexity. This technology is still difficult to introduce in laboratories working in the area of histocompatibility and immunogenetics. There were some unresolved technical issues concerning labor intensity of a gene library preparation, and too long time required for processing of the databases produced. Noteworthy, the bioinformatic tools in NGS rarely generated a readable report of HLA typing.
Taking this into account, an automated approach to the high-resolutionHLA typing was developed, being adapted for the 454 GS Junior, covering 17 exons of the following genes: HLA-A, -B, -C, -DQB1, -DPB1, -DRB1, -DRB3, -DRB4 и -DRB5 (DRB3 / 4/5). However, exact data on 5”- and 3”- untranslated sequences (UTR) are not available for all the alleles. Therefore, binding of primers in these regions cannot be definitely predicted, thus missing some alleles from analysis or nucleotide shifts due to suboptimal amplification primers. Another potential problem may be due to simultaneous amplification of pseudogenes since their sequences are quite similar to those of target HLA genes.
Despite these issues, NGS has a great future in the area. An opportunity of the whole-gene analysis should ultimately lead to better understanding of HLA role for HSCT outcomes and the course of the primary disease. Moreover, the zero alleles could be better defined. So far, we cannot exploit this benefit. However, some questions will be answered, e.g., assessing a “complete” HLA allele database and overcoming existing hurdles when assembling a single complete allelic sequence.
At the initial step, a maximal length of sequences readable with Myseq was limited of 2×150 bp. However, the manufacturer (Illumina) improves its software, as well as chemical equipment. Hence, their last version (version 3) allows sequencing of ca.300 bp, in order to cover one exon within each sequenced amplicon, thus diminishing potential error rates and simplifying the work process.
Main steps of the NGS as performed with Illumina platform
- Template generation: the complete sequences of HLA genes are amplified with complementary primers in a single reaction, using Long-Range DNA polymerase.
- Double-stranded DNA fragmentation by means of specific fragmentase optimized by its size for the given HLA locus
- Preparation of gene libraries: Appropriate reagents are used for the end repair, 5’ phosphorylation of poly-A and poly-T ends for subsequent adaptors’ ligation.
- Adapters’ ligation and their indexing.
- Mixing of the samples and their loading to the NGS sequencer.
- Analysis of results (Fig.1)
Following generation of a gene library, the DNA molecules are bound to the solid phase carrier, the s.c. flow well, by means of direct and reverse oligonucleotide adapters. During generation of the gene library, the complementary (adapter) sequences are fixed to the templates at the 5”- and 3”-ends, thus allowing them to bind the flow well surface covered with adapter molecules. To enhance the sequencing signal, molecular clusters are generated from one DNA molecule by means of solid-phase amplification bridge, resulting in up to 1000 tightly packed copies of the initial template in the flow well. Hence, each cluster corresponds to a single read by the end of sequencing. The adapter oligonucleotides are bound to the surface of flow well, both for the capture of sequenced templates molecules, and primers for bridge amplification. Sequencing is then carried out by means of chemical reaction “reversible terminator’” All 4 fluorescently labeled oligonucleotides are simultaneously driven through the flow well. Their labels are intended for termination of the chain synthesis. Therefore, only one nucleotide type is added for each polymerization cycle. The nonbound nucleotides are washed off. The labels and terminator are removed following CCD-assisted conversion of the pictures, in order to allow further elongation of the chain at the next sequencing cycle. To facilitate the multiplexing procedure, the adapter sequences are followed by the s.c. index sequences (DNA barcoding). These DNA barcodes are attached to the target fragments during the library design, with a unique code for given sample. Double indexing is possible by differential DNA barcoding from 5” to 3” end. In the course of four sequencing rounds, two indexing reads and two reads of target sequences are initiated. The data are accumulated for each single cluster and are used for generation of the output files in two directions of sequencing.
HLA data analysis with MiSeq platform
Miseq generates a working file in FASTQ format with two corresponding files (resp., direct and reverse reads). All the files contain equal numbers of the read sequences presented in the same order. Moreover, all the sequences read are of equal length according to the chosen specifications. Homogeneous structure of initial data provides some benefits for the pre-analytic data handling. Upon filtration and cutting, some algorithms are required here which are more applicable than with platforms generating variable reading length (e.g., Roche 454, or Ion Torrent systems). Data quality obtained with Miseq is high enough to perform HLA typing.
To assign the HLA alleles, software is required which allows communicating with updated HLA databases (IMGT last version, renewed 4 times a year). A number of such commercial packages are currently available. To date, the solutions with an open initial code are also offered, as follows:
HLAminer: http://genomemedicine.com/content/4/12/95
seq2HLA: http://genomemedicine.com/content/4/12/102
Athlates: http://nar.oxfordjournals.org/content/41/14/e142. long
HLAforest: http://www.plosone.org/article/info%3Adoi%2F10. 1371%2Fjournal.pone.0067885
PHLAT: http://www.biomedcentral.com/1471-2164/15/325
According to the guidelines of clinical trials and in order to observe acting regulations (European Federation for Immunogenetics (EFI), American Society for Histocompatibility and Immunogenetics (ASHI) standards), extensive studies of QC and QA are required for development of appropriate in-house reagents, and their testing for HLA typing bases on the Miseq platform. They should include both developments of the tests, and methods of automated data mining, and their presentation. Sufficient time is needed for design, feasibility evaluation, and implementation of NGS working protocols. The optimal choice of NGS platform and proper protocol is a difficult task for any individual setting in this rapidly developing technology [14].
Modern protocols of sequencing and data retrieval/handling differ, generally, in the aims of analysis, i.e., whole-genome evaluation (from 5’UTR to 3’UTR), or exome studies. So far, a limit of 400 bp exists for the length of sequencing, independently of the working protocol applied. This length of DNA fragment is sufficient to reading of whole exons and providing unequivocal clonal information for these exons. To sequence longer PCR products, a series of overlapping fragments is required, followed by their subsequent assembly (shotgun sequencing). The working processes under these two protocols significantly differ in labor intensity and complexity.
The output data are collected for each molecular cluster and used for generation of the output files containing results of direct and reverse reads as displayed on Fig. 2 [5]
imgThe detailed information concerns HLA-A genotype assignment, as show in the master layer. Direct and reverse sequence reads are aligned to exons 2, 3, and 4, with partial coverage of intronic regions [33]. Read numbers and sequence direction are shown in white cells. For example, exon 4 is represented by two (alleles 1 and 2) direct and reverse sequences, with 90 coverages, and, respectively, 81 nucleotides read, and two reverse sequences with 82 and 86 nucleotides read. Low case: exon map, consensus sequence, and genotype assigned. Stepwise distribution of the sequences at exon2/intron 2 border is also shown, and an HLA-A * 01: 01: 01: 01, 24: 02: 01: 01 combination is proposed with zero mismatch conditions (MM master layer) as compared with the database. A non-expressed (А* 01: 01: 01: 02 N), and low-expressed (А * 24: 02: 01: 02 L) variants are discriminated by means of exclusion of intron 2 variants, as shown in the noncoding column (N-C). Discordances in phase layers 3 and 4 (ММ3, MM4) demonstrate potential ambiguities which could occur if the SNP phase in exons 3 and 2 could not be revealed (with conventional Sanger sequencing). Other detectable alleles, A * 24: 02: 01: 03 (change in intron 3), and A * 24: 02: 40 (synonymous substitution in exon 5) cannot be excluded, and therefore, are displayed in a similar way with zero mismatches. [5]
Factors to consider in cost analysis of in-depth sequencing
Reagents for the entire process include those used for conventional pre-analytic methods (e.g., DNA extraction, quality assessment, and initial low-resolution typing step. Additional expenditures are a subject to some ambiguities, due to different prices reagents and equipment offered by distinct manufacturers. Moreover, it should be stressed that all commercial NGS systems are of closed type, thus causing broad variations in prices for the entire NGS procedure per single DNA sample, and depending on the per year capacity of the given HLA typing laboratory.
However, even taking into account maintenance costs (estimated 10% equipment cost), usage of core facilities or shared equipment, the Sanger sequencing (220 K) proves to be twice more expensive than NGS (variable, all less than Sanger), as shown in Fig.3. I.e., the sample preparation costs remain the same however the sequencing cost decreases (Fig. 4).
img img imgCurrent progress and future directions
In 2005, Rochе produced the first NGS instrument, the Genome Sequencer 20. By means of this device, 100-bp reads are possible, and in future, up to 20 Mbp could be sequenced. Over last decade, rapid progress in NGS technologies has led to revolutionary changes in general genomics and its applications for clinical sequencing or medical exomics (RNA expression analysis). However, PCR-SSO and PCR-sequencing still remain the first-line methods in HLA typing. Meanhwile, commercial kits for NGS-assisted HLA typing have been recently appeared [8]. Hence, elucidation of the whole HLA gene sequence will bring a knowledge which will be of value for medical science in sooner time. However, this knowledge will be insufficient to the total understanding of MHS and HLA interactions. One should assess the sequences and haplotypes of other HLA segments, including regulatory (non-coding) regions [12]. Further analysis should determine transcriptional factors and events regulating the entire HLA unit, including HLA genes and relevant gene structures. They all should be carefully studied, in order to get complete HLA description, i.e., HLA-omics [4]. These efforts will bring real clinical benefits for the patients. Novel methods of HLA typing will be helpful to more accurate medical examination, assignment of biologically diverse clinical groups and individualized treatment of the patients.
Conclusions
A routine tissue-typing laboratory should be flexible, providing valid and reproducible HLA data within short time period. NGS-based HLA analysis is performed with 100% reliability, and well fits the tasks of HLA typing in unrelated donors, in concordance with EFI and ASHI policies. This work process well corresponds to the working schedules for medium- and high-capacity laboratories, thus being potentially attractive to the donor registries.
Recently introduced next-generation sequencing techniques have a facilitating potential for the high-resolution genotyping via decrease of general uncertainty, like as due to extended sequencing regions. The entire set of MHS genes could be sequenced by means of these technologies, in order to uncover the latent immune determinants relevant to autoaggresive reactions [7, 25]. In near future, the whole exome sequencing may be considered a method of decoding some genetic variants, influencing HSCT outcomes. The NGS approaches will be a quite effective and cost-effective technology when evaluating histocompatibility parameters and immunogenetic interactions.
Conflict of interests
No conflict of interests is declared.
References
- Bentley G, Higuchi R, Hoglund B, Goodridge D, Sayer D, Trachtenberg EA, Erlich HA: High-resolution, high-throughput HLA genotyping by nextgeneration sequencing. Tissue Antigens 2009; 74: 393–403.
- Blazar BR, Murphy WJ, Abedi M: Advances in graftversus- host disease biology and therapy. Nat Rev Immunol 2012; 12: 443–458.
- Brunstein CG, Eapen M, Ahn KW, Appelbaum FR, Ballen KK, Champlin RE, Cutler C, Kan F, Laughlin MJ, Soiffer RJ, Weisdorf DJ, Woolfrey A, Wagner JE: Reduced-intensity conditioning transplantation in acute leukemia: the effect of source of unrelated donor stem cells on outcomes. Blood 2012; 119: 5591–5598.
- Danzer M, Polin H, Proll J, Hofer K, Fae I, Fischer GF, Gabriel C: Highthroughput sequence-based typing strategy for HLA-DRB1 based on real-time polymerase chain reaction. Hum Immunol 2007; 68: 915–917.
- Danzer M, Niklas N, Stabentheiner S, Hofer K, Pröll J, Stückler C, Raml E, Polinand H, Gabriel C. Rapid, scalable and highly automated HLA genotyping using next-generation sequencing: a transition from research to diagnostics. BMC Genomics 2013, 14: 221.
- Dunn PP: Human leucocyte antigen typing: techniques and technology, a critical appraisal. Int J Immunogenet 2011; 38: 463–473.
- Elsner HA, Blasczyk R: Immunogenetics of HLA null alleles: implications for blood stem cell transplantation. Tissue Antigens 2004; 64: 687–695.
- Erlich H: HLA DNA typing: past, present, and future. Tissue Antigens 2012; 80: 1–11.
- Erlich RL, Jia X, Anderson S, Banks E, Gao X, Carrington M, Gupta N, DePristo MA, Henn MR, Lennon NJ, de Bakker PI: Next-generation sequencing for HLA typing of class I loci. BMC Genomics 2011; 12: 42.
- Fleischhauer K, Shaw BE, Gooley T, Malkki M, Bardy P, Bignon JD, Dubois V, Horowitz MM, Madrigal JA, Morishima Y, Oudshoorn M, Ringden O, Spellman S, Velardi A, Zino E, Petersdorf EW: Effect of T-cell-epitope matching at HLA-DPB1 in recipients of unrelated-donor haemopoieticcell transplantation: a retrospective study. Lancet Oncol 2012; 13: 366–374.
- Gabriel C, Danzer M, Hackl C, Kopal G, Hufnagl P, Hofer K, Polin H, Stabentheiner S, Proll J: Rapid high-throughput human leukocyte antigen typing by massively parallel pyrosequencing for high-resolution allele identification. Hum Immunol; 2009, 70: 960–964.
- Harismendy O, Frazer K: Method for improving sequence coverage uniformity of targeted genomic intervals amplified by LR-PCR using Illumina GA sequencing-by-synthesis technology. Biotechniques 2009;46:229–231.
- Holcomb CL, Hoglund B, Anderson MW, Blake LA, Bohme I, Egholm M, Ferriola D, Gabriel C, Gelber SE, Goodridge D, Hawbecker S, Klein R, Ladner M, Lind C, Monos D, Pando MJ, Proll J, Sayer DC, Schmitz-Agheguian G, Simen BB, Thiele B, Trachtenberg EA, Tyan DB, Wassmuth R, White S, Erlich HA: A multi-site study using high-resolution HLA genotyping by next generation sequencing. Tissue Antigens 2011, 77: 206–217.
- Huse SM, Huber JA, Morrison HG, Sogin ML, Welch DM: Accuracy and quality of massively parallel DNA pyrosequencing. Genome Biol 2007; 8: R143.
- Kotsch K, Wehling J, Blasczyk R: Sequencing of HLA class II genes based on the conserved diversity of the non-coding regions: sequencing based typing of HLA-DRB genes. Tissue Antigens 1999; 53: 486–497.
- Lank SM, Golbach BA, Creager HM, Wiseman RW, Keskin DB, Reinherz EL, Brusic V, O’Connor DH: Ultra-high resolution HLA genotyping and allele discovery by highly multiplexed cDNA amplicon pyrosequencing. BMC Genomics 2012; 13: 378.
- Lank SM, Wiseman RW, Dudley DM, O’Connor DH: A novel single cDNA amplicon pyrosequencing method for high-throughput, cost-effective sequence-based HLA class I genotyping. Hum Immunol 2010; 71: 1011–1017.
- Lebedeva TV, Mastromarino SA, Lee E, Ohashi M, Alosco SM, Yu N: Resolution of HLA class I sequence-based typing ambiguities by groupspecific sequencing primers. Tissue Antigens 2011, 77: 247–250.
- Baxter-Lowe LA. Tailoring NGS for smaller volume labs. 42nd ASHI Annual Meeting. St. Louis, MO, Poster report (Sept 28, 2016).
- Li HW, Sykes M: Emerging concepts in haematopoietic cell transplantation. Nat Rev Immunol 2012; 12: 403–416.
- Lind C, Ferriola D, Mackiewicz K, Heron S, Rogers M, Slavich L, Walker R, Hsiao T, McLaughlin L, D’Arcy M, Gai X, Goodridge D, Sayer D, Monos D: Next-generation sequencing: the solution for high-resolution, unambiguous human leukocyte antigen typing. Hum Immunol 2010; 71: 1033–1042.
- McDevitt SL, Bredeson JV, Roy SW, Lane JA, Noble JA. HAPCAD: An open-source tool to detect PCR crossovers in next-generation sequencing generated HLA data. Hum Immunol. 2016; 77 (3): 257-63. doi: 10.1016/j.humimm. 2016.01.013.
- Metzker ML: Sequencing technologies – the next generation. Nat Rev Genet 2010; 11: 31–46.
- Petersdorf EW: Optimal HLA matching in hematopoietic cell transplantation. Curr Opin Immunol 2008; 20: 588–593.
- Proll J, Danzer M, Stabentheiner S, Niklas N, Hackl C, Hofer K, Atzmuller S, Hufnagl P, Gully C, Hauser H, Krieger O, Gabriel C: Sequence capture and next generation resequencing of the MHC region highlights potential transplantation determinants in HLA identical haematopoietic stem cell transplantation. DNA Res 2011; 18: 201–210.
- Robinson J, Halliwell JA, Hayhurst JD, Flicek P, Parham P, Marsh SG. The IPD and IMGT/HLA database: allele variant databases. Nucleic Acids Res 2015; 43(Database issue): D423-31. doi: 10.1093/nar/gku1161.
- Shaw BE, Arguello R, Garcia-Sepulveda CA, Madrigal JA: The impact of HLA genotyping on survival following unrelated donor haematopoietic stem cell transplantation. Br J Haematol 2010; 150: 251–258.
- Shiina T, Suzuki S, Ozaki Y, Taira H, Kikkawa E, Shigenari A, Oka A, UmemuraT, Joshita S, Takahashi O, Hayashi Y, Paumen M, Katsuyama Y, Mitsunaga S, Ota M, Kulski JK, Inoko H: Super high resolution for single molecule sequence-based typing of classical HLA loci at the 8-digit level using next generation sequencers. Tissue Antigens 2012; 80: 305–316.
- Marsh SG, WHO Nomenclature Committee for Factors of the HLA System. Nomenclature for factors of the HLA system, update March 2016. Human Immunology 2016; 77 (8): 706-709.
- Voorter CE, Mulkers E, Liebelt P, Sleyster E, van den Berg-Loonen EM: Reanalysis of sequence-based HLA-A, -B and -Cw typings: how ambiguous is today’s SBT typing tomorrow. Tissue Antigens 2007; 70: 383–389.
- Voelkerding KV, Dames SA, Durtschi JD. Next-generation sequencing: from basic research to diagnostics. Clin Chem 2009; 55: 641–658.
- Wang C, Krishnakumar S, Wilhelmy J, Babrzadeh F, Stepanyan L, Su LF, Levinson D, Fernandez-Vina MA, Davis RW, Davis MM, Mindrinos M: Highthroughput, high-fidelity HLA genotyping with deep sequencing. Proc Natl Acad Sci USA 2012; 109: 8676–8681.
- Weimer ET, Montgomery M, Petraroia R, Crawford J, Schmitz JL. Performance characteristics and validation of next-generation sequencing for human leucocyte antigen typing. J Mol Diagnostics 2016; 18 (5): 668-675.
" ["DETAIL_TEXT_TYPE"]=> string(4) "html" ["~DETAIL_TEXT_TYPE"]=> string(4) "html" ["PREVIEW_TEXT"]=> string(0) "" ["~PREVIEW_TEXT"]=> string(0) "" ["PREVIEW_TEXT_TYPE"]=> string(4) "text" ["~PREVIEW_TEXT_TYPE"]=> string(4) "text" ["PREVIEW_PICTURE"]=> NULL ["~PREVIEW_PICTURE"]=> NULL ["LANG_DIR"]=> string(4) "/ru/" ["~LANG_DIR"]=> string(4) "/ru/" ["SORT"]=> string(3) "500" ["~SORT"]=> string(3) "500" ["CODE"]=> string(100) "sovremennye-metody-i-vozmozhnosti-sekvenirovaniya-sleduyushchego-pokoleniya-ngs-dlya-tipirovaniya-si" ["~CODE"]=> string(100) "sovremennye-metody-i-vozmozhnosti-sekvenirovaniya-sleduyushchego-pokoleniya-ngs-dlya-tipirovaniya-si" ["EXTERNAL_ID"]=> string(3) "694" ["~EXTERNAL_ID"]=> string(3) "694" ["IBLOCK_TYPE_ID"]=> string(7) "journal" ["~IBLOCK_TYPE_ID"]=> string(7) "journal" ["IBLOCK_CODE"]=> string(7) "volumes" ["~IBLOCK_CODE"]=> string(7) "volumes" ["IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["~IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["LID"]=> string(2) "s2" ["~LID"]=> string(2) "s2" ["EDIT_LINK"]=> NULL ["DELETE_LINK"]=> NULL ["DISPLAY_ACTIVE_FROM"]=> string(0) "" ["IPROPERTY_VALUES"]=> array(18) { ["ELEMENT_META_TITLE"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["ELEMENT_META_KEYWORDS"]=> string(260) "Секвенирование следуюшего поколения (NGS) технологические решения HLA-типирование аллогенная трансплантация гемопоэтических стволовых клеток" ["ELEMENT_META_DESCRIPTION"]=> string(269) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLACurrent methods and opportunities of next-generation sequencing (NGS) for HLA typing" ["ELEMENT_PREVIEW_PICTURE_FILE_ALT"]=> string(3546) "Область генома, кодирующая лейкоциты антигенов человека (HLA) – это сегмент длиной 3,6 миллиона пар оснований на хромосоме 6(p21). Сложная по антигенному составу система HLA содержит важные молекулы, участвующие в трансплантационном иммунитете. Специфические генные последовательности генов HLA довольно сложно расшифровать с помощью классических методов прямого секвенирования в связи с их техническими ограничениями, сложным составом генов HLA, и возрастающим числом новых аллелей, что требует новых методов генотипирования. Секвенирование следуюшего поколения (NGS) является методом, который может обеспечить полное решение проблемы HLA-типирования. За последнее десятилетие разработка NGS обеспечила более легкий способ полногеномного анализа у человека, в том числе для типирования генов HLA. Несколько вариантов высокопроизводительных методов оценки HLA, основанные на мультиплексных аналитических технологиях NGS были разработаны на основе различных технологических платформ. В этом обзоре мы обсуждаем возможные области применения и прогресс в оборудовании для NGS в плане глубокого типирования системы HLA, с особым вниманием к будущим аспектам его клинического использования. Общеизвестный полиморфизм генов HLA и минимальные межаллельные различия представляют собой особую проблему. Поэтому некоторые вопросы, связанные с биоинформатикой и углубленной обработкой данных для более эффективного анализа HLA в контексте оценки совместимости донора и реципиента. В этом плане различными производителями предложены специализированные компьютерные программы для анализа больших баз данных, получаемых посредством технологий NGS. Наконец, эти высокопроизводительные подходы позволяют минимизировать финансовые затраты на 1 образец, особенно в крупных HLA-лабораториях, где соответствующие расходы на NGS-исследования стали теперь значительно затрат, возникающих при использовании классических технологий секвенирования по Сэнгеру." ["ELEMENT_PREVIEW_PICTURE_FILE_TITLE"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["ELEMENT_DETAIL_PICTURE_FILE_ALT"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["ELEMENT_DETAIL_PICTURE_FILE_TITLE"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["SECTION_META_TITLE"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["SECTION_META_KEYWORDS"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["SECTION_META_DESCRIPTION"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["SECTION_PICTURE_FILE_ALT"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["SECTION_PICTURE_FILE_TITLE"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["SECTION_PICTURE_FILE_NAME"]=> string(100) "sovremennye-metody-i-vozmozhnosti-sekvenirovaniya-sleduyushchego-pokoleniya-ngs-dlya-tipirovaniya-si" ["SECTION_DETAIL_PICTURE_FILE_ALT"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["SECTION_DETAIL_PICTURE_FILE_TITLE"]=> string(185) "Современные методы и возможности секвенирования следующего поколения (NGS) для типирования системы HLA" ["SECTION_DETAIL_PICTURE_FILE_NAME"]=> string(100) "sovremennye-metody-i-vozmozhnosti-sekvenirovaniya-sleduyushchego-pokoleniya-ngs-dlya-tipirovaniya-si" ["ELEMENT_PREVIEW_PICTURE_FILE_NAME"]=> string(100) "sovremennye-metody-i-vozmozhnosti-sekvenirovaniya-sleduyushchego-pokoleniya-ngs-dlya-tipirovaniya-si" ["ELEMENT_DETAIL_PICTURE_FILE_NAME"]=> string(100) "sovremennye-metody-i-vozmozhnosti-sekvenirovaniya-sleduyushchego-pokoleniya-ngs-dlya-tipirovaniya-si" } ["FIELDS"]=> array(1) { ["IBLOCK_SECTION_ID"]=> string(2) "21" } ["PROPERTIES"]=> array(18) { ["KEYWORDS"]=> array(36) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(4) { [0]=> string(4) "8467" [1]=> string(4) "8468" [2]=> string(4) "8469" [3]=> string(4) "8470" } ["VALUE"]=> array(4) { [0]=> string(3) "692" [1]=> string(3) "693" [2]=> string(3) "607" [3]=> string(3) "282" } ["DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(4) { [0]=> string(3) "692" [1]=> string(3) "693" [2]=> string(3) "607" [3]=> string(3) "282" } ["~DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" } ["SUBMITTED"]=> array(36) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8414" ["VALUE"]=> string(22) "09/14/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "09/14/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL } ["ACCEPTED"]=> array(36) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8415" ["VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL } ["PUBLISHED"]=> array(36) { ["ID"]=> string(2) "22" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Дата публикации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "PUBLISHED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "22" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Дата публикации" ["~DEFAULT_VALUE"]=> NULL } ["CONTACT"]=> array(36) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8416" ["VALUE"]=> string(3) "691" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "691" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHORS"]=> array(36) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(1) { [0]=> string(4) "8471" } ["VALUE"]=> array(1) { [0]=> string(3) "691" } ["DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(1) { [0]=> string(3) "691" } ["~DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_RU"]=> array(36) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8418" ["VALUE"]=> array(2) { ["TEXT"]=> string(213) "<sup>1</sup>Юрий А. Серов, <sup>1</sup>Ильдар M. Бархатов, <sup>1</sup>Антон С. Климов, <sup>2</sup>Андрей С. Беркос" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(165) "1Юрий А. Серов, 1Ильдар M. Бархатов, 1Антон С. Климов, 2Андрей С. Беркос" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_RU"]=> array(36) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8419" ["VALUE"]=> array(2) { ["TEXT"]=> string(545) "<sup>1</sup>НИИ детской онкологии, гематологии и трансплантации им. Р. М. Горбачевой, Первый Санкт-Петербургский<br> государственный медицинский университет им. И. П. Павлова, Санкт-Петебург, Россия;<br> <sup>2</sup>Российский НИИ гематологии и трансфузиологии, Санкт-Петербург, Россия<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(503) "1НИИ детской онкологии, гематологии и трансплантации им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. И. П. Павлова, Санкт-Петебург, Россия;
2Российский НИИ гематологии и трансфузиологии, Санкт-Петербург, Россия
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_RU"]=> array(36) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8420" ["VALUE"]=> array(2) { ["TEXT"]=> string(3546) "Область генома, кодирующая лейкоциты антигенов человека (HLA) – это сегмент длиной 3,6 миллиона пар оснований на хромосоме 6(p21). Сложная по антигенному составу система HLA содержит важные молекулы, участвующие в трансплантационном иммунитете. Специфические генные последовательности генов HLA довольно сложно расшифровать с помощью классических методов прямого секвенирования в связи с их техническими ограничениями, сложным составом генов HLA, и возрастающим числом новых аллелей, что требует новых методов генотипирования. Секвенирование следуюшего поколения (NGS) является методом, который может обеспечить полное решение проблемы HLA-типирования. За последнее десятилетие разработка NGS обеспечила более легкий способ полногеномного анализа у человека, в том числе для типирования генов HLA. Несколько вариантов высокопроизводительных методов оценки HLA, основанные на мультиплексных аналитических технологиях NGS были разработаны на основе различных технологических платформ. В этом обзоре мы обсуждаем возможные области применения и прогресс в оборудовании для NGS в плане глубокого типирования системы HLA, с особым вниманием к будущим аспектам его клинического использования. Общеизвестный полиморфизм генов HLA и минимальные межаллельные различия представляют собой особую проблему. Поэтому некоторые вопросы, связанные с биоинформатикой и углубленной обработкой данных для более эффективного анализа HLA в контексте оценки совместимости донора и реципиента. В этом плане различными производителями предложены специализированные компьютерные программы для анализа больших баз данных, получаемых посредством технологий NGS. Наконец, эти высокопроизводительные подходы позволяют минимизировать финансовые затраты на 1 образец, особенно в крупных HLA-лабораториях, где соответствующие расходы на NGS-исследования стали теперь значительно затрат, возникающих при использовании классических технологий секвенирования по Сэнгеру." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(3546) "Область генома, кодирующая лейкоциты антигенов человека (HLA) – это сегмент длиной 3,6 миллиона пар оснований на хромосоме 6(p21). Сложная по антигенному составу система HLA содержит важные молекулы, участвующие в трансплантационном иммунитете. Специфические генные последовательности генов HLA довольно сложно расшифровать с помощью классических методов прямого секвенирования в связи с их техническими ограничениями, сложным составом генов HLA, и возрастающим числом новых аллелей, что требует новых методов генотипирования. Секвенирование следуюшего поколения (NGS) является методом, который может обеспечить полное решение проблемы HLA-типирования. За последнее десятилетие разработка NGS обеспечила более легкий способ полногеномного анализа у человека, в том числе для типирования генов HLA. Несколько вариантов высокопроизводительных методов оценки HLA, основанные на мультиплексных аналитических технологиях NGS были разработаны на основе различных технологических платформ. В этом обзоре мы обсуждаем возможные области применения и прогресс в оборудовании для NGS в плане глубокого типирования системы HLA, с особым вниманием к будущим аспектам его клинического использования. Общеизвестный полиморфизм генов HLA и минимальные межаллельные различия представляют собой особую проблему. Поэтому некоторые вопросы, связанные с биоинформатикой и углубленной обработкой данных для более эффективного анализа HLA в контексте оценки совместимости донора и реципиента. В этом плане различными производителями предложены специализированные компьютерные программы для анализа больших баз данных, получаемых посредством технологий NGS. Наконец, эти высокопроизводительные подходы позволяют минимизировать финансовые затраты на 1 образец, особенно в крупных HLA-лабораториях, где соответствующие расходы на NGS-исследования стали теперь значительно затрат, возникающих при использовании классических технологий секвенирования по Сэнгеру." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["DOI"]=> array(36) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8421" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-63-70" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-63-70" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_EN"]=> array(36) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8422" ["VALUE"]=> array(2) { ["TEXT"]=> string(165) "<sup>1</sup>Youri A. Serov, <sup>1</sup>Ildar M. Barkhatov, <sup>1</sup>Anton S. Klimov, <sup>2</sup>Andrey S. Berkos" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(117) "1Youri A. Serov, 1Ildar M. Barkhatov, 1Anton S. Klimov, 2Andrey S. Berkos" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_EN"]=> array(36) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8423" ["VALUE"]=> array(2) { ["TEXT"]=> string(783) "<sup>1</sup>Research Institute of Children Oncology, Hematology and Transplantation, The First State I. Pavlov Medical University,<br> St. Petersburg, Russia<br> <sup>2</sup>Russian Research Institute of Hematology and Transfusiology, Federal Medical-Biological Agency, St. Petersburg, Russia<br> <br> Youri A. Serov, MD, Ph. D., CHS(ABHI), The Head of Biobank<br> Research Unit, Raisa Gorbacheva Memorial Institute of Children’s<br> Hematology, Oncology and Transplantation, The First<br> St. Petersburg I. P. Pavlov State Medical University, Leo Tolstoy<br> str. 6-8, 197022, St. Petersburg, Russia<br> <br> Phone: +7(812) 338-6260 (office); +7(981) 973-8041 (mob.)<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(693) "1Research Institute of Children Oncology, Hematology and Transplantation, The First State I. Pavlov Medical University,
St. Petersburg, Russia
2Russian Research Institute of Hematology and Transfusiology, Federal Medical-Biological Agency, St. Petersburg, Russia
Youri A. Serov, MD, Ph. D., CHS(ABHI), The Head of Biobank
Research Unit, Raisa Gorbacheva Memorial Institute of Children’s
Hematology, Oncology and Transplantation, The First
St. Petersburg I. P. Pavlov State Medical University, Leo Tolstoy
str. 6-8, 197022, St. Petersburg, Russia
Phone: +7(812) 338-6260 (office); +7(981) 973-8041 (mob.)
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_EN"]=> array(36) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8424" ["VALUE"]=> array(2) { ["TEXT"]=> string(1211) "The region encoding human leukocyte antigens (HLA) is a 3.6-Mb segment at chromosome 6p21. A highly complex HLA system comprises important molecules involved in transplantation immunity, classical direct sequencing methods making it very complicating to resolve the genomic specificities of HLA genes, due of the complex nature of them. Current limitations of existing methods and the increasing rate of new alleles, strongly demanding for a new methodology approach for the HLA genotyping. Next-Generation Sequencing (NGS) is a method that can provide a complete solution to the HLA typing problem. Last decade, the development of (NGS) has shown the easy way for whole-genome analysis in individuals. HLA typing has also benefited from NGS technologies. Several high-throughput HLA- NGS-based typing methods were developed using different technological platforms. In this review, we summarize the possible applications and instrumentation developments of NGS in the in-depth HLA typing, with a focus on future clinical applications. However, some issues of bioinformatics and data mining remain an obstacle to more effective analysis of HLA analysis in the context of donor/recipient compatibility assessment." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(1211) "The region encoding human leukocyte antigens (HLA) is a 3.6-Mb segment at chromosome 6p21. A highly complex HLA system comprises important molecules involved in transplantation immunity, classical direct sequencing methods making it very complicating to resolve the genomic specificities of HLA genes, due of the complex nature of them. Current limitations of existing methods and the increasing rate of new alleles, strongly demanding for a new methodology approach for the HLA genotyping. Next-Generation Sequencing (NGS) is a method that can provide a complete solution to the HLA typing problem. Last decade, the development of (NGS) has shown the easy way for whole-genome analysis in individuals. HLA typing has also benefited from NGS technologies. Several high-throughput HLA- NGS-based typing methods were developed using different technological platforms. In this review, we summarize the possible applications and instrumentation developments of NGS in the in-depth HLA typing, with a focus on future clinical applications. However, some issues of bioinformatics and data mining remain an obstacle to more effective analysis of HLA analysis in the context of donor/recipient compatibility assessment." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["NAME_EN"]=> array(36) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8425" ["VALUE"]=> string(84) "Current methods and opportunities of next-generation sequencing (NGS) for HLA typing" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(84) "Current methods and opportunities of next-generation sequencing (NGS) for HLA typing" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" } ["FULL_TEXT_RU"]=> array(36) { ["ID"]=> string(2) "42" ["TIMESTAMP_X"]=> string(19) "2015-09-07 20:29:18" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(23) "Полный текст" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(12) "FULL_TEXT_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "42" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(23) "Полный текст" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["PDF_RU"]=> array(36) { ["ID"]=> string(2) "43" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF RUS" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_RU" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "43" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8472" ["VALUE"]=> string(3) "278" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "278" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(7) "PDF RUS" ["~DEFAULT_VALUE"]=> string(0) "" } ["PDF_EN"]=> array(36) { ["ID"]=> string(2) "44" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF ENG" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "44" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8473" ["VALUE"]=> string(3) "279" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "279" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(7) "PDF ENG" ["~DEFAULT_VALUE"]=> string(0) "" } ["NAME_LONG"]=> array(36) { ["ID"]=> string(2) "45" ["TIMESTAMP_X"]=> string(19) "2023-04-13 00:55:00" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(72) "Название (для очень длинных заголовков)" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "NAME_LONG" ["DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "45" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(80) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(72) "Название (для очень длинных заголовков)" ["~DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } } } ["DISPLAY_PROPERTIES"]=> array(13) { ["AUTHOR_EN"]=> array(37) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8422" ["VALUE"]=> array(2) { ["TEXT"]=> string(165) "<sup>1</sup>Youri A. Serov, <sup>1</sup>Ildar M. Barkhatov, <sup>1</sup>Anton S. Klimov, <sup>2</sup>Andrey S. Berkos" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(117) "1Youri A. Serov, 1Ildar M. Barkhatov, 1Anton S. Klimov, 2Andrey S. Berkos" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(117) "1Youri A. Serov, 1Ildar M. Barkhatov, 1Anton S. Klimov, 2Andrey S. Berkos" } ["SUMMARY_EN"]=> array(37) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8424" ["VALUE"]=> array(2) { ["TEXT"]=> string(1211) "The region encoding human leukocyte antigens (HLA) is a 3.6-Mb segment at chromosome 6p21. A highly complex HLA system comprises important molecules involved in transplantation immunity, classical direct sequencing methods making it very complicating to resolve the genomic specificities of HLA genes, due of the complex nature of them. Current limitations of existing methods and the increasing rate of new alleles, strongly demanding for a new methodology approach for the HLA genotyping. Next-Generation Sequencing (NGS) is a method that can provide a complete solution to the HLA typing problem. Last decade, the development of (NGS) has shown the easy way for whole-genome analysis in individuals. HLA typing has also benefited from NGS technologies. Several high-throughput HLA- NGS-based typing methods were developed using different technological platforms. In this review, we summarize the possible applications and instrumentation developments of NGS in the in-depth HLA typing, with a focus on future clinical applications. However, some issues of bioinformatics and data mining remain an obstacle to more effective analysis of HLA analysis in the context of donor/recipient compatibility assessment." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(1211) "The region encoding human leukocyte antigens (HLA) is a 3.6-Mb segment at chromosome 6p21. A highly complex HLA system comprises important molecules involved in transplantation immunity, classical direct sequencing methods making it very complicating to resolve the genomic specificities of HLA genes, due of the complex nature of them. Current limitations of existing methods and the increasing rate of new alleles, strongly demanding for a new methodology approach for the HLA genotyping. Next-Generation Sequencing (NGS) is a method that can provide a complete solution to the HLA typing problem. Last decade, the development of (NGS) has shown the easy way for whole-genome analysis in individuals. HLA typing has also benefited from NGS technologies. Several high-throughput HLA- NGS-based typing methods were developed using different technological platforms. In this review, we summarize the possible applications and instrumentation developments of NGS in the in-depth HLA typing, with a focus on future clinical applications. However, some issues of bioinformatics and data mining remain an obstacle to more effective analysis of HLA analysis in the context of donor/recipient compatibility assessment." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(1211) "The region encoding human leukocyte antigens (HLA) is a 3.6-Mb segment at chromosome 6p21. A highly complex HLA system comprises important molecules involved in transplantation immunity, classical direct sequencing methods making it very complicating to resolve the genomic specificities of HLA genes, due of the complex nature of them. Current limitations of existing methods and the increasing rate of new alleles, strongly demanding for a new methodology approach for the HLA genotyping. Next-Generation Sequencing (NGS) is a method that can provide a complete solution to the HLA typing problem. Last decade, the development of (NGS) has shown the easy way for whole-genome analysis in individuals. HLA typing has also benefited from NGS technologies. Several high-throughput HLA- NGS-based typing methods were developed using different technological platforms. In this review, we summarize the possible applications and instrumentation developments of NGS in the in-depth HLA typing, with a focus on future clinical applications. However, some issues of bioinformatics and data mining remain an obstacle to more effective analysis of HLA analysis in the context of donor/recipient compatibility assessment." } ["DOI"]=> array(37) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8421" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-63-70" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-63-70" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-63-70" } ["NAME_EN"]=> array(37) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8425" ["VALUE"]=> string(84) "Current methods and opportunities of next-generation sequencing (NGS) for HLA typing" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(84) "Current methods and opportunities of next-generation sequencing (NGS) for HLA typing" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(84) "Current methods and opportunities of next-generation sequencing (NGS) for HLA typing" } ["ORGANIZATION_EN"]=> array(37) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8423" ["VALUE"]=> array(2) { ["TEXT"]=> string(783) "<sup>1</sup>Research Institute of Children Oncology, Hematology and Transplantation, The First State I. Pavlov Medical University,<br> St. Petersburg, Russia<br> <sup>2</sup>Russian Research Institute of Hematology and Transfusiology, Federal Medical-Biological Agency, St. Petersburg, Russia<br> <br> Youri A. Serov, MD, Ph. D., CHS(ABHI), The Head of Biobank<br> Research Unit, Raisa Gorbacheva Memorial Institute of Children’s<br> Hematology, Oncology and Transplantation, The First<br> St. Petersburg I. P. Pavlov State Medical University, Leo Tolstoy<br> str. 6-8, 197022, St. Petersburg, Russia<br> <br> Phone: +7(812) 338-6260 (office); +7(981) 973-8041 (mob.)<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(693) "1Research Institute of Children Oncology, Hematology and Transplantation, The First State I. Pavlov Medical University,
St. Petersburg, Russia
2Russian Research Institute of Hematology and Transfusiology, Federal Medical-Biological Agency, St. Petersburg, Russia
Youri A. Serov, MD, Ph. D., CHS(ABHI), The Head of Biobank
Research Unit, Raisa Gorbacheva Memorial Institute of Children’s
Hematology, Oncology and Transplantation, The First
St. Petersburg I. P. Pavlov State Medical University, Leo Tolstoy
str. 6-8, 197022, St. Petersburg, Russia
Phone: +7(812) 338-6260 (office); +7(981) 973-8041 (mob.)
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(693) "1Research Institute of Children Oncology, Hematology and Transplantation, The First State I. Pavlov Medical University,
St. Petersburg, Russia
2Russian Research Institute of Hematology and Transfusiology, Federal Medical-Biological Agency, St. Petersburg, Russia
Youri A. Serov, MD, Ph. D., CHS(ABHI), The Head of Biobank
Research Unit, Raisa Gorbacheva Memorial Institute of Children’s
Hematology, Oncology and Transplantation, The First
St. Petersburg I. P. Pavlov State Medical University, Leo Tolstoy
str. 6-8, 197022, St. Petersburg, Russia
Phone: +7(812) 338-6260 (office); +7(981) 973-8041 (mob.)
" } ["AUTHORS"]=> array(38) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(1) { [0]=> string(4) "8471" } ["VALUE"]=> array(1) { [0]=> string(3) "691" } ["DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(1) { [0]=> string(3) "691" } ["~DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(57) "Youri A. Serov" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["AUTHOR_RU"]=> array(37) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8418" ["VALUE"]=> array(2) { ["TEXT"]=> string(213) "<sup>1</sup>Юрий А. Серов, <sup>1</sup>Ильдар M. Бархатов, <sup>1</sup>Антон С. Климов, <sup>2</sup>Андрей С. Беркос" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(165) "1Юрий А. Серов, 1Ильдар M. Бархатов, 1Антон С. Климов, 2Андрей С. Беркос" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(165) "1Юрий А. Серов, 1Ильдар M. Бархатов, 1Антон С. Климов, 2Андрей С. Беркос" } ["SUBMITTED"]=> array(37) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8414" ["VALUE"]=> string(22) "09/14/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "09/14/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "09/14/2016 12:00:00 am" } ["ACCEPTED"]=> array(37) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8415" ["VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "11/25/2016 12:00:00 am" } ["KEYWORDS"]=> array(38) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(4) { [0]=> string(4) "8467" [1]=> string(4) "8468" [2]=> string(4) "8469" [3]=> string(4) "8470" } ["VALUE"]=> array(4) { [0]=> string(3) "692" [1]=> string(3) "693" [2]=> string(3) "607" [3]=> string(3) "282" } ["DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(4) { [0]=> string(3) "692" [1]=> string(3) "693" [2]=> string(3) "607" [3]=> string(3) "282" } ["~DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(4) { [0]=> string(118) "Секвенирование следуюшего поколения (NGS)" [1]=> string(89) "технологические решения" [2]=> string(70) "HLA-типирование" [3]=> string(156) "аллогенная трансплантация гемопоэтических стволовых клеток" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["CONTACT"]=> array(38) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8416" ["VALUE"]=> string(3) "691" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "691" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(57) "Youri A. Serov" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["SUMMARY_RU"]=> array(37) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8420" ["VALUE"]=> array(2) { ["TEXT"]=> string(3546) "Область генома, кодирующая лейкоциты антигенов человека (HLA) – это сегмент длиной 3,6 миллиона пар оснований на хромосоме 6(p21). Сложная по антигенному составу система HLA содержит важные молекулы, участвующие в трансплантационном иммунитете. Специфические генные последовательности генов HLA довольно сложно расшифровать с помощью классических методов прямого секвенирования в связи с их техническими ограничениями, сложным составом генов HLA, и возрастающим числом новых аллелей, что требует новых методов генотипирования. Секвенирование следуюшего поколения (NGS) является методом, который может обеспечить полное решение проблемы HLA-типирования. За последнее десятилетие разработка NGS обеспечила более легкий способ полногеномного анализа у человека, в том числе для типирования генов HLA. Несколько вариантов высокопроизводительных методов оценки HLA, основанные на мультиплексных аналитических технологиях NGS были разработаны на основе различных технологических платформ. В этом обзоре мы обсуждаем возможные области применения и прогресс в оборудовании для NGS в плане глубокого типирования системы HLA, с особым вниманием к будущим аспектам его клинического использования. Общеизвестный полиморфизм генов HLA и минимальные межаллельные различия представляют собой особую проблему. Поэтому некоторые вопросы, связанные с биоинформатикой и углубленной обработкой данных для более эффективного анализа HLA в контексте оценки совместимости донора и реципиента. В этом плане различными производителями предложены специализированные компьютерные программы для анализа больших баз данных, получаемых посредством технологий NGS. Наконец, эти высокопроизводительные подходы позволяют минимизировать финансовые затраты на 1 образец, особенно в крупных HLA-лабораториях, где соответствующие расходы на NGS-исследования стали теперь значительно затрат, возникающих при использовании классических технологий секвенирования по Сэнгеру." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(3546) "Область генома, кодирующая лейкоциты антигенов человека (HLA) – это сегмент длиной 3,6 миллиона пар оснований на хромосоме 6(p21). Сложная по антигенному составу система HLA содержит важные молекулы, участвующие в трансплантационном иммунитете. Специфические генные последовательности генов HLA довольно сложно расшифровать с помощью классических методов прямого секвенирования в связи с их техническими ограничениями, сложным составом генов HLA, и возрастающим числом новых аллелей, что требует новых методов генотипирования. Секвенирование следуюшего поколения (NGS) является методом, который может обеспечить полное решение проблемы HLA-типирования. За последнее десятилетие разработка NGS обеспечила более легкий способ полногеномного анализа у человека, в том числе для типирования генов HLA. Несколько вариантов высокопроизводительных методов оценки HLA, основанные на мультиплексных аналитических технологиях NGS были разработаны на основе различных технологических платформ. В этом обзоре мы обсуждаем возможные области применения и прогресс в оборудовании для NGS в плане глубокого типирования системы HLA, с особым вниманием к будущим аспектам его клинического использования. Общеизвестный полиморфизм генов HLA и минимальные межаллельные различия представляют собой особую проблему. Поэтому некоторые вопросы, связанные с биоинформатикой и углубленной обработкой данных для более эффективного анализа HLA в контексте оценки совместимости донора и реципиента. В этом плане различными производителями предложены специализированные компьютерные программы для анализа больших баз данных, получаемых посредством технологий NGS. Наконец, эти высокопроизводительные подходы позволяют минимизировать финансовые затраты на 1 образец, особенно в крупных HLA-лабораториях, где соответствующие расходы на NGS-исследования стали теперь значительно затрат, возникающих при использовании классических технологий секвенирования по Сэнгеру." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(3546) "Область генома, кодирующая лейкоциты антигенов человека (HLA) – это сегмент длиной 3,6 миллиона пар оснований на хромосоме 6(p21). Сложная по антигенному составу система HLA содержит важные молекулы, участвующие в трансплантационном иммунитете. Специфические генные последовательности генов HLA довольно сложно расшифровать с помощью классических методов прямого секвенирования в связи с их техническими ограничениями, сложным составом генов HLA, и возрастающим числом новых аллелей, что требует новых методов генотипирования. Секвенирование следуюшего поколения (NGS) является методом, который может обеспечить полное решение проблемы HLA-типирования. За последнее десятилетие разработка NGS обеспечила более легкий способ полногеномного анализа у человека, в том числе для типирования генов HLA. Несколько вариантов высокопроизводительных методов оценки HLA, основанные на мультиплексных аналитических технологиях NGS были разработаны на основе различных технологических платформ. В этом обзоре мы обсуждаем возможные области применения и прогресс в оборудовании для NGS в плане глубокого типирования системы HLA, с особым вниманием к будущим аспектам его клинического использования. Общеизвестный полиморфизм генов HLA и минимальные межаллельные различия представляют собой особую проблему. Поэтому некоторые вопросы, связанные с биоинформатикой и углубленной обработкой данных для более эффективного анализа HLA в контексте оценки совместимости донора и реципиента. В этом плане различными производителями предложены специализированные компьютерные программы для анализа больших баз данных, получаемых посредством технологий NGS. Наконец, эти высокопроизводительные подходы позволяют минимизировать финансовые затраты на 1 образец, особенно в крупных HLA-лабораториях, где соответствующие расходы на NGS-исследования стали теперь значительно затрат, возникающих при использовании классических технологий секвенирования по Сэнгеру." } ["ORGANIZATION_RU"]=> array(37) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8419" ["VALUE"]=> array(2) { ["TEXT"]=> string(545) "<sup>1</sup>НИИ детской онкологии, гематологии и трансплантации им. Р. М. Горбачевой, Первый Санкт-Петербургский<br> государственный медицинский университет им. И. П. Павлова, Санкт-Петебург, Россия;<br> <sup>2</sup>Российский НИИ гематологии и трансфузиологии, Санкт-Петербург, Россия<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(503) "1НИИ детской онкологии, гематологии и трансплантации им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. И. П. Павлова, Санкт-Петебург, Россия;
2Российский НИИ гематологии и трансфузиологии, Санкт-Петербург, Россия
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(503) "1НИИ детской онкологии, гематологии и трансплантации им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. И. П. Павлова, Санкт-Петебург, Россия;
2Российский НИИ гематологии и трансфузиологии, Санкт-Петербург, Россия
" } } } [6]=> array(49) { ["IBLOCK_SECTION_ID"]=> string(2) "21" ["~IBLOCK_SECTION_ID"]=> string(2) "21" ["ID"]=> string(3) "685" ["~ID"]=> string(3) "685" ["IBLOCK_ID"]=> string(1) "2" ["~IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["~NAME"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["ACTIVE_FROM"]=> NULL ["~ACTIVE_FROM"]=> NULL ["TIMESTAMP_X"]=> string(22) "03/30/2017 07:32:49 pm" ["~TIMESTAMP_X"]=> string(22) "03/30/2017 07:32:49 pm" ["DETAIL_PAGE_URL"]=> string(161) "/en/archive/vypusk-5-nomer-4/eksperimentalnye-issledovaniya/gennaya-terapiya-lentivirusnymi-vektorami-v-gemo-poeticheskikh-stvolovykh-kletkakh-pri-vrozhdennykh-/" ["~DETAIL_PAGE_URL"]=> string(161) "/en/archive/vypusk-5-nomer-4/eksperimentalnye-issledovaniya/gennaya-terapiya-lentivirusnymi-vektorami-v-gemo-poeticheskikh-stvolovykh-kletkakh-pri-vrozhdennykh-/" ["LIST_PAGE_URL"]=> string(12) "/en/archive/" ["~LIST_PAGE_URL"]=> string(12) "/en/archive/" ["DETAIL_TEXT"]=> string(28175) "
Introduction
Rare diseases, of which 80% are inherited disorders, affect some 6% of the human population, amounting in Europe to around 30 million people. Since many result in chronic disability and cost-intensive care, the impact is disproportional and may well be over 20% of health care costs, although the current lack of registration, contrary to cancer, makes exact estimates difficult. Most of the approximately 7500 known inherited diseases lack a curative intervention, are managed by symptomatic treatment and, in case of a family history, by prenatal diagnosis. A small minority is included in newborn screening programs. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation, (iv) strict regulation of transgene product levels is not required, (v) the transgene produces levels sufficient for sustained alleviation of symptoms or cure, and (vi) adverse immune responses to the transgene product are not expected, do not interfere with efficacy, or can be successfully counteracted or circumvented. Many of the more than 100 primary immune deficiencies and around 40 lysosomal storage disorders meet those criteria.
This brief review covers development and prospect of gene therapy for inherited immune deficiencies and lysosomal storage disorders, extending an earlier review [45].
Initial development of hematopoietic stem cell gene therapy
Hematopoietic stem cell gene therapy has been developed over more than 20 years. The pioneering trials for X-linked severe combined immune deficiency (SCID) using gammaretroviral gene transfer vectors resulted in successful restoration of T cell immunity [17, 22] in 18 patients and in long-term survival for 17 patients out of 20, a survival rate similar to HLA-identical BM transplantation [35]. Unfortunately, in 5 patients autonomous T cell clones developed into leukemia, among which 1 patient did not survive. In the context of European collaborative projects, the pathogenesis was rapidly elucidated, resulting in a series of publications on mechanisms involved in gammaretroviral mutagenesis and oncogenesis [3,12,13,28,31,32,37]. Briefly, gammaretroviral vectors generally integrate near the transcription start sites of expressed genes with a preference for proto-oncogenes, which results in aberrant expression driven by the promoter/ enhancer of the therapeutic transgene and may result in a preleukemic state. It is not excluded that the phenotypes of the treated diseases co-predispose to leukemia development [38], given the absence of leukemia in the ADA-SCID trial [1, 4], a 25% incidence in the X-linked SCID trials and an over 75% incidence in a gammaretroviral Wiskott-Aldrich trial [6].
Development of lentiviral vector gene therapy in inherited storage disorders
The gammaretroviral vectors have been replaced by HIV-1 derived lentiviral vectors [29,36,48], which lack the propensity for integration near proto-oncogenes and have the added advantage of integrating into quiescent cells, such as longterm repopulating stem cells. In addition, third generation lentiviral vectors made self-inactivating (SIN) by deletion of enhancer regions from the long terminal repeat sequences reduce the risk of influencing nearby genes, resulting in favorable safety profiles [8]. Systematic disease specific efficacy and safety evaluations, including codon optimization and careful promoter selection have enabled initial clinical trials using these vectors for selected metabolic storage disorders (Table 1), which include adrenoleukodystrophy (ALD), metachromatic leukodystrophy (MLD), and Hurler (MPS I), Pompe (GSD II) and Fabry diseases. We have focused on Pompe disease, the only disorder so far developed for stem cell gene therapy [42] in which allogeneic stem cell transplantation has not been applied due to lack of enzyme expression in the hematopoietic system [25] and in addition studied stem cell gene therapy for Hurler syndrome.
Hurler Syndrome and Pompe disease
Hurler Syndrome (Mucopolysaccharidosis type I, OMIM # 252800) is a lethal autosomal recessive storage disorder caused by a deficiency of the lysosomal enzyme α-L-iduronidase (IDUA; EC 3.2.1.76). The deficiency leads to insufficient degradation of glycoaminoglycans (GAG’s’) that interferes with normal cellular function and causes a multisystem disorder affecting the CNS, liver, skeleton, lungs and sensory organs (corneal clouding and deafness), average expected life-span approximately 5-10 years. Treatment consists of alloSCT, currently the only therapeutic option to establish long-term survival and protection of the CNS. Enzyme replacement has a systemic effect but does not reach the brain or the skeletal bones. AlloSCT does not improve the skeleton pathology either. Although clinical outcome of SCT has improved by the use of umbilical cord stem cells, progressive bone disease persists, leading to severe handicaps. An international long term clinical follow-up cohort of Hurler patients treated with alloSCT including >80% of the patients successfully transplanted worldwide [2] showed that certain genotypes, age at HSCT, and poor performance at HSCT are predictors for poor skeletal and neurodevelopmental outcome. These findings warrant the development of a single curative gene therapy approach.
Pompe disease (glycogen storage disease type II, acid maltase deficiency, OMIM # 232300) is a rare autosomal recessive lysosomal storage disorder caused by mutations in the gene-encoding acid α-glucosidase (EC 3.2.1.20) [40]. Severe mutations cause complete enzyme deficiency, resulting in the classic infantile form of Pompe disease, which was first
described by the Dutch pathologist J.C.Pompe [33]. Symptoms are caused by glycogen accumulation, mainly in skeletal, cardiac and smooth muscle, but also in other tissues, including the central and peripheral nervous system. In the first months of life, patients present with progressive muscle weakness, hypertrophic cardiomyopathy, respiratory problems and feeding difficulties. If untreated, this leads to death before the age of one year [39]. Older children and adults may have up to 20-30% residual enzyme activity and show a more slowly progressive phenotype. The symptoms generally result from weakness of the (proximal) skeletal muscles. These patients eventually become wheelchair bound and ventilator dependent in late childhood or adulthood.
Enzyme replacement therapy (ERT) by administration of recombinant acid α-glucosidase [16,40] (Myozyme®) is currently the only effective treatment, requiring high dose biweekly administration. Although of considerable benefit to many patients, ERT is not curative, requires life-long administration, may result in immune responses to the recombinant enzyme [41] and, partly due to the high doses required for clinical efficacy, the costs are extremely high. Therefore, a corrective intervention with curative intent represents an unmet medical need.
Efficacy and safety evaluation of lentiviral vector gene therapy in the Hurler and Pompe mouse models
The natural course of Hurler’s disease is invalidating and lethal, and the drawbacks of the current therapeutic modalities justify a gene therapy approach. An IDUA knock-out mouse model has been developed suitable to study gene therapeutic approaches and made available to our research. A gene therapy study with HSC in the IDUA KO mice showed improvement of disease pathology, including the brain, but bone remodeling was not reported [14]. Using a codon optimized IDUA gene driven by a cellular promoter in a lentiviral vector to transduce hematopoietic stem cells, we demonstrated long-term expression in the hematopoietic system resulting in full correction of the Hurler phenotype (unpublished) without any severe adverse effects. Similar results and a further safety analysis were obtained in a parallel study [43,44], completing the preclinical analysis enabling an initial clinical trial in selected patients.
In the initial evaluation of lentiviral stem cell gene therapy for Pompe disease using an efficient overnight transduction protocol [42], we demonstrated that approximately 30% successfully transduced cells present in the bone marrow after sublethal total body irradiation as conditioning for transplantation resulted in high levels of α-glucosidase. Restoration of α-glucosidase activity in target tissues by uptake through the mannose-6-phosphate receptor reduced glycogen storage proportional to the enzyme levels achieved, with full correction of glycogen storage in liver and spleen, correction of the life-threatening cardiomyopathy, significantly improved respiration and improved, but not fully normalized skeletal muscle function. Of particular interest was the demonstration of robust immune tolerance to the recombinant transgene product. In the follow-up study (manuscript in preparation), codon-optimization of the therapeutic transgene resulted in full correction of the phenotype including skeletal muscles. Remarkably, although Pompe disease does not result in mental retardation or other neuronal problems, also brain glycogen levels normalized entirely, with all astrocytes, which play a key role in glycogen storage and glycogenolysis in the brain, showing active acid α-glucosidase activity. Apparently the microglia descendants of hematopoietic stem cells, which are capable of passing the blood-brain-barrier, as we originally demonstrated in the mouse model of Krabbe disease [24], provide sufficient acid α-glucosidase to normalize glycogen levels also in neuronal tissue. Up till now, hematopoietic stem cell gene therapy is the only approach to achieve both robust immune tolerance to the transgene product and efficacy in bypassing the blood/ brain barrier, as has also been observed by others [43].
Clinical implementation of stem cell gene therapy in primary immune deficiencies
A summary of the current developments is provided in Table 2. Briefly, successful clinical trials are ongoing for X-linked SCID, ADA-SCID and Wiskott-Aldrich syndrome, while
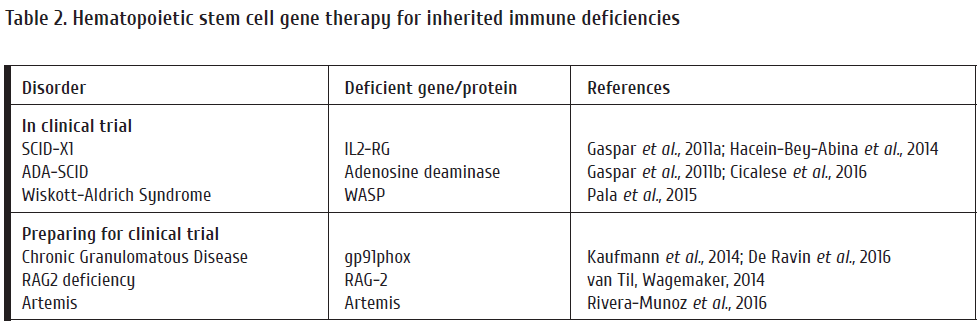
clinical trials are being prepared for RAG2 deficiency, chronic granulomatous disease and Artemis. Gene therapy for ADA-SCID, which is difficult to treat with allogeneic stem cell transplantation, has recently been registered as an advanced therapy medicinal product and will soon be considered as the standard treatment for this disease.
Further developments
The future development of stem cell gene therapy efficacy and safety would obviously benefit considerably from non-cytoreductive preparation of the patients to enable engraftment of the gene-corrected cells, ex vivo stem cell expansion both to promote engraftment of transduced cells and to enable selection of stem cells for transplantation, lineage specific expression of the therapeutic transgene, targeted gene delivery, and eventually gene editing of the deficient mutant genes. Promoting engraftment by temporary mobilization of endogenous stem cells to open the stem cell niches in the bone marrow has been proposed [9] and applied successfully in the X-SCID mouse model [26]. An initial success has recently been reported in gene editing [20]. If the current clinical trials using lentiviral stem cell gene transfer prove efficacious and safe, its rapid clinical implementation in a variety of eligible inherited disorders will become within reach in the interest of the patients involved and thereby of health care and its costs.
Funding
Funding was provided by the European Commission’s 5th, 6th and 7th Framework Programs, Contracts QLK3-CT- 2001-00427-INHERINET, LSHB-CT-2004-005242-CONSERT, LSHB-CT-2006-19038-Magselectofection, Grant Agreement 222878-PERSIST and Grant agreement 261387 CELL-PID, and by the Netherlands Health Research and Development Organization ZonMW (Translational Gene Therapy program grants 43100016 and 43400010).
Author Disclosure Statement
The author declares no competing financial interest.
References
- Aiuti A, Cassani B, Andolfi G, Mirolo M, Biasco L, Recchia A, Urbinati F, Valacca C, Scaramuzza S, Aker M, Slavin S, Cazzola M, Sartori D, Ambrosi A, Di Serio C, Roncarolo MG, Mavilio F, Bordignon C. Multilineage hematopoietic reconstitution without clonal selection in ADA-SCID patients treated with stem cell gene therapy. J Clin Invest 2007; 117 (8): 2233-2240.
- Aldenhoven M, Wynn RF, Orchard PJ, O’Meara A, Veys P, Fischer A,Valayannopoulos V, Neven B, Rovelli A, Prasad VK, Tolar J, Allewelt H, Jones SA, Parini R, Renard M, Bordon V, Wulffraat NM, de Koning TJ, Shapiro EG, Kurtzberg J, Boelens JJ. Long-term outcome of Hurler syndrome patients after hematopoietic cell transplantation: an international multicenter study. Blood 2015; 125 (13): 2164-2172.
- Baum C, von Kalle C, Staal FJ, Li Z, Fehse B, Schmidt M, Weerkamp F, Karlsson S, Wagemaker G, Williams DA. Chance or necessity? Insertional mutagenesis in gene therapy and its consequences. Mol Ther 2004; 9 (1): 5-13.
- Biasco L, Ambrosi A, Pellin D, Bartholomae C, Brigida I, Roncarolo MG, Di Serio C, von Kalle C, Schmidt M, Aiuti A. Integration profile of retroviralvector in gene therapy treated patients is cell-specific according to gene expression and chromatin conformation of target cell. EMBO Mol Med 2011; 3 (2): 89-101.
- Biffi A, Montini E, Lorioli L, Cesani M, Fumagalli F, Plati T, Baldoli C, Martino S, Calabria A, Canale S, Benedicenti F, Vallanti G, Biasco L, Leo S, Kabbara N, Zanetti G, Rizzo WB, Mehta NA, Cicalese MP, Casiraghi M, Boelens JJ, Del Carro U, Dow DJ, Schmidt M, Assanelli A, Neduva V, Di Serio C, Stupka E, Gardner J, von Kalle C, Bordignon C, Ciceri F, Rovelli A, Roncarolo MG, Aiuti A, Sessa M, Naldini L. Lentiviral hematopoietic stem cell gene therapy benefits metachromatic leukodystrophy. Science 2013;341(6148):1233158.
- Braun CJ, Boztug K, Paruzynski A, Witzel M, Schwarzer A, Rothe M, Modlich U, Beier R, Göhring G, Steinemann D, Fronza R, Ball CR, Haemmerle R, Naundorf S, Kühlcke K, Rose M, Fraser C, Mathias L, Ferrari R, Abboud MR, Al- Herz W, Kondratenko I, Maródi L, Glimm H, Schlegelberger B, Schambach A, Albert MH, Schmidt M, von Kalle C, Klein C. Gene therapy for Wiskott-Aldrichsyndrome-longterm efficacy and genotoxicity. Sci Transl Med 2014; 6 (227): 227ra33.
- Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy.Cartier N, Hacein- Bey-Abina S, Bartholomae CC, Veres G, Schmidt M, Kutschera I, Vidaud M, Abel U, Dal-Cortivo L, Caccavelli L, Mahlaoui N, Kiermer V, Mittelstaedt D, Bellesme C, Lahlou N, Lefrère F, Blanche S, Audit M, Payen E, Leboulch P, l’Homme B, Bougnères P, Von Kalle C, Fischer A, Cavazzana- Calvo M, Aubourg P. Science 2009; 326 (5954): 818-823.
- Cesana D, Ranzani M, Volpin M, Bartholomae C, Duros C, Artus A, Merella S, Benedicenti F, Sergi Sergi L, Sanvito F, Brombin C, Nonis A, Serio CD, Doglioni C, von Kalle C, Schmidt M, Cohen-Haguenauer O, Naldini L, Montini E. Uncovering and dissecting the genotoxicity of self-inactivating lentiviral vectors in vivo. Mol Ther 2014; 22 (4): 774-785.
- Chen J, Larochelle A, Fricker S, Bridger G, Dunbar CE, Abkowitz JL. Mobilization as a preparative regimen for hematopoietic stem cell transplantation. Blood 2006; 107 (9): 3764-3771.
- Cicalese MP, Ferrua F, Castagnaro L, Pajno R, Barzaghi F, Giannelli S, Dionisio F, Brigida I, Bonopane M, Casiraghi M, Tabucchi A, Carlucci F, Grunebaum E, Adeli M, Bredius RG, Puck JM, Stepensky P, Tezcan I, Rolfe K, De Boever E, Reinhardt RR, Appleby J, Ciceri F, Roncarolo MG, Aiuti A. Update on the safety and efficacy of retroviral gene therapy for immunodeficiency due to adenosine deaminase deficiency. Blood 2016; 128 (1): 45-54.
- De Ravin SS, Reik A, Liu PQ, Li L, Wu X, Su L, Raley C, Theobald N, Choi U, Song AH, Chan A, Pearl JR, Paschon DE, Lee J, Newcombe H, Koontz S, Sweeney C, Shivak DA, Zarember KA, Peshwa MV, Gregory PD, Urnov FD, Malech HL. Targeted gene addition in human CD34(+) hematopoietic cells for correction of X-linked chronic granulomatous disease. Nat Biotechnol 2016; 34 (4): 424-429.
- Deichmann A, Brugman MH, Bartholomae CC, Schwarzwaelder K, Verstegen MM, Howe SJ, Arens A, Ott MG, Hoelzer D, Seger R, Grez M, Hacein-Bey-Abina S, Cavazzana- Calvo M, Fischer A, Paruzynski A, Gabriel R, Glimm H, Abel U, Cattoglio C, Mavilio F, Cassani B, Aiuti A, Dunbar CE, Baum C, Gaspar HB, Thrasher AJ, von Kalle C, Schmidt M, Wagemaker G. Insertion sites in engrafted cells cluster within a limited repertoire of genomic areas after gammaretroviral vector gene therapy. Mol Ther 2011; 19 (11): 2031- 2039.
- Deichmann A, Hacein-Bey-Abina S, Schmidt M, Garrigue A, Brugman MH, Hu J, Glimm H, Gyapay G, Prum B, Fraser CC, Fischer N, Schwarzwaelder K, Siegler ML, de Ridder D, Pike-Overzet K, Howe SJ, Thrasher AJ, Wagemaker G, Abel U, Staal FJ, Delabesse E, Villeval JL, Aronow B, Hue C, Prinz C, Wissler M, Klanke C, Weissenbach J, Alexander I, Fischer A, von Kalle C, Cavazzana-Calvo M. Vector integration is nonrandom and clustered and influences the fate of lymphopoiesis in SCID-X1 gene therapy. J Clin Invest 2007; 117 (8): 2225-2232.
- Desmaris N, Verot L, Puech JP, Caillaud C, Vanier MT, Heard JM. Prevention of neuropathology in the mouse model of Hurler syndrome. Ann Neurol 2004; 56 (1): 68-76.
- Enquist IB, Nilsson E, Ooka A, Månsson JE, Olsson K, Ehinger M, Brady RO, Richter J, Karlsson S. Effective cell and gene therapy in a murine model of Gaucher disease. Proc Natl Acad Sci USA 2006; 103 (37): 13819-13824.
- Fuller M, Van der Ploeg A, Reuser AJ, Anson DS, Hopwood JJ. Isolation and characterization of a recombinant, precursor form of lysosomal acid alpha-glucosidase. Eur J Biochem 1995; 234 (3): 903-909.
- Gaspar HB, Parsley KL, Howe S, King D, Gilmour KC, Sinclair J, Brouns G, Schmidt M, Von Kalle C, Barington T, Jakobsen MA, Christensen HO, Al Ghonaium A, White HN, Smith JL, Levinsky RJ, Ali RR, Kinnon C, Thrasher AJ. Gene therapy of X-linked severe combined immunodeficiency by use of a pseudotyped gammaretroviral vector. Lancet 2004 ; 364 (9452): 2181-2187.
- Gaspar HB, Cooray S, Gilmour KC, Parsley KL, Adams S, Howe SJ, Al Ghonaium A, Bayford J, Brown L, Davies EG, Kinnon C, Thrasher AJ. Long-term persistence of a polyclonal T cell repertoire after gene therapy for X-linked severe combined immunodeficiency. Sci Transl Med 2011; 3 (97): 97ra79.
- Gaspar HB, Cooray S, Gilmour KC, Parsley KL, Zhang F, Adams S, Bjorkegren E, Bayford J, Brown L, Davies EG, Veys P, Fairbanks L, Bordon V, Petropoulou T, Kinnon C, Thrasher AJ. Hematopoietic stem cell gene therapy for adenosine deaminase-deficient severe combined immunodeficiency leads to long-term immunological recovery and metabolic correction. Sci Transl Med 2011; 3 (97): 97ra80.
- Genovese P, Schiroli G, Escobar G, Di Tomaso T, Firrito C, Calabria A, Moi D, Mazzieri R, Bonini C, Holmes MC, Gregory PD, van der Burg M, Gentner B, MontiniE, Lombardo A, Naldini L. Targeted genome editing in human repopulating haematopoietic stem cells. Nature 2014; 510 (7504): 235-240.
- Gentner B, Visigalli I, Hiramatsu H, Lechman E, Ungari S, Giustacchini A, Schira G, Amendola M, Quattrini A, Martino S, Orlacchio A, Dick JE, Biffi A, Naldini L. Identification of hematopoietic stem cell-specific miRNAs enables gene therapy of globoid cell leukodystrophy. Sci Transl Med 2010; 2 (58): 58ra84.
- Hacein-Bey-Abina S, Hauer J, Lim A, Picard C, Wang GP, Berry CC, Martinache C, Rieux-Laucat F, Latour S, Belohradsky BH, Leiva L, Sorensen R, Debré M, Casanova JL, Blanche S, Durandy A, Bushman FD, Fischer A, Cavazzana- Calvo M. Efficacy of gene therapy for X-linked severe combined immunodeficiency. N Engl J Med 2010; 363 (4): 355-364.
- Hacein-Bey-Abina S, Pai SY, Gaspar HB, Armant M, Berry CC, Blanche S, Bleesing J, Blondeau J, de Boer H, Buckland KF, Caccavelli L, Cros G, De Oliveira S, Fernández KS, Guo D, Harris CE, Hopkins G, Lehmann LE, Lim A, London WB, van der Loo JC, Malani N, Male F, Malik P, Marinovic MA, McNicol AM, Moshous D, Neven B, Oleastro M, Picard C, Ritz J, Rivat C, Schambach A, Shaw KL, Sherman EA, Silberstein LE, Six E, Touzot F, Tsytsykova A, Xu-Bayford J, Baum C, Bushman FD, Fischer A, Kohn DB, Filipovich AH, Notarangelo LD, Cavazzana M, Williams DA, Thrasher AJ. A modified γ-retrovirus vector for X-linked severe combined immunodeficiency. N Engl J Med 2014; 371 (15): 1407-1417.
- Hoogerbrugge PM, Suzuki K, Suzuki K, Poorthuis BJ, Kobayashi T, Wagemaker G, van Bekkum DW. Donor-derived cells in the central nervous system of twitcher mice after bone marrow transplantation. Science 1988; 239 (4843): 1035-1038.
- Hoogerbrugge PM, Wagemaker G, van Bekkum DW, Reuser AJ, vd Ploeg AT. Bone marrow transplantation for Pompé’s disease. N Engl J Med 1986; 315 (1): 65-66.
- Huston MW, Riegman AR, Yadak R, van Helsdingen Y, de Boer H, van Til NP, Wagemaker G. Pretransplant mobilization with granulocyte colony-stimulating factor improves B-cell reconstitution by lentiviral vector gene therapy in SCID-X1 mice. Hum Gene Ther 2014; 25 (10): 905-914.
- Kaufmann KB, Chiriaco M, Siler U, Finocchi A, Reichenbach J, Stein S, Grez M. Gene therapy for chronic granulomatous disease: current status and future perspectives. Curr Gene Ther 2014; 14 (6): 447-460.
- Kustikova OS, Geiger H, Li Z, Brugman MH, Chambers SM, Shaw CA, Pike-Overzet K, de Ridder D, Staal FJ, von Keudell G, Cornils K, Nattamai KJ, Modlich U, Wagemaker G, Goodell MA, Fehse B, Baum C. Retroviral vector insertion sites associated with dominant hematopoietic clones mark “stemness” pathways. Blood 2007; 109 (5): 1897-1907.
- Naldini L, Blömer U, Gallay P, Ory D, Mulligan R, Gage FH, Verma IM, Trono D. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 1996; 272 (5259): 263-267.
- Pala F, Morbach H, Castiello MC, Schickel JN, Scaramuzza S, Chamberlain N, Cassani B, Glauzy S, Romberg N, Candotti F, Aiuti A, Bosticardo M, Villa A, Meffre E. Lentiviral- mediated gene therapy restores B cell tolerance in Wiskott-Aldrich syndrome patients. J Clin Invest 2015; 125 (10): 3941-3951.
- Pike-Overzet K, de Ridder D, Weerkamp F, Baert MR, Verstegen MM, Brugman MH, Howe SJ, Reinders MJ, Thrasher AJ, Wagemaker G, van Dongen JJ, Staal FJ. Gene therapy: is IL2RG oncogenic in T-cell development? Nature 2006; 443 (7109): E5; discussion E6-7.
- Pike-Overzet K, van der Burg M, Wagemaker G, van Dongen JJ, Staal FJ. New insights and unresolved issues regarding insertional mutagenesis in X-linked SCID gene therapy. Mol Ther 2007; 15 (11): 1910-1916.
- Pompe JC. Over idiopathische hypertrophie van het hart. 1932; N. T. v. G. 76.I.3.
- Rivera-Munoz P, Abramowski V, Jacquot S, André P, Charrier S, Lipson-Ruffert K, Fischer A, Galy A, Cavazzana M, de Villartay JP. Lymphopoiesis in transgenic mice over-expressing Artemis. Gene Ther 2016; 23 (2): 176-186.
- Rocha V, Wagner JE Jr, Sobocinski KA, Klein JP, Zhang MJ, Horowitz MM, Gluckman E. Graft-versus-host disease in children who have received a cord-blood or bone marrow transplant from an HLA-identical sibling. Eurocord and International Bone Marrow Transplant Registry Working Committee on Alternative Donor and Stem Cell Sources. N Engl J Med 2000; 342 (25): 1846-1854.
- Schambach A, Bohne J, Baum C, Hermann FG, Egerer L, von Laer D, Giroglou T. Woodchuck hepatitis virus post-transcriptional regulatory element deleted from X protein and promoter sequences enhances retroviral vector titer and expression. Gene Ther 2006; 13 (7): 641-645.
- Schwarzwaelder K, Howe SJ, Schmidt M, Brugman MH, Deichmann A, Glimm H, Schmidt S, Prinz C, Wissler M, King DJ, Zhang F, Parsley KL, Gilmour KC, Sinclair J, Bayford J, Peraj R, Pike-Overzet K, Staal FJ, de Ridder D, Kinnon C, Abel U, Wagemaker G, Gaspar HB, Thrasher AJ, von Kalle C. Gammaretrovirus-mediated correction of SCID-X1 is associated with skewed vector integration site distribution in vivo. J Clin Invest 2007; 117 (8): 2241-2249.
- Shou Y, Ma Z, Lu T, Sorrentino BP. Unique risk factors for insertional mutagenesis in a mouse model of XSCID gene therapy. Proc Natl Acad Sci USA 2006; 103 (31): 11730-11735.
- van den Hout HM, Hop W, van Diggelen OP, Smeitink JA, Smit GP, Poll-The BT, Bakker HD, Loonen MC, de Klerk JB, Reuser AJ, van der Ploeg AT. The natural course of infantile Pompe’s disease: 20 original cases compared with 133 cases from the literature. Pediatrics 2003; 112 (2): 332-340.
- van der Ploeg AT, Reuser AJ. Pompe’s disease. Lancet 2008 Oct 11; 372 (9646): 1342-53.
- van Gelder CM, Hoogeveen-Westerveld M, Kroos MA, Plug I, van der Ploeg AT, Reuser AJ. Enzyme therapy and immune response in relation to CRIM status: the Dutch experience in classic infantile Pompe disease. J Inherit Metab Dis. 2015; 38 (2): 305-314.
- van Til NP, Wagemaker G. Lentiviral gene transduction of mouse and human hematopoietic stem cells. Methods Mol Biol 2014; 1185: 311-319.
- Visigalli I, Delai S, Politi LS, Di Domenico C, Cerri F, Mrak E, D’Isa R, Ungaro D, Stok M, Sanvito F, Mariani E, Staszewsky L, Godi C, Russo I, Cecere F, Del Carro U, Rubinacci A, Brambilla R, Quattrini A, Di Natale P, Ponder K, Naldini L, Biffi A. Gene therapy augments the efficacy of hematopoietic cell transplantation and fully corrects mucopolysaccharidosis type I phenotype in the mouse model. Blood 2010; 116 (24): 5130-5139.
- Visigalli I, Delai S, Ferro F, Cecere F, Vezzoli M, Sanvito F, Chanut F, Benedicenti F, Spinozzi G, Wynn R, Calabria A, Naldini L, Montini E, Cristofori P, Biffi A. Preclinical testing of the safety and tolerability of LV-mediated above normal alpha-L-iduronidase expression in murine and human hematopoietic cells using toxicology and biodistribution GLP studies. Hum Gene Ther 2016 Jul 18.
- Wagemaker G. Lentiviral hematopoietic stem cell gene therapy in inherited metabolic disorders. Hum Gene Ther 2014; 25 (10): 862-865.
- Walia JS, Neschadim A, Lopez-Perez O, Alayoubi A, Fan X, Carpentier S, Madden M, Lee CJ, Cheung F, Jaffray DA, Levade T, McCart JA, Medin JA. Autologous transplantation of lentivector/acid ceramidase-transduced hematopoietic cells in nonhuman primates. Hum Gene Ther 2011; 22 (6): 679-687.
- Yoshimitsu M, Higuchi K, Ramsubir S, Nonaka T, Rasaiah VI, Siatskas C, Liang SB, Murray GJ, Brady RO, Medin JA. Efficient correction of Fabry mice and patient cells mediated by lentiviral transduction of hematopoietic stem/progenitor cells. Gene Ther 2007; 14 (3): 256-265.
- Zhang F, Thornhill SI, Howe SJ, Ulaganathan M, Schambach A, Sinclair J, Kinnon C, Gaspar HB, Antoniou M, Thrasher AJ. Lentiviral vectors containing an enhancer-less ubiquitously acting chromatin opening element (UCOE) provide highly reproducible and stable transgene expression in hematopoietic cells. Blood. 2007; 110 (5): 1448-1457.
" ["~DETAIL_TEXT"]=> string(28175) "
Introduction
Rare diseases, of which 80% are inherited disorders, affect some 6% of the human population, amounting in Europe to around 30 million people. Since many result in chronic disability and cost-intensive care, the impact is disproportional and may well be over 20% of health care costs, although the current lack of registration, contrary to cancer, makes exact estimates difficult. Most of the approximately 7500 known inherited diseases lack a curative intervention, are managed by symptomatic treatment and, in case of a family history, by prenatal diagnosis. A small minority is included in newborn screening programs. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation, (iv) strict regulation of transgene product levels is not required, (v) the transgene produces levels sufficient for sustained alleviation of symptoms or cure, and (vi) adverse immune responses to the transgene product are not expected, do not interfere with efficacy, or can be successfully counteracted or circumvented. Many of the more than 100 primary immune deficiencies and around 40 lysosomal storage disorders meet those criteria.
This brief review covers development and prospect of gene therapy for inherited immune deficiencies and lysosomal storage disorders, extending an earlier review [45].
Initial development of hematopoietic stem cell gene therapy
Hematopoietic stem cell gene therapy has been developed over more than 20 years. The pioneering trials for X-linked severe combined immune deficiency (SCID) using gammaretroviral gene transfer vectors resulted in successful restoration of T cell immunity [17, 22] in 18 patients and in long-term survival for 17 patients out of 20, a survival rate similar to HLA-identical BM transplantation [35]. Unfortunately, in 5 patients autonomous T cell clones developed into leukemia, among which 1 patient did not survive. In the context of European collaborative projects, the pathogenesis was rapidly elucidated, resulting in a series of publications on mechanisms involved in gammaretroviral mutagenesis and oncogenesis [3,12,13,28,31,32,37]. Briefly, gammaretroviral vectors generally integrate near the transcription start sites of expressed genes with a preference for proto-oncogenes, which results in aberrant expression driven by the promoter/ enhancer of the therapeutic transgene and may result in a preleukemic state. It is not excluded that the phenotypes of the treated diseases co-predispose to leukemia development [38], given the absence of leukemia in the ADA-SCID trial [1, 4], a 25% incidence in the X-linked SCID trials and an over 75% incidence in a gammaretroviral Wiskott-Aldrich trial [6].
Development of lentiviral vector gene therapy in inherited storage disorders
The gammaretroviral vectors have been replaced by HIV-1 derived lentiviral vectors [29,36,48], which lack the propensity for integration near proto-oncogenes and have the added advantage of integrating into quiescent cells, such as longterm repopulating stem cells. In addition, third generation lentiviral vectors made self-inactivating (SIN) by deletion of enhancer regions from the long terminal repeat sequences reduce the risk of influencing nearby genes, resulting in favorable safety profiles [8]. Systematic disease specific efficacy and safety evaluations, including codon optimization and careful promoter selection have enabled initial clinical trials using these vectors for selected metabolic storage disorders (Table 1), which include adrenoleukodystrophy (ALD), metachromatic leukodystrophy (MLD), and Hurler (MPS I), Pompe (GSD II) and Fabry diseases. We have focused on Pompe disease, the only disorder so far developed for stem cell gene therapy [42] in which allogeneic stem cell transplantation has not been applied due to lack of enzyme expression in the hematopoietic system [25] and in addition studied stem cell gene therapy for Hurler syndrome.
Hurler Syndrome and Pompe disease
Hurler Syndrome (Mucopolysaccharidosis type I, OMIM # 252800) is a lethal autosomal recessive storage disorder caused by a deficiency of the lysosomal enzyme α-L-iduronidase (IDUA; EC 3.2.1.76). The deficiency leads to insufficient degradation of glycoaminoglycans (GAG’s’) that interferes with normal cellular function and causes a multisystem disorder affecting the CNS, liver, skeleton, lungs and sensory organs (corneal clouding and deafness), average expected life-span approximately 5-10 years. Treatment consists of alloSCT, currently the only therapeutic option to establish long-term survival and protection of the CNS. Enzyme replacement has a systemic effect but does not reach the brain or the skeletal bones. AlloSCT does not improve the skeleton pathology either. Although clinical outcome of SCT has improved by the use of umbilical cord stem cells, progressive bone disease persists, leading to severe handicaps. An international long term clinical follow-up cohort of Hurler patients treated with alloSCT including >80% of the patients successfully transplanted worldwide [2] showed that certain genotypes, age at HSCT, and poor performance at HSCT are predictors for poor skeletal and neurodevelopmental outcome. These findings warrant the development of a single curative gene therapy approach.
Pompe disease (glycogen storage disease type II, acid maltase deficiency, OMIM # 232300) is a rare autosomal recessive lysosomal storage disorder caused by mutations in the gene-encoding acid α-glucosidase (EC 3.2.1.20) [40]. Severe mutations cause complete enzyme deficiency, resulting in the classic infantile form of Pompe disease, which was first

described by the Dutch pathologist J.C.Pompe [33]. Symptoms are caused by glycogen accumulation, mainly in skeletal, cardiac and smooth muscle, but also in other tissues, including the central and peripheral nervous system. In the first months of life, patients present with progressive muscle weakness, hypertrophic cardiomyopathy, respiratory problems and feeding difficulties. If untreated, this leads to death before the age of one year [39]. Older children and adults may have up to 20-30% residual enzyme activity and show a more slowly progressive phenotype. The symptoms generally result from weakness of the (proximal) skeletal muscles. These patients eventually become wheelchair bound and ventilator dependent in late childhood or adulthood.
Enzyme replacement therapy (ERT) by administration of recombinant acid α-glucosidase [16,40] (Myozyme®) is currently the only effective treatment, requiring high dose biweekly administration. Although of considerable benefit to many patients, ERT is not curative, requires life-long administration, may result in immune responses to the recombinant enzyme [41] and, partly due to the high doses required for clinical efficacy, the costs are extremely high. Therefore, a corrective intervention with curative intent represents an unmet medical need.
Efficacy and safety evaluation of lentiviral vector gene therapy in the Hurler and Pompe mouse models
The natural course of Hurler’s disease is invalidating and lethal, and the drawbacks of the current therapeutic modalities justify a gene therapy approach. An IDUA knock-out mouse model has been developed suitable to study gene therapeutic approaches and made available to our research. A gene therapy study with HSC in the IDUA KO mice showed improvement of disease pathology, including the brain, but bone remodeling was not reported [14]. Using a codon optimized IDUA gene driven by a cellular promoter in a lentiviral vector to transduce hematopoietic stem cells, we demonstrated long-term expression in the hematopoietic system resulting in full correction of the Hurler phenotype (unpublished) without any severe adverse effects. Similar results and a further safety analysis were obtained in a parallel study [43,44], completing the preclinical analysis enabling an initial clinical trial in selected patients.
In the initial evaluation of lentiviral stem cell gene therapy for Pompe disease using an efficient overnight transduction protocol [42], we demonstrated that approximately 30% successfully transduced cells present in the bone marrow after sublethal total body irradiation as conditioning for transplantation resulted in high levels of α-glucosidase. Restoration of α-glucosidase activity in target tissues by uptake through the mannose-6-phosphate receptor reduced glycogen storage proportional to the enzyme levels achieved, with full correction of glycogen storage in liver and spleen, correction of the life-threatening cardiomyopathy, significantly improved respiration and improved, but not fully normalized skeletal muscle function. Of particular interest was the demonstration of robust immune tolerance to the recombinant transgene product. In the follow-up study (manuscript in preparation), codon-optimization of the therapeutic transgene resulted in full correction of the phenotype including skeletal muscles. Remarkably, although Pompe disease does not result in mental retardation or other neuronal problems, also brain glycogen levels normalized entirely, with all astrocytes, which play a key role in glycogen storage and glycogenolysis in the brain, showing active acid α-glucosidase activity. Apparently the microglia descendants of hematopoietic stem cells, which are capable of passing the blood-brain-barrier, as we originally demonstrated in the mouse model of Krabbe disease [24], provide sufficient acid α-glucosidase to normalize glycogen levels also in neuronal tissue. Up till now, hematopoietic stem cell gene therapy is the only approach to achieve both robust immune tolerance to the transgene product and efficacy in bypassing the blood/ brain barrier, as has also been observed by others [43].
Clinical implementation of stem cell gene therapy in primary immune deficiencies
A summary of the current developments is provided in Table 2. Briefly, successful clinical trials are ongoing for X-linked SCID, ADA-SCID and Wiskott-Aldrich syndrome, while

clinical trials are being prepared for RAG2 deficiency, chronic granulomatous disease and Artemis. Gene therapy for ADA-SCID, which is difficult to treat with allogeneic stem cell transplantation, has recently been registered as an advanced therapy medicinal product and will soon be considered as the standard treatment for this disease.
Further developments
The future development of stem cell gene therapy efficacy and safety would obviously benefit considerably from non-cytoreductive preparation of the patients to enable engraftment of the gene-corrected cells, ex vivo stem cell expansion both to promote engraftment of transduced cells and to enable selection of stem cells for transplantation, lineage specific expression of the therapeutic transgene, targeted gene delivery, and eventually gene editing of the deficient mutant genes. Promoting engraftment by temporary mobilization of endogenous stem cells to open the stem cell niches in the bone marrow has been proposed [9] and applied successfully in the X-SCID mouse model [26]. An initial success has recently been reported in gene editing [20]. If the current clinical trials using lentiviral stem cell gene transfer prove efficacious and safe, its rapid clinical implementation in a variety of eligible inherited disorders will become within reach in the interest of the patients involved and thereby of health care and its costs.
Funding
Funding was provided by the European Commission’s 5th, 6th and 7th Framework Programs, Contracts QLK3-CT- 2001-00427-INHERINET, LSHB-CT-2004-005242-CONSERT, LSHB-CT-2006-19038-Magselectofection, Grant Agreement 222878-PERSIST and Grant agreement 261387 CELL-PID, and by the Netherlands Health Research and Development Organization ZonMW (Translational Gene Therapy program grants 43100016 and 43400010).
Author Disclosure Statement
The author declares no competing financial interest.
References
- Aiuti A, Cassani B, Andolfi G, Mirolo M, Biasco L, Recchia A, Urbinati F, Valacca C, Scaramuzza S, Aker M, Slavin S, Cazzola M, Sartori D, Ambrosi A, Di Serio C, Roncarolo MG, Mavilio F, Bordignon C. Multilineage hematopoietic reconstitution without clonal selection in ADA-SCID patients treated with stem cell gene therapy. J Clin Invest 2007; 117 (8): 2233-2240.
- Aldenhoven M, Wynn RF, Orchard PJ, O’Meara A, Veys P, Fischer A,Valayannopoulos V, Neven B, Rovelli A, Prasad VK, Tolar J, Allewelt H, Jones SA, Parini R, Renard M, Bordon V, Wulffraat NM, de Koning TJ, Shapiro EG, Kurtzberg J, Boelens JJ. Long-term outcome of Hurler syndrome patients after hematopoietic cell transplantation: an international multicenter study. Blood 2015; 125 (13): 2164-2172.
- Baum C, von Kalle C, Staal FJ, Li Z, Fehse B, Schmidt M, Weerkamp F, Karlsson S, Wagemaker G, Williams DA. Chance or necessity? Insertional mutagenesis in gene therapy and its consequences. Mol Ther 2004; 9 (1): 5-13.
- Biasco L, Ambrosi A, Pellin D, Bartholomae C, Brigida I, Roncarolo MG, Di Serio C, von Kalle C, Schmidt M, Aiuti A. Integration profile of retroviralvector in gene therapy treated patients is cell-specific according to gene expression and chromatin conformation of target cell. EMBO Mol Med 2011; 3 (2): 89-101.
- Biffi A, Montini E, Lorioli L, Cesani M, Fumagalli F, Plati T, Baldoli C, Martino S, Calabria A, Canale S, Benedicenti F, Vallanti G, Biasco L, Leo S, Kabbara N, Zanetti G, Rizzo WB, Mehta NA, Cicalese MP, Casiraghi M, Boelens JJ, Del Carro U, Dow DJ, Schmidt M, Assanelli A, Neduva V, Di Serio C, Stupka E, Gardner J, von Kalle C, Bordignon C, Ciceri F, Rovelli A, Roncarolo MG, Aiuti A, Sessa M, Naldini L. Lentiviral hematopoietic stem cell gene therapy benefits metachromatic leukodystrophy. Science 2013;341(6148):1233158.
- Braun CJ, Boztug K, Paruzynski A, Witzel M, Schwarzer A, Rothe M, Modlich U, Beier R, Göhring G, Steinemann D, Fronza R, Ball CR, Haemmerle R, Naundorf S, Kühlcke K, Rose M, Fraser C, Mathias L, Ferrari R, Abboud MR, Al- Herz W, Kondratenko I, Maródi L, Glimm H, Schlegelberger B, Schambach A, Albert MH, Schmidt M, von Kalle C, Klein C. Gene therapy for Wiskott-Aldrichsyndrome-longterm efficacy and genotoxicity. Sci Transl Med 2014; 6 (227): 227ra33.
- Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy.Cartier N, Hacein- Bey-Abina S, Bartholomae CC, Veres G, Schmidt M, Kutschera I, Vidaud M, Abel U, Dal-Cortivo L, Caccavelli L, Mahlaoui N, Kiermer V, Mittelstaedt D, Bellesme C, Lahlou N, Lefrère F, Blanche S, Audit M, Payen E, Leboulch P, l’Homme B, Bougnères P, Von Kalle C, Fischer A, Cavazzana- Calvo M, Aubourg P. Science 2009; 326 (5954): 818-823.
- Cesana D, Ranzani M, Volpin M, Bartholomae C, Duros C, Artus A, Merella S, Benedicenti F, Sergi Sergi L, Sanvito F, Brombin C, Nonis A, Serio CD, Doglioni C, von Kalle C, Schmidt M, Cohen-Haguenauer O, Naldini L, Montini E. Uncovering and dissecting the genotoxicity of self-inactivating lentiviral vectors in vivo. Mol Ther 2014; 22 (4): 774-785.
- Chen J, Larochelle A, Fricker S, Bridger G, Dunbar CE, Abkowitz JL. Mobilization as a preparative regimen for hematopoietic stem cell transplantation. Blood 2006; 107 (9): 3764-3771.
- Cicalese MP, Ferrua F, Castagnaro L, Pajno R, Barzaghi F, Giannelli S, Dionisio F, Brigida I, Bonopane M, Casiraghi M, Tabucchi A, Carlucci F, Grunebaum E, Adeli M, Bredius RG, Puck JM, Stepensky P, Tezcan I, Rolfe K, De Boever E, Reinhardt RR, Appleby J, Ciceri F, Roncarolo MG, Aiuti A. Update on the safety and efficacy of retroviral gene therapy for immunodeficiency due to adenosine deaminase deficiency. Blood 2016; 128 (1): 45-54.
- De Ravin SS, Reik A, Liu PQ, Li L, Wu X, Su L, Raley C, Theobald N, Choi U, Song AH, Chan A, Pearl JR, Paschon DE, Lee J, Newcombe H, Koontz S, Sweeney C, Shivak DA, Zarember KA, Peshwa MV, Gregory PD, Urnov FD, Malech HL. Targeted gene addition in human CD34(+) hematopoietic cells for correction of X-linked chronic granulomatous disease. Nat Biotechnol 2016; 34 (4): 424-429.
- Deichmann A, Brugman MH, Bartholomae CC, Schwarzwaelder K, Verstegen MM, Howe SJ, Arens A, Ott MG, Hoelzer D, Seger R, Grez M, Hacein-Bey-Abina S, Cavazzana- Calvo M, Fischer A, Paruzynski A, Gabriel R, Glimm H, Abel U, Cattoglio C, Mavilio F, Cassani B, Aiuti A, Dunbar CE, Baum C, Gaspar HB, Thrasher AJ, von Kalle C, Schmidt M, Wagemaker G. Insertion sites in engrafted cells cluster within a limited repertoire of genomic areas after gammaretroviral vector gene therapy. Mol Ther 2011; 19 (11): 2031- 2039.
- Deichmann A, Hacein-Bey-Abina S, Schmidt M, Garrigue A, Brugman MH, Hu J, Glimm H, Gyapay G, Prum B, Fraser CC, Fischer N, Schwarzwaelder K, Siegler ML, de Ridder D, Pike-Overzet K, Howe SJ, Thrasher AJ, Wagemaker G, Abel U, Staal FJ, Delabesse E, Villeval JL, Aronow B, Hue C, Prinz C, Wissler M, Klanke C, Weissenbach J, Alexander I, Fischer A, von Kalle C, Cavazzana-Calvo M. Vector integration is nonrandom and clustered and influences the fate of lymphopoiesis in SCID-X1 gene therapy. J Clin Invest 2007; 117 (8): 2225-2232.
- Desmaris N, Verot L, Puech JP, Caillaud C, Vanier MT, Heard JM. Prevention of neuropathology in the mouse model of Hurler syndrome. Ann Neurol 2004; 56 (1): 68-76.
- Enquist IB, Nilsson E, Ooka A, Månsson JE, Olsson K, Ehinger M, Brady RO, Richter J, Karlsson S. Effective cell and gene therapy in a murine model of Gaucher disease. Proc Natl Acad Sci USA 2006; 103 (37): 13819-13824.
- Fuller M, Van der Ploeg A, Reuser AJ, Anson DS, Hopwood JJ. Isolation and characterization of a recombinant, precursor form of lysosomal acid alpha-glucosidase. Eur J Biochem 1995; 234 (3): 903-909.
- Gaspar HB, Parsley KL, Howe S, King D, Gilmour KC, Sinclair J, Brouns G, Schmidt M, Von Kalle C, Barington T, Jakobsen MA, Christensen HO, Al Ghonaium A, White HN, Smith JL, Levinsky RJ, Ali RR, Kinnon C, Thrasher AJ. Gene therapy of X-linked severe combined immunodeficiency by use of a pseudotyped gammaretroviral vector. Lancet 2004 ; 364 (9452): 2181-2187.
- Gaspar HB, Cooray S, Gilmour KC, Parsley KL, Adams S, Howe SJ, Al Ghonaium A, Bayford J, Brown L, Davies EG, Kinnon C, Thrasher AJ. Long-term persistence of a polyclonal T cell repertoire after gene therapy for X-linked severe combined immunodeficiency. Sci Transl Med 2011; 3 (97): 97ra79.
- Gaspar HB, Cooray S, Gilmour KC, Parsley KL, Zhang F, Adams S, Bjorkegren E, Bayford J, Brown L, Davies EG, Veys P, Fairbanks L, Bordon V, Petropoulou T, Kinnon C, Thrasher AJ. Hematopoietic stem cell gene therapy for adenosine deaminase-deficient severe combined immunodeficiency leads to long-term immunological recovery and metabolic correction. Sci Transl Med 2011; 3 (97): 97ra80.
- Genovese P, Schiroli G, Escobar G, Di Tomaso T, Firrito C, Calabria A, Moi D, Mazzieri R, Bonini C, Holmes MC, Gregory PD, van der Burg M, Gentner B, MontiniE, Lombardo A, Naldini L. Targeted genome editing in human repopulating haematopoietic stem cells. Nature 2014; 510 (7504): 235-240.
- Gentner B, Visigalli I, Hiramatsu H, Lechman E, Ungari S, Giustacchini A, Schira G, Amendola M, Quattrini A, Martino S, Orlacchio A, Dick JE, Biffi A, Naldini L. Identification of hematopoietic stem cell-specific miRNAs enables gene therapy of globoid cell leukodystrophy. Sci Transl Med 2010; 2 (58): 58ra84.
- Hacein-Bey-Abina S, Hauer J, Lim A, Picard C, Wang GP, Berry CC, Martinache C, Rieux-Laucat F, Latour S, Belohradsky BH, Leiva L, Sorensen R, Debré M, Casanova JL, Blanche S, Durandy A, Bushman FD, Fischer A, Cavazzana- Calvo M. Efficacy of gene therapy for X-linked severe combined immunodeficiency. N Engl J Med 2010; 363 (4): 355-364.
- Hacein-Bey-Abina S, Pai SY, Gaspar HB, Armant M, Berry CC, Blanche S, Bleesing J, Blondeau J, de Boer H, Buckland KF, Caccavelli L, Cros G, De Oliveira S, Fernández KS, Guo D, Harris CE, Hopkins G, Lehmann LE, Lim A, London WB, van der Loo JC, Malani N, Male F, Malik P, Marinovic MA, McNicol AM, Moshous D, Neven B, Oleastro M, Picard C, Ritz J, Rivat C, Schambach A, Shaw KL, Sherman EA, Silberstein LE, Six E, Touzot F, Tsytsykova A, Xu-Bayford J, Baum C, Bushman FD, Fischer A, Kohn DB, Filipovich AH, Notarangelo LD, Cavazzana M, Williams DA, Thrasher AJ. A modified γ-retrovirus vector for X-linked severe combined immunodeficiency. N Engl J Med 2014; 371 (15): 1407-1417.
- Hoogerbrugge PM, Suzuki K, Suzuki K, Poorthuis BJ, Kobayashi T, Wagemaker G, van Bekkum DW. Donor-derived cells in the central nervous system of twitcher mice after bone marrow transplantation. Science 1988; 239 (4843): 1035-1038.
- Hoogerbrugge PM, Wagemaker G, van Bekkum DW, Reuser AJ, vd Ploeg AT. Bone marrow transplantation for Pompé’s disease. N Engl J Med 1986; 315 (1): 65-66.
- Huston MW, Riegman AR, Yadak R, van Helsdingen Y, de Boer H, van Til NP, Wagemaker G. Pretransplant mobilization with granulocyte colony-stimulating factor improves B-cell reconstitution by lentiviral vector gene therapy in SCID-X1 mice. Hum Gene Ther 2014; 25 (10): 905-914.
- Kaufmann KB, Chiriaco M, Siler U, Finocchi A, Reichenbach J, Stein S, Grez M. Gene therapy for chronic granulomatous disease: current status and future perspectives. Curr Gene Ther 2014; 14 (6): 447-460.
- Kustikova OS, Geiger H, Li Z, Brugman MH, Chambers SM, Shaw CA, Pike-Overzet K, de Ridder D, Staal FJ, von Keudell G, Cornils K, Nattamai KJ, Modlich U, Wagemaker G, Goodell MA, Fehse B, Baum C. Retroviral vector insertion sites associated with dominant hematopoietic clones mark “stemness” pathways. Blood 2007; 109 (5): 1897-1907.
- Naldini L, Blömer U, Gallay P, Ory D, Mulligan R, Gage FH, Verma IM, Trono D. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 1996; 272 (5259): 263-267.
- Pala F, Morbach H, Castiello MC, Schickel JN, Scaramuzza S, Chamberlain N, Cassani B, Glauzy S, Romberg N, Candotti F, Aiuti A, Bosticardo M, Villa A, Meffre E. Lentiviral- mediated gene therapy restores B cell tolerance in Wiskott-Aldrich syndrome patients. J Clin Invest 2015; 125 (10): 3941-3951.
- Pike-Overzet K, de Ridder D, Weerkamp F, Baert MR, Verstegen MM, Brugman MH, Howe SJ, Reinders MJ, Thrasher AJ, Wagemaker G, van Dongen JJ, Staal FJ. Gene therapy: is IL2RG oncogenic in T-cell development? Nature 2006; 443 (7109): E5; discussion E6-7.
- Pike-Overzet K, van der Burg M, Wagemaker G, van Dongen JJ, Staal FJ. New insights and unresolved issues regarding insertional mutagenesis in X-linked SCID gene therapy. Mol Ther 2007; 15 (11): 1910-1916.
- Pompe JC. Over idiopathische hypertrophie van het hart. 1932; N. T. v. G. 76.I.3.
- Rivera-Munoz P, Abramowski V, Jacquot S, André P, Charrier S, Lipson-Ruffert K, Fischer A, Galy A, Cavazzana M, de Villartay JP. Lymphopoiesis in transgenic mice over-expressing Artemis. Gene Ther 2016; 23 (2): 176-186.
- Rocha V, Wagner JE Jr, Sobocinski KA, Klein JP, Zhang MJ, Horowitz MM, Gluckman E. Graft-versus-host disease in children who have received a cord-blood or bone marrow transplant from an HLA-identical sibling. Eurocord and International Bone Marrow Transplant Registry Working Committee on Alternative Donor and Stem Cell Sources. N Engl J Med 2000; 342 (25): 1846-1854.
- Schambach A, Bohne J, Baum C, Hermann FG, Egerer L, von Laer D, Giroglou T. Woodchuck hepatitis virus post-transcriptional regulatory element deleted from X protein and promoter sequences enhances retroviral vector titer and expression. Gene Ther 2006; 13 (7): 641-645.
- Schwarzwaelder K, Howe SJ, Schmidt M, Brugman MH, Deichmann A, Glimm H, Schmidt S, Prinz C, Wissler M, King DJ, Zhang F, Parsley KL, Gilmour KC, Sinclair J, Bayford J, Peraj R, Pike-Overzet K, Staal FJ, de Ridder D, Kinnon C, Abel U, Wagemaker G, Gaspar HB, Thrasher AJ, von Kalle C. Gammaretrovirus-mediated correction of SCID-X1 is associated with skewed vector integration site distribution in vivo. J Clin Invest 2007; 117 (8): 2241-2249.
- Shou Y, Ma Z, Lu T, Sorrentino BP. Unique risk factors for insertional mutagenesis in a mouse model of XSCID gene therapy. Proc Natl Acad Sci USA 2006; 103 (31): 11730-11735.
- van den Hout HM, Hop W, van Diggelen OP, Smeitink JA, Smit GP, Poll-The BT, Bakker HD, Loonen MC, de Klerk JB, Reuser AJ, van der Ploeg AT. The natural course of infantile Pompe’s disease: 20 original cases compared with 133 cases from the literature. Pediatrics 2003; 112 (2): 332-340.
- van der Ploeg AT, Reuser AJ. Pompe’s disease. Lancet 2008 Oct 11; 372 (9646): 1342-53.
- van Gelder CM, Hoogeveen-Westerveld M, Kroos MA, Plug I, van der Ploeg AT, Reuser AJ. Enzyme therapy and immune response in relation to CRIM status: the Dutch experience in classic infantile Pompe disease. J Inherit Metab Dis. 2015; 38 (2): 305-314.
- van Til NP, Wagemaker G. Lentiviral gene transduction of mouse and human hematopoietic stem cells. Methods Mol Biol 2014; 1185: 311-319.
- Visigalli I, Delai S, Politi LS, Di Domenico C, Cerri F, Mrak E, D’Isa R, Ungaro D, Stok M, Sanvito F, Mariani E, Staszewsky L, Godi C, Russo I, Cecere F, Del Carro U, Rubinacci A, Brambilla R, Quattrini A, Di Natale P, Ponder K, Naldini L, Biffi A. Gene therapy augments the efficacy of hematopoietic cell transplantation and fully corrects mucopolysaccharidosis type I phenotype in the mouse model. Blood 2010; 116 (24): 5130-5139.
- Visigalli I, Delai S, Ferro F, Cecere F, Vezzoli M, Sanvito F, Chanut F, Benedicenti F, Spinozzi G, Wynn R, Calabria A, Naldini L, Montini E, Cristofori P, Biffi A. Preclinical testing of the safety and tolerability of LV-mediated above normal alpha-L-iduronidase expression in murine and human hematopoietic cells using toxicology and biodistribution GLP studies. Hum Gene Ther 2016 Jul 18.
- Wagemaker G. Lentiviral hematopoietic stem cell gene therapy in inherited metabolic disorders. Hum Gene Ther 2014; 25 (10): 862-865.
- Walia JS, Neschadim A, Lopez-Perez O, Alayoubi A, Fan X, Carpentier S, Madden M, Lee CJ, Cheung F, Jaffray DA, Levade T, McCart JA, Medin JA. Autologous transplantation of lentivector/acid ceramidase-transduced hematopoietic cells in nonhuman primates. Hum Gene Ther 2011; 22 (6): 679-687.
- Yoshimitsu M, Higuchi K, Ramsubir S, Nonaka T, Rasaiah VI, Siatskas C, Liang SB, Murray GJ, Brady RO, Medin JA. Efficient correction of Fabry mice and patient cells mediated by lentiviral transduction of hematopoietic stem/progenitor cells. Gene Ther 2007; 14 (3): 256-265.
- Zhang F, Thornhill SI, Howe SJ, Ulaganathan M, Schambach A, Sinclair J, Kinnon C, Gaspar HB, Antoniou M, Thrasher AJ. Lentiviral vectors containing an enhancer-less ubiquitously acting chromatin opening element (UCOE) provide highly reproducible and stable transgene expression in hematopoietic cells. Blood. 2007; 110 (5): 1448-1457.
" ["DETAIL_TEXT_TYPE"]=> string(4) "html" ["~DETAIL_TEXT_TYPE"]=> string(4) "html" ["PREVIEW_TEXT"]=> string(0) "" ["~PREVIEW_TEXT"]=> string(0) "" ["PREVIEW_TEXT_TYPE"]=> string(4) "text" ["~PREVIEW_TEXT_TYPE"]=> string(4) "text" ["PREVIEW_PICTURE"]=> NULL ["~PREVIEW_PICTURE"]=> NULL ["LANG_DIR"]=> string(4) "/ru/" ["~LANG_DIR"]=> string(4) "/ru/" ["SORT"]=> string(3) "500" ["~SORT"]=> string(3) "500" ["CODE"]=> string(100) "gennaya-terapiya-lentivirusnymi-vektorami-v-gemo-poeticheskikh-stvolovykh-kletkakh-pri-vrozhdennykh-" ["~CODE"]=> string(100) "gennaya-terapiya-lentivirusnymi-vektorami-v-gemo-poeticheskikh-stvolovykh-kletkakh-pri-vrozhdennykh-" ["EXTERNAL_ID"]=> string(3) "685" ["~EXTERNAL_ID"]=> string(3) "685" ["IBLOCK_TYPE_ID"]=> string(7) "journal" ["~IBLOCK_TYPE_ID"]=> string(7) "journal" ["IBLOCK_CODE"]=> string(7) "volumes" ["~IBLOCK_CODE"]=> string(7) "volumes" ["IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["~IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["LID"]=> string(2) "s2" ["~LID"]=> string(2) "s2" ["EDIT_LINK"]=> NULL ["DELETE_LINK"]=> NULL ["DISPLAY_ACTIVE_FROM"]=> string(0) "" ["IPROPERTY_VALUES"]=> array(18) { ["ELEMENT_META_TITLE"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["ELEMENT_META_KEYWORDS"]=> string(216) "гемопоэтические стволовые клетки лентивирусный вектор генная терапия врожденные иммунодефициты лизосомные болезни" ["ELEMENT_META_DESCRIPTION"]=> string(353) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимовLentiviral Hematopoietic Stem Cell Gene Therapy in Inherited Immune and Lysosomal Enzyme Deficiencies" ["ELEMENT_PREVIEW_PICTURE_FILE_ALT"]=> string(2620) "Редкие болезни поражают, в целом, миллионы людей во всем мире. Многие из этих являются наследственными заболеваниями, ведущими к инвалидности и требующими дорогостоящего ухода. Генная терапия гемопоэтическими стволовыми клетками (ГТГСК) разработана за последние 20 лет. На современном уровне генная терапия выполнима при заболеваниях, для которых (1) есть идентифицированный генетический дефект, (2) диагноз ставится достаточно рано для действенного терапевтического вмешательства, (3) имеется специфическая экспериментальная модель для оценки эффективности и безопасности лечения. Соответствующие терапевтические трансгены должны также отвечать определенным биологическим критериям. Лентивирусные векторы третьего поколения выполнены самоинактивирующимися (SIN), путем делеции энхансерных участков из LTR-последовательностей, тем самым снижая риск воздействия на соседние гены, что приводит к достаточным уровням безопасности. В настоящее время лентивирусная ГТГСК вступила в фазу начального клинического внедрения для лечения иммунодефицитов и лизосомных болезней накопления. Мы обсуждаем начальные клинические испытания с применением этих векторов для некоторых метаболических болезней накопления, которые включают адренолейкодистрофию, метахроматическую лейкодистрофию, синдром Гурлер (MPS I), Помпе (GSD II), и болезнь Фабри. Данный краткий обзор обобщает развитие и современное клиническое внедрение этих подходов." ["ELEMENT_PREVIEW_PICTURE_FILE_TITLE"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["ELEMENT_DETAIL_PICTURE_FILE_ALT"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["ELEMENT_DETAIL_PICTURE_FILE_TITLE"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["SECTION_META_TITLE"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["SECTION_META_KEYWORDS"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["SECTION_META_DESCRIPTION"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["SECTION_PICTURE_FILE_ALT"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["SECTION_PICTURE_FILE_TITLE"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["SECTION_PICTURE_FILE_NAME"]=> string(100) "gennaya-terapiya-lentivirusnymi-vektorami-v-gemo-poeticheskikh-stvolovykh-kletkakh-pri-vrozhdennykh-" ["SECTION_DETAIL_PICTURE_FILE_ALT"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["SECTION_DETAIL_PICTURE_FILE_TITLE"]=> string(252) "Генная терапия лентивирусными векторами в гемо- поэтических стволовых клетках при врожденных дефицитах иммунитета и лизосомных энзимов" ["SECTION_DETAIL_PICTURE_FILE_NAME"]=> string(100) "gennaya-terapiya-lentivirusnymi-vektorami-v-gemo-poeticheskikh-stvolovykh-kletkakh-pri-vrozhdennykh-" ["ELEMENT_PREVIEW_PICTURE_FILE_NAME"]=> string(100) "gennaya-terapiya-lentivirusnymi-vektorami-v-gemo-poeticheskikh-stvolovykh-kletkakh-pri-vrozhdennykh-" ["ELEMENT_DETAIL_PICTURE_FILE_NAME"]=> string(100) "gennaya-terapiya-lentivirusnymi-vektorami-v-gemo-poeticheskikh-stvolovykh-kletkakh-pri-vrozhdennykh-" } ["FIELDS"]=> array(1) { ["IBLOCK_SECTION_ID"]=> string(2) "21" } ["PROPERTIES"]=> array(18) { ["KEYWORDS"]=> array(36) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(5) { [0]=> string(4) "8396" [1]=> string(4) "8397" [2]=> string(4) "8398" [3]=> string(4) "8399" [4]=> string(4) "8400" } ["VALUE"]=> array(5) { [0]=> string(3) "686" [1]=> string(3) "687" [2]=> string(3) "688" [3]=> string(3) "689" [4]=> string(3) "690" } ["DESCRIPTION"]=> array(5) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(5) { [0]=> string(3) "686" [1]=> string(3) "687" [2]=> string(3) "688" [3]=> string(3) "689" [4]=> string(3) "690" } ["~DESCRIPTION"]=> array(5) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" } ["SUBMITTED"]=> array(36) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8371" ["VALUE"]=> string(22) "11/28/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/28/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL } ["ACCEPTED"]=> array(36) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8372" ["VALUE"]=> string(22) "12/09/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "12/09/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL } ["PUBLISHED"]=> array(36) { ["ID"]=> string(2) "22" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Дата публикации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "PUBLISHED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "22" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Дата публикации" ["~DEFAULT_VALUE"]=> NULL } ["CONTACT"]=> array(36) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8373" ["VALUE"]=> string(3) "433" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "433" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHORS"]=> array(36) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(1) { [0]=> string(4) "8401" } ["VALUE"]=> array(1) { [0]=> string(3) "433" } ["DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(1) { [0]=> string(3) "433" } ["~DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_RU"]=> array(36) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8388" ["VALUE"]=> array(2) { ["TEXT"]=> string(31) "Герард Вагемакер" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(31) "Герард Вагемакер" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_RU"]=> array(36) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8389" ["VALUE"]=> array(2) { ["TEXT"]=> string(617) "Университет Эразмус, Роттердам, Нидерланды; Центр исследования и разработки стволовых клеток, Университет<br> Хашеттепе, Анкара, Турция; НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый<br> Санкт-Петербургский государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(599) "Университет Эразмус, Роттердам, Нидерланды; Центр исследования и разработки стволовых клеток, Университет
Хашеттепе, Анкара, Турция; НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый
Санкт-Петербургский государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_RU"]=> array(36) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8390" ["VALUE"]=> array(2) { ["TEXT"]=> string(2620) "Редкие болезни поражают, в целом, миллионы людей во всем мире. Многие из этих являются наследственными заболеваниями, ведущими к инвалидности и требующими дорогостоящего ухода. Генная терапия гемопоэтическими стволовыми клетками (ГТГСК) разработана за последние 20 лет. На современном уровне генная терапия выполнима при заболеваниях, для которых (1) есть идентифицированный генетический дефект, (2) диагноз ставится достаточно рано для действенного терапевтического вмешательства, (3) имеется специфическая экспериментальная модель для оценки эффективности и безопасности лечения. Соответствующие терапевтические трансгены должны также отвечать определенным биологическим критериям. Лентивирусные векторы третьего поколения выполнены самоинактивирующимися (SIN), путем делеции энхансерных участков из LTR-последовательностей, тем самым снижая риск воздействия на соседние гены, что приводит к достаточным уровням безопасности. В настоящее время лентивирусная ГТГСК вступила в фазу начального клинического внедрения для лечения иммунодефицитов и лизосомных болезней накопления. Мы обсуждаем начальные клинические испытания с применением этих векторов для некоторых метаболических болезней накопления, которые включают адренолейкодистрофию, метахроматическую лейкодистрофию, синдром Гурлер (MPS I), Помпе (GSD II), и болезнь Фабри. Данный краткий обзор обобщает развитие и современное клиническое внедрение этих подходов." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2620) "Редкие болезни поражают, в целом, миллионы людей во всем мире. Многие из этих являются наследственными заболеваниями, ведущими к инвалидности и требующими дорогостоящего ухода. Генная терапия гемопоэтическими стволовыми клетками (ГТГСК) разработана за последние 20 лет. На современном уровне генная терапия выполнима при заболеваниях, для которых (1) есть идентифицированный генетический дефект, (2) диагноз ставится достаточно рано для действенного терапевтического вмешательства, (3) имеется специфическая экспериментальная модель для оценки эффективности и безопасности лечения. Соответствующие терапевтические трансгены должны также отвечать определенным биологическим критериям. Лентивирусные векторы третьего поколения выполнены самоинактивирующимися (SIN), путем делеции энхансерных участков из LTR-последовательностей, тем самым снижая риск воздействия на соседние гены, что приводит к достаточным уровням безопасности. В настоящее время лентивирусная ГТГСК вступила в фазу начального клинического внедрения для лечения иммунодефицитов и лизосомных болезней накопления. Мы обсуждаем начальные клинические испытания с применением этих векторов для некоторых метаболических болезней накопления, которые включают адренолейкодистрофию, метахроматическую лейкодистрофию, синдром Гурлер (MPS I), Помпе (GSD II), и болезнь Фабри. Данный краткий обзор обобщает развитие и современное клиническое внедрение этих подходов." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["DOI"]=> array(36) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8375" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-56-62" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-56-62" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_EN"]=> array(36) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8391" ["VALUE"]=> array(2) { ["TEXT"]=> string(17) "Gerard Wagemaker*" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(17) "Gerard Wagemaker*" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_EN"]=> array(36) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8392" ["VALUE"]=> array(2) { ["TEXT"]=> string(599) "Erasmus University Rotterdam, The Netherlands; Center for Stem Cell Research and Development, Hacettepe University, Ankara, Turkey;<br> Raisa Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia<br> <br> Gerard Wagemaker, PhD, Professor, Erasmus University Rotterdam,<br> c/o PO Box 37048<br> 3005 LA Rotterdam, The Netherlands<br> <br> Phone: +31-6-51619585<br> E-mail: <a href="mailto:g.wagemaker@genetherapy.nl">g.wagemaker@genetherapy.nl</a><br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(523) "Erasmus University Rotterdam, The Netherlands; Center for Stem Cell Research and Development, Hacettepe University, Ankara, Turkey;
Raisa Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
Gerard Wagemaker, PhD, Professor, Erasmus University Rotterdam,
c/o PO Box 37048
3005 LA Rotterdam, The Netherlands
Phone: +31-6-51619585
E-mail: g.wagemaker@genetherapy.nl
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_EN"]=> array(36) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8393" ["VALUE"]=> array(2) { ["TEXT"]=> string(1328) "Rare diseases affect millions of people worldwide. Many of those are inherited disorders resulting in chronic disability and requiring cost-intensive care. Hematopoietic stem cell gene therapy has been developed over more than 20 years. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation. Appropriate therapeutic transgenes should also comply with certain biological criteria. Third-generation lentiviral vectors have been made self-inactivating (SIN) by deletion of enhancer regions from the LTR sequences thus reducing the risk of influencing nearby genes, resulting in favorable safety profiles. At the present time, lentiviral hematopoietic stem cell gene therapy has entered the stage of initial clinical implementation for immune deficiencies and lysosomal storage disorders. We discuss initial clinical trials using these vectors for selected metabolic storage disorders, which include adrenoleukodystrophy, metachromatic leukodystrophy, Hurler (MPS I), Pompe (GSD II), and Fabry diseases. This brief review summarizes the development and current clinical implementation of these approaches." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(1328) "Rare diseases affect millions of people worldwide. Many of those are inherited disorders resulting in chronic disability and requiring cost-intensive care. Hematopoietic stem cell gene therapy has been developed over more than 20 years. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation. Appropriate therapeutic transgenes should also comply with certain biological criteria. Third-generation lentiviral vectors have been made self-inactivating (SIN) by deletion of enhancer regions from the LTR sequences thus reducing the risk of influencing nearby genes, resulting in favorable safety profiles. At the present time, lentiviral hematopoietic stem cell gene therapy has entered the stage of initial clinical implementation for immune deficiencies and lysosomal storage disorders. We discuss initial clinical trials using these vectors for selected metabolic storage disorders, which include adrenoleukodystrophy, metachromatic leukodystrophy, Hurler (MPS I), Pompe (GSD II), and Fabry diseases. This brief review summarizes the development and current clinical implementation of these approaches." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["NAME_EN"]=> array(36) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8376" ["VALUE"]=> string(101) "Lentiviral Hematopoietic Stem Cell Gene Therapy in Inherited Immune and Lysosomal Enzyme Deficiencies" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(101) "Lentiviral Hematopoietic Stem Cell Gene Therapy in Inherited Immune and Lysosomal Enzyme Deficiencies" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" } ["FULL_TEXT_RU"]=> array(36) { ["ID"]=> string(2) "42" ["TIMESTAMP_X"]=> string(19) "2015-09-07 20:29:18" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(23) "Полный текст" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(12) "FULL_TEXT_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "42" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(23) "Полный текст" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["PDF_RU"]=> array(36) { ["ID"]=> string(2) "43" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF RUS" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_RU" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "43" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8394" ["VALUE"]=> string(3) "266" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "266" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF RUS" ["~DEFAULT_VALUE"]=> string(0) "" } ["PDF_EN"]=> array(36) { ["ID"]=> string(2) "44" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF ENG" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "44" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8395" ["VALUE"]=> string(3) "267" ["DESCRIPTION"]=> NULL ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "267" ["~DESCRIPTION"]=> NULL ["~NAME"]=> string(7) "PDF ENG" ["~DEFAULT_VALUE"]=> string(0) "" } ["NAME_LONG"]=> array(36) { ["ID"]=> string(2) "45" ["TIMESTAMP_X"]=> string(19) "2023-04-13 00:55:00" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(72) "Название (для очень длинных заголовков)" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "NAME_LONG" ["DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "45" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(80) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(72) "Название (для очень длинных заголовков)" ["~DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } } } ["DISPLAY_PROPERTIES"]=> array(13) { ["AUTHOR_EN"]=> array(37) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8391" ["VALUE"]=> array(2) { ["TEXT"]=> string(17) "Gerard Wagemaker*" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(17) "Gerard Wagemaker*" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(17) "Gerard Wagemaker*" } ["SUMMARY_EN"]=> array(37) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8393" ["VALUE"]=> array(2) { ["TEXT"]=> string(1328) "Rare diseases affect millions of people worldwide. Many of those are inherited disorders resulting in chronic disability and requiring cost-intensive care. Hematopoietic stem cell gene therapy has been developed over more than 20 years. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation. Appropriate therapeutic transgenes should also comply with certain biological criteria. Third-generation lentiviral vectors have been made self-inactivating (SIN) by deletion of enhancer regions from the LTR sequences thus reducing the risk of influencing nearby genes, resulting in favorable safety profiles. At the present time, lentiviral hematopoietic stem cell gene therapy has entered the stage of initial clinical implementation for immune deficiencies and lysosomal storage disorders. We discuss initial clinical trials using these vectors for selected metabolic storage disorders, which include adrenoleukodystrophy, metachromatic leukodystrophy, Hurler (MPS I), Pompe (GSD II), and Fabry diseases. This brief review summarizes the development and current clinical implementation of these approaches." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(1328) "Rare diseases affect millions of people worldwide. Many of those are inherited disorders resulting in chronic disability and requiring cost-intensive care. Hematopoietic stem cell gene therapy has been developed over more than 20 years. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation. Appropriate therapeutic transgenes should also comply with certain biological criteria. Third-generation lentiviral vectors have been made self-inactivating (SIN) by deletion of enhancer regions from the LTR sequences thus reducing the risk of influencing nearby genes, resulting in favorable safety profiles. At the present time, lentiviral hematopoietic stem cell gene therapy has entered the stage of initial clinical implementation for immune deficiencies and lysosomal storage disorders. We discuss initial clinical trials using these vectors for selected metabolic storage disorders, which include adrenoleukodystrophy, metachromatic leukodystrophy, Hurler (MPS I), Pompe (GSD II), and Fabry diseases. This brief review summarizes the development and current clinical implementation of these approaches." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(1328) "Rare diseases affect millions of people worldwide. Many of those are inherited disorders resulting in chronic disability and requiring cost-intensive care. Hematopoietic stem cell gene therapy has been developed over more than 20 years. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation. Appropriate therapeutic transgenes should also comply with certain biological criteria. Third-generation lentiviral vectors have been made self-inactivating (SIN) by deletion of enhancer regions from the LTR sequences thus reducing the risk of influencing nearby genes, resulting in favorable safety profiles. At the present time, lentiviral hematopoietic stem cell gene therapy has entered the stage of initial clinical implementation for immune deficiencies and lysosomal storage disorders. We discuss initial clinical trials using these vectors for selected metabolic storage disorders, which include adrenoleukodystrophy, metachromatic leukodystrophy, Hurler (MPS I), Pompe (GSD II), and Fabry diseases. This brief review summarizes the development and current clinical implementation of these approaches." } ["DOI"]=> array(37) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8375" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-56-62" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-56-62" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-56-62" } ["NAME_EN"]=> array(37) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8376" ["VALUE"]=> string(101) "Lentiviral Hematopoietic Stem Cell Gene Therapy in Inherited Immune and Lysosomal Enzyme Deficiencies" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(101) "Lentiviral Hematopoietic Stem Cell Gene Therapy in Inherited Immune and Lysosomal Enzyme Deficiencies" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(101) "Lentiviral Hematopoietic Stem Cell Gene Therapy in Inherited Immune and Lysosomal Enzyme Deficiencies" } ["ORGANIZATION_EN"]=> array(37) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8392" ["VALUE"]=> array(2) { ["TEXT"]=> string(599) "Erasmus University Rotterdam, The Netherlands; Center for Stem Cell Research and Development, Hacettepe University, Ankara, Turkey;<br> Raisa Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia<br> <br> Gerard Wagemaker, PhD, Professor, Erasmus University Rotterdam,<br> c/o PO Box 37048<br> 3005 LA Rotterdam, The Netherlands<br> <br> Phone: +31-6-51619585<br> E-mail: <a href="mailto:g.wagemaker@genetherapy.nl">g.wagemaker@genetherapy.nl</a><br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(523) "Erasmus University Rotterdam, The Netherlands; Center for Stem Cell Research and Development, Hacettepe University, Ankara, Turkey;
Raisa Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
Gerard Wagemaker, PhD, Professor, Erasmus University Rotterdam,
c/o PO Box 37048
3005 LA Rotterdam, The Netherlands
Phone: +31-6-51619585
E-mail: g.wagemaker@genetherapy.nl
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(523) "Erasmus University Rotterdam, The Netherlands; Center for Stem Cell Research and Development, Hacettepe University, Ankara, Turkey;
Raisa Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
Gerard Wagemaker, PhD, Professor, Erasmus University Rotterdam,
c/o PO Box 37048
3005 LA Rotterdam, The Netherlands
Phone: +31-6-51619585
E-mail: g.wagemaker@genetherapy.nl
" } ["AUTHORS"]=> array(38) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(1) { [0]=> string(4) "8401" } ["VALUE"]=> array(1) { [0]=> string(3) "433" } ["DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(1) { [0]=> string(3) "433" } ["~DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(59) "Gerard Wagemaker" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["AUTHOR_RU"]=> array(37) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8388" ["VALUE"]=> array(2) { ["TEXT"]=> string(31) "Герард Вагемакер" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(31) "Герард Вагемакер" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(31) "Герард Вагемакер" } ["SUBMITTED"]=> array(37) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8371" ["VALUE"]=> string(22) "11/28/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/28/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "11/28/2016 12:00:00 am" } ["ACCEPTED"]=> array(37) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8372" ["VALUE"]=> string(22) "12/09/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "12/09/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "12/09/2016 12:00:00 am" } ["KEYWORDS"]=> array(38) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(5) { [0]=> string(4) "8396" [1]=> string(4) "8397" [2]=> string(4) "8398" [3]=> string(4) "8399" [4]=> string(4) "8400" } ["VALUE"]=> array(5) { [0]=> string(3) "686" [1]=> string(3) "687" [2]=> string(3) "688" [3]=> string(3) "689" [4]=> string(3) "690" } ["DESCRIPTION"]=> array(5) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(5) { [0]=> string(3) "686" [1]=> string(3) "687" [2]=> string(3) "688" [3]=> string(3) "689" [4]=> string(3) "690" } ["~DESCRIPTION"]=> array(5) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" [4]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(5) { [0]=> string(106) "гемопоэтические стволовые клетки" [1]=> string(83) "лентивирусный вектор" [2]=> string(71) "генная терапия" [3]=> string(93) "врожденные иммунодефициты" [4]=> string(79) "лизосомные болезни" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["CONTACT"]=> array(38) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8373" ["VALUE"]=> string(3) "433" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "433" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(59) "Gerard Wagemaker" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["SUMMARY_RU"]=> array(37) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8390" ["VALUE"]=> array(2) { ["TEXT"]=> string(2620) "Редкие болезни поражают, в целом, миллионы людей во всем мире. Многие из этих являются наследственными заболеваниями, ведущими к инвалидности и требующими дорогостоящего ухода. Генная терапия гемопоэтическими стволовыми клетками (ГТГСК) разработана за последние 20 лет. На современном уровне генная терапия выполнима при заболеваниях, для которых (1) есть идентифицированный генетический дефект, (2) диагноз ставится достаточно рано для действенного терапевтического вмешательства, (3) имеется специфическая экспериментальная модель для оценки эффективности и безопасности лечения. Соответствующие терапевтические трансгены должны также отвечать определенным биологическим критериям. Лентивирусные векторы третьего поколения выполнены самоинактивирующимися (SIN), путем делеции энхансерных участков из LTR-последовательностей, тем самым снижая риск воздействия на соседние гены, что приводит к достаточным уровням безопасности. В настоящее время лентивирусная ГТГСК вступила в фазу начального клинического внедрения для лечения иммунодефицитов и лизосомных болезней накопления. Мы обсуждаем начальные клинические испытания с применением этих векторов для некоторых метаболических болезней накопления, которые включают адренолейкодистрофию, метахроматическую лейкодистрофию, синдром Гурлер (MPS I), Помпе (GSD II), и болезнь Фабри. Данный краткий обзор обобщает развитие и современное клиническое внедрение этих подходов." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2620) "Редкие болезни поражают, в целом, миллионы людей во всем мире. Многие из этих являются наследственными заболеваниями, ведущими к инвалидности и требующими дорогостоящего ухода. Генная терапия гемопоэтическими стволовыми клетками (ГТГСК) разработана за последние 20 лет. На современном уровне генная терапия выполнима при заболеваниях, для которых (1) есть идентифицированный генетический дефект, (2) диагноз ставится достаточно рано для действенного терапевтического вмешательства, (3) имеется специфическая экспериментальная модель для оценки эффективности и безопасности лечения. Соответствующие терапевтические трансгены должны также отвечать определенным биологическим критериям. Лентивирусные векторы третьего поколения выполнены самоинактивирующимися (SIN), путем делеции энхансерных участков из LTR-последовательностей, тем самым снижая риск воздействия на соседние гены, что приводит к достаточным уровням безопасности. В настоящее время лентивирусная ГТГСК вступила в фазу начального клинического внедрения для лечения иммунодефицитов и лизосомных болезней накопления. Мы обсуждаем начальные клинические испытания с применением этих векторов для некоторых метаболических болезней накопления, которые включают адренолейкодистрофию, метахроматическую лейкодистрофию, синдром Гурлер (MPS I), Помпе (GSD II), и болезнь Фабри. Данный краткий обзор обобщает развитие и современное клиническое внедрение этих подходов." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(2620) "Редкие болезни поражают, в целом, миллионы людей во всем мире. Многие из этих являются наследственными заболеваниями, ведущими к инвалидности и требующими дорогостоящего ухода. Генная терапия гемопоэтическими стволовыми клетками (ГТГСК) разработана за последние 20 лет. На современном уровне генная терапия выполнима при заболеваниях, для которых (1) есть идентифицированный генетический дефект, (2) диагноз ставится достаточно рано для действенного терапевтического вмешательства, (3) имеется специфическая экспериментальная модель для оценки эффективности и безопасности лечения. Соответствующие терапевтические трансгены должны также отвечать определенным биологическим критериям. Лентивирусные векторы третьего поколения выполнены самоинактивирующимися (SIN), путем делеции энхансерных участков из LTR-последовательностей, тем самым снижая риск воздействия на соседние гены, что приводит к достаточным уровням безопасности. В настоящее время лентивирусная ГТГСК вступила в фазу начального клинического внедрения для лечения иммунодефицитов и лизосомных болезней накопления. Мы обсуждаем начальные клинические испытания с применением этих векторов для некоторых метаболических болезней накопления, которые включают адренолейкодистрофию, метахроматическую лейкодистрофию, синдром Гурлер (MPS I), Помпе (GSD II), и болезнь Фабри. Данный краткий обзор обобщает развитие и современное клиническое внедрение этих подходов." } ["ORGANIZATION_RU"]=> array(37) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8389" ["VALUE"]=> array(2) { ["TEXT"]=> string(617) "Университет Эразмус, Роттердам, Нидерланды; Центр исследования и разработки стволовых клеток, Университет<br> Хашеттепе, Анкара, Турция; НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый<br> Санкт-Петербургский государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(599) "Университет Эразмус, Роттердам, Нидерланды; Центр исследования и разработки стволовых клеток, Университет
Хашеттепе, Анкара, Турция; НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый
Санкт-Петербургский государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(599) "Университет Эразмус, Роттердам, Нидерланды; Центр исследования и разработки стволовых клеток, Университет
Хашеттепе, Анкара, Турция; НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый
Санкт-Петербургский государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия
" } } } [7]=> array(49) { ["IBLOCK_SECTION_ID"]=> string(2) "21" ["~IBLOCK_SECTION_ID"]=> string(2) "21" ["ID"]=> string(3) "700" ["~ID"]=> string(3) "700" ["IBLOCK_ID"]=> string(1) "2" ["~IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["~NAME"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["ACTIVE_FROM"]=> NULL ["~ACTIVE_FROM"]=> NULL ["TIMESTAMP_X"]=> string(22) "05/03/2017 09:38:32 am" ["~TIMESTAMP_X"]=> string(22) "05/03/2017 09:38:32 am" ["DETAIL_PAGE_URL"]=> string(161) "/en/archive/vypusk-5-nomer-4/eksperimentalnye-issledovaniya/vliyanie-funktsionalnykh-kharakteristik-stromalnykh-kletok-kostnogo-mozga-retsipienta-na-prizhivleni/" ["~DETAIL_PAGE_URL"]=> string(161) "/en/archive/vypusk-5-nomer-4/eksperimentalnye-issledovaniya/vliyanie-funktsionalnykh-kharakteristik-stromalnykh-kletok-kostnogo-mozga-retsipienta-na-prizhivleni/" ["LIST_PAGE_URL"]=> string(12) "/en/archive/" ["~LIST_PAGE_URL"]=> string(12) "/en/archive/" ["DETAIL_TEXT"]=> string(23125) "
Introduction
Morphological and functional patterns of malignant cells are largely determined by specific somatic gene aberrations, which represent the major factor in biology of oncohematological disorders. Meanwhile, some data suggest expression of tumor markers in non-hematopoietic bone marrow cells from the stromal microenvironment [2, 6]. Hence, potential mechanisms of leukemogenesis, like as specific gene mutations, may include certain effects of cellular environment which may promote proliferation and development of hematopoietic stem cells [7, 16]. The so-called bone marrow niches are represented by endothelium, stromal cells, macrophages etc. The role of bone marrow stromal cells (BMSC) in hematopoietic regulation was first described by A.Ya Friedenstein fifty years ago. There are several types of stromal cells, in particular, mesenchymal stem cells (MSC) which are best studied for their potential clinical applications. MSCs are shown to suppress immune conflicts in allogeneic transplantation [10]; to replace and restore functions of the damaged non-hematopoietic tissues [14], support hematopoiesis when co-transplanted with hematopoietic stem cells (HSCs) [11]. These effects are based on favorable actions upon proliferation and differentiation of different hematopoietic lineages, e.g., myeloid and lymphoid progenitors. Hence, studies of stromal cells seem to be quite rational in view of their possible involvement into pathogenesis of oncohematological disorders.
It is known that, despite successful engraftment of donor-derived hematopoietic stem cells after allogeneic stem cell transplantation (alloSCT), the mesenchymal stem cells, are, in general, of host origin, even years post-transplant [15]. Some patients after alloSCT do not recover their stem cells despite receiving large amounts of CD34+ blood progenitors. Possible basis for the graft failure may be due to affection of hematopoietic niche by conditioning chemotherapy which could disturb its supportive (nursing) capacity. A number of researchers have identified the cells of osteoblast lineage to be a key cellular component of the HSC niche. These cells are responsible for bone formation [4, 19]. In addition, some transplant-associated factors might affect the osteoblasts, along with direct driving of hematopoiesis. Thus, it may be assumed that a difference in morphology and functional activity of the bone marrow stromal populations before the transplant could sufficiently influence engraftment kinetics of the donor hematopoietic cells.
The aim of this study was to investigate changes in functional characteristics of bone marrow stromal cells (BMSC) in the patients with pre-treated hematological diseases and evaluate their significance in the post-transplant reconstitution of hematopoiesis.
Materials and Methods
We have studied bone marrow samples from thirty-eight patients with acute leukemia (0 to 47 years old). Twenty healthy donors (13 to 52 years old) served as control group. Bone marrow was obtained by sternal punctures. Separation of mononuclear cells was performed by Ficoll density gradient (1,077 g/l) followed by explantation to culture flasks containing alpha-MEM (Biolot, Russia) supplemented with 20% of fetal calf serum and antibiotics (Penicillin, 100 U/ml, and Streptomycin, 100 U/ml, Gibco). Cell cultures were maintained in sterile closed dishes at 37 °С, 100% humidity and 5% СО2. All the culture variants were studied in duplicate. To evaluate proliferative characteristics of BMSC, we have counted fibroblastic colony-forming units (CFU-F). After 10 days of culture, the cell colonies were fixed with 96% ethanol for 30 min, stained by Romanovsky-Giemsa (1:10 dilution) for another 30 min. The fibroblastic colonies were classified into large (>20 cells) and small ones (<20 cells).
To induce osteogenic and adipogenic differentiation of BMSCs, a standard conditioning medium was discarded at days 7 to 10 of primary cultures, and adherent cells were supplemented with fresh culture medium, or with the same medium supplied with differentiation-inducing agents. Osteogenic differentiation was achieved by addition of-glycerophosphate (7×10-3 М); dexamethasone (1×10-8 М); ascorbic acid (2×10-4 М) (Sigma, St Louis, MO, USA). Adipogenic differentiation of the BMSC was induced by dexamethasone (1×10-7 М); insulin (1×10-9 М). To detect osteocytic differentiation, the adherent cell layer was stained according to von Kossa method, or with Alizarin Red stain (Sigma, St Louis, MO, USA). Osteocytes were identified by typical calcium inclusions (black or red colour, respectively). To visualize adipocytic differentiation in cultured cell populations, the adherent fraction was fixed with formol vapor for 10 min., and incubated in the working Sudan Red O solution (Sigma- Aldrich, St Louis, MO, USA) for 1 hour at room temperature. Adipocytes were identified by orange lipid inclusions. In vitro hematopoietic support produced by BMSCs was evaluated in agar drop liquid culture system [1]. A monolayer of adherent marrow cells was used as a source of colony- stimulating factors. Bone marrow mononuclears from healthy donors were used as target cells. The cell colonies (CFU-GM) were counted at day 7 of culture. Colony-forming activity (CFA) and cluster-forming ability (ClFA) was assessed by colony numbers per culture and their sizes, i.e., small colonies, 20 to 40 cells; medium colonies, 41-100 cells; large colonies, >100 cells. Big cell clusters contained 10 to 19 cells; small clusters, 5-9 cells.
In parallel to the mentioned test system, hemostimulating activity of BMSC co-cultured with bone marrow mononuclears was tested in a liquid phase. The average term of coculture was 10-12 days. In order to analyze differentiation potential of hematopoietic progenitors, the colony-forming assays were performed in 100 μL Iscove’s modified Dulbecco’s medium (IMDM) plus 2% fetal bovine serum (Biolot, Russia), then added to 1 mL methylcellulose medium containing a cocktail of recombinant human stem cell factor, granulocyte-macrophage colony-stimulating factor, interleukin- 3, (MethoCult H4534, Stem Cell Technologies) before and after the co-culture initiation. The cells plated in 35-mm Petri dishes were cultured in a fully humidified environment with 5% CO2 at 37°C for 14 days.
Moreover, we measured expression of several genes responsible for cell adherence (SELECTIN, CXCR4, VCAM and PECAM). To this purpose, we performed reverse transcription of mRNAs from cell lysates. Real-time PCR with gene-specific primers was carried out in a standard reaction mix (Syntol, Russia). The post-PCR amplicon amounts were evaluated by means of a DNA-binding SYBR Green dye. The relative gene expression was normalized for a reference ABL gene.
Statistical evaluation was performed with STATISTICA software, version 6.0. In cases of statistically significant differences, we performed pairwise comparisons of the series using non-parametric Mann-Whitney criterion for two independent samples. The correlation quotients (r) were calculated using Spearman criterion. Statistical significance between experimental series was proven at confidence levels of p<0.05.
Results and Discussion
Comparison of BMSCs functional characteristics in patients and healthy donors A comparative assessment of fibroblast colony-forming units (CFU-F) CM in healthy donors and patients with acute leukemia shows a significant increase in big fibroblastic colonies of AML and ALL patients (p <0.05). (Fig. 1) These changes in proliferative ability of recipient stromal cells may occur due to the influence of malignant cell clones [18], or by previous rounds of chemotherapy [17]. One should discern between these potential causal factors.
Meanwhile, the number of CFU-F capable for adipogenic and osteogenic differentiation in acute leukemia was also significantly higher (p <0.05) (Fig. 2). A typical fibroblastic CFU is shown in Fig. 3. These results are in accordance with data presented by Borojevic et al., who showed similar changes of osteogenic marker expression and alkaline phosphatase activity in stromal cells from MDS patients [3]. Moreover, we find higher numbers of adipogenic progenitors in stroma from leukemia patients (AML and ALL), thus, probably, exerting a negative impact upon normal hematopoiesis. These data were recently described also by Le et al. [9].
When testing hemostimulating activity of stromal cells in soft agar, we observed a significant increase in hematopoiesis-supportive ability by stromal cells in acute myeloid leukemia (AML) (p <0.05), compared with healthy donors, whereas BMSC in patients with acute lymphoblastic leukemia, did not show similar properties (Fig. 4). This finding may suggest a possible role of stromal microenvironment in supporting malignant cell clones, as described elsewhere [13].
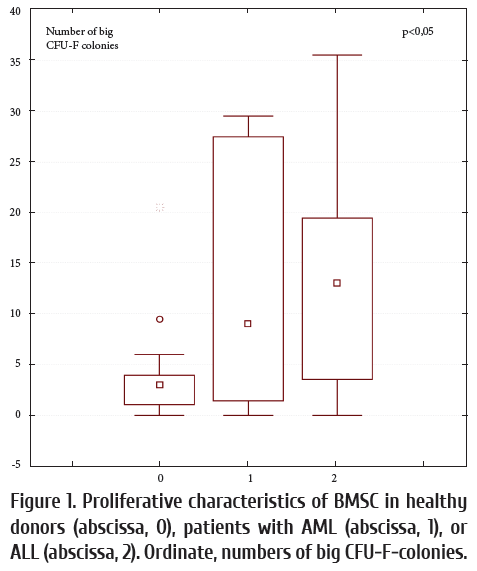
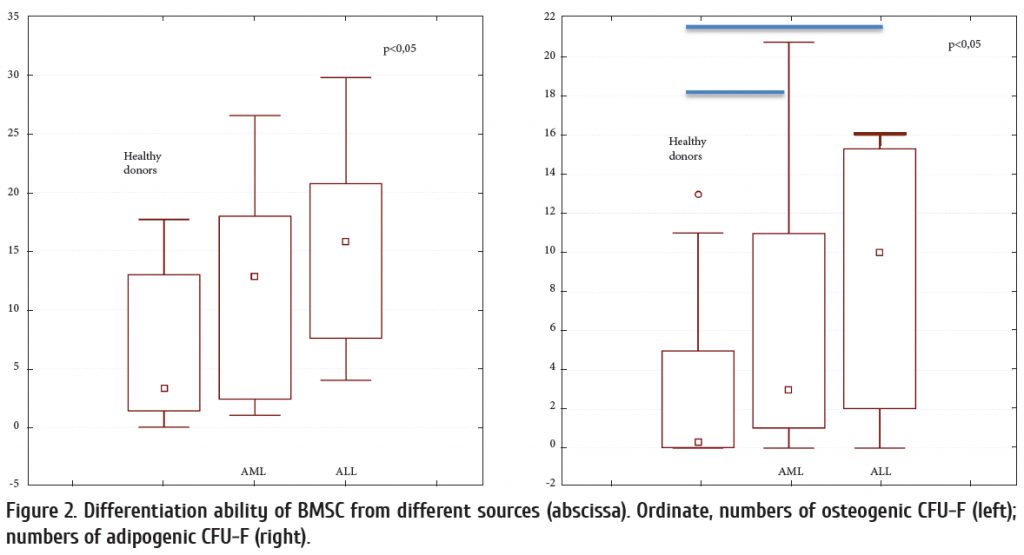
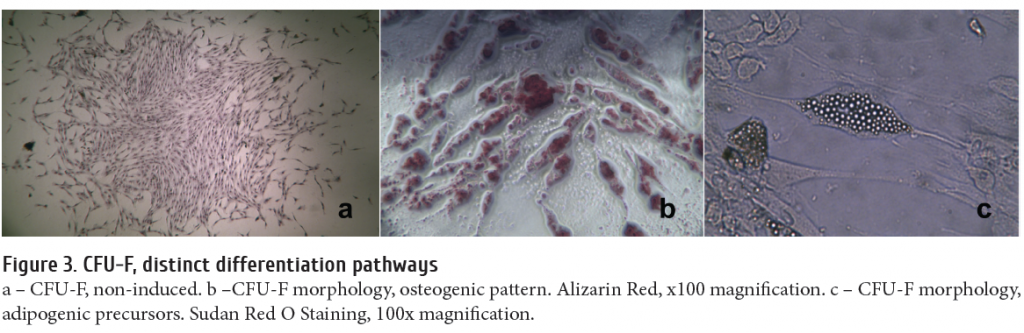
We have also revealed significant differences concerning expression of genes controlling adherence of hematopoietic cells to their substrates in bone marrow. We have found that stromal cells from AL patients exhibited higher expression of SELECTIN and VECAM genes in comparison to BMSC from healthy donors (p=0.008 and p=0.04, respectively, data not shown) in 17 patients with acute leukemia and healthy donors. Noteworthy, similar assays of PECAM and CXCR4 expression levels did not reveal such differences between stromal cells from AL patients and normal donors. However,
we have found a trend for decrease in CXCR4 gene expression among older leukemia patients (p=0.05). It should be noted that changed expression of adherence factors may be important over post-transplant period since they determine both engraftment kinetics of donor cells and graft functioning at later terms [5].
Role of BMSC supportive characteristics in hematopoietic engraftment kinetics
When analyzing a potentially predictive role of BMSC functional characteristics for the graft reconstitution, we have revealed a higher in vitro colony-forming ability of recipient BMSCs associated with longer terms of donor cell engraftment. A direct correlation was found between the large and small CFU-F colony numbers and leukocyte recovery terms (>1.0 peripheral leukocytes/μL), i.e., R = 0.34, p = 0.00006 and R = 0.23, p = 0.006, respectively. A similar trend was observed, when assessing connections with blood neutrophil recovery (R= 0.28, p = 0.001 and R = 0.21, p = 0.012, respectively, for large and small CFU-F colonies, data not shown).
Hence, when detecting in vitro hemostimulating activity of recipient stromal cells in comparison to in vivo hematopoietic recovery, we found a direct correlation be tween the number of CFU-GM colonies in soft agar cultures, and the recovery terms for leukocytes and platelets (> 20/L), R = 0.58 and R = 0.48, respectively. Meanwhile, analysis of colony formation in methylcellulose culture has shown an inverse correlation, i.e., shorter engraftment terms for blood leukocytes and platelets, with increasing support of colony-forming ability by stromal BMSCs derived from the recipient. Hence, it may indicate effects of short-distance factors promoting hematopoietic reconstitution after transplantation which may effectively support hematopoiesis in liquid culture but not in soft
agar. At the same time, while the higher hematopoietic progenitor colony formation in methylcellulose correlated with increased probability of post-transplant relapse (p <0.05). In the culture system “drop-agar liquid medium” such patterns have not been identified (Fig. 5), indicating that the contribution relapse in expectation of the same factors.
Moreover, we studied some relationships between quantitative parameters of osteogenic and adipogenic CFU-F, and the rates of hematopoietic engraftment. We have shown that the recovery time for leukocytes and platelets is reduced in the patients with higher in vitro osteogenic
potential of their CFU-F (p = 0,034), as well as a significantly reduced probability of graft hypofunction in cases of a higher number of adipogenic CFU-Fs in recipient stromal populations (p <0.05) (Fig. 6). A number of studies have shown that adipocytes act primarily as a negative regulator of hematopoiesis and are able to lengthen the periods of post-transplant reconstitution of hematopoiesis [12]. In turn, osteoblasts are seen as one of the key components of the hematopoietic microenvironment, regulating adhesion, homing and engraftment of HSCs during post-transplant period. There is the evidence of a leukemic clone inhibition by osteoblasts, thus creating favorable conditions for normal hematopoiesis [8].
When assessing a probability of acute graft-versus-host disease (aGvHD), we have shown that a higher risk of this common HSCT complication was associated with increased proliferative capacity characteristics of the recipient stromal cells, as expressed by CFU-F growth, and higher relative contents of large CFU-F colonies (>100 cells). Interestingly, despite our data, which point to hemostimulating ability of osteoblast progenitors and their role in faster post-transplant recovery, we have not received data which suggest higher risk of relapse in cases of increased osteogenic CFU-F numbers (p >0.05) There is also an association between increased numbers of osteogenic CFU-F and risk of aGvHD development (p >0.05) (Fig. 7).
Another evidence concerns mRNA expression of adhesion molecules (Selectin, Pecam, VCAM and CXCR4) in BMSCs. We have revealed shorter engraftment terms for leukocytes and platelets in the patients with increased mRNA expression of adhesion molecules in patient BMSC (p <0.05) (data not shown). However, the increase of these markers expression was associated with a higher probability of relapse and reduced post-transplant chimerism stability (p <0.05) (Fig. 8).
Conclusions
- Our findigs indicate the presence of higher proliferative activity and hemostimulating ability of BMSC in acute leukemia patients compared with healthy donors
- These biological effects of stroma may be ascribed to preceding chemotherapy and myelosuppression which affected stromal cells, or its interaction with the tumor clone.
- Functional characteristics of patients’ stromal cells may influence on posttransplant hematopoiesis recovery. Thus increase of the osteogenic differentiation is associated with a faster recovery of hematopoiesis, whereas adipogenic differentiation is associated with less probability of graft hypofunction. Some short-range haemostimulating factors may be responsible for such effect.
- Increased pre-transplant expression of some genes controlling HSC-homing molecules in recipient stroma is associated with sooner hematopoietic recovery, however, with higher occurence of post-transplant relapses.
Acknowledgement
The study was supported by The Russian Foundation for Basic Studies, Grant № 15-04-08679
References
- Afanasyev BV, Kulibaba TG, Zabelina TS, Lukasheva TN, Smirnova GA. Agar cloning of hematopoietic cells from the patients with different forms of hematopoietic dysplasia (clinics vs culture). Ter Arkhiv. 1982; 8:97-103.
- Blau O, Hofmann WK, Baldus CD, Thiel G, Serbent V, Schümann E, Thiel E, Blau IW.. Chromosomal aberrations in bone marrow mesenchymal stroma cells from patients with myelodysplastic syndrome and acute myeloblastic leukemia. Exp Hematol. 2007; 35 (2):221-229.
- Borojevic R, Roela RA, Rodarte RS, Thiago LS, Pasini FS, Conti FM, Rossi MI, Reis LF, Lopes LF, Brentani MM. Bone marrow stroma in childhood myelodysplastic syndrome: Composition, ability to sustain hematopoiesis in vitro, and altered gene expression. Leuk Res. 2004; 28 (8):831-844.
- Calvi LM, Adams GB, Weibrecht KW, Weber JM, Olson DP, Knight MC, Martin RP, Schipani E, Divieti P, Bringhurst FR, Milner LA, Kronenberg HM, Scadden DT. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature. 2003; 425 (6960):841-846.
- Chamberlain G, Fox J, Ashton B, Middleton J. Concise review: mesenchymal stem cells: their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells. 2007; 25 (11):2739-2749.
- Flores-Figueroa E, Arana-Trejo RM, Gutiérrez-Espíndola G, Pérez-Cabrera A, Mayani H. Mesenchymal stem cells in myelodysplastic syndromes: Phenotypic and cytogenetic characterization. Leuk Res. 2005; 29 (2):215-224.
- Konopleva M, Tabe Y, Zeng Z, Andreeff M. Therapeutic targeting of microenvironmental interactions in leukemia: Mechanisms and approaches. Drug Resist Updat. 2009; 12 (4-5):103-113.
- Krevvata M, Silva BC, Manavalan JS, Galan-Diez M, Kode A, Matthews BG, Park D, Zhang CA, Galili N, Nickolas TL, Dempster DW, Dougall WJ T-F. Inhibition of leukemia cell engraftment and disease progression in mice by osteoblasts. Blood. 2014; 124 (18):2834-2846.
- Le Y, Fraineau S, Chandran P, Sabloff M, Brand M, Lavoie JR, Gagne R, Rosu-Myles M, Yauk CL, Richardson RB, Allan DS. Adipogenic mesenchymal stromal cells from bone marrow and their hematopoietic supportive role: towards understanding the permissive marrow microenvironment in acute myeloid leukemia. Stem Cell Rev Reports. 2016; 12 (2):235-244.
- Le Blanc K. Mesenchymal stromal cells: Tissue repair and immune modulation. Cytotherapy. 2006; 8 (6):559-561.
- Le Blanc K, Samuelsson H, Gustafsson B, Remberger M, Sundberg B, Arvidson J, Ljungman P, Lönnies H, Nava S, Ringdén O. Transplantation of mesenchymal stem cells to enhance engraftment of hematopoietic stem cells. Leukemia. 2007; 21 (8):1733-1738.
- Naveiras O, Nardi V, Wenzel PL, Hauschka PV, Fahey F, Daley GQ. Bone-marrow adipocytes as negative regulators of the haematopoietic microenvironment. Nature. 2009; 460 (7252):259-263.
- Podar K, Richardson PG, Hideshima T, Chauhan D, Anderson KC. The malignant clone and the bone-marrow environment. Best Pract Res Clin Haematol. 2007; 20 (4):597-612.
- Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;4;276(5309):71-74.
- Rieger K, Marinets O, Fietz T, Körper S, Sommer D, Mücke C, Reufi B, Blau WI, Thiel E, Knauf WU. Mesenchymal stem cells remain of host origin even a long time after allogeneic peripheral blood stem cell or bone marrow transplantation. Exp Hematol. 2005; 33 (5):605-611.
- Scadden DT. The stem cell niche in health and leukemic disease. Best Pr Res Clin Haematol. 2007; 20 (1):19-27.
- Shipounova IN, Petinati NA, Bigildeev AE, Drize NJ, Sorokina TV, Kuzmina LA, Parovichnikova EN, Savchenko VG. Alterations of the bone marrow stromal microenvironment in adult patients with acute myeloid and lymphoblastic leukemias before and after allogeneic hematopoietic stem cell transplantation. Leuk Lymphoma. 2017; 58 (2):408-417.
- Yang G-C, Xu Y-H, Chen H-X, Wang X-J. Acute Lymphoblastic Leukemia Cells Inhibit the Differentiation of Bone Mesenchymal Stem Cells into Osteoblasts In Vitro by Activating Notch Signaling. Stem Cells Int. 2015; 2015:1-11.
- Zhang J, Niu C, Ye L, Huang H, He X, Tong WG, Ross J, Haug J, Johnson T, Feng JQ, Harris S, Wiedemann LM, Mishina Y, Li L. Identification of the haematopoietic stem cell niche and control of the niche size. Nature. 2003; 425:836-841.
" ["~DETAIL_TEXT"]=> string(23125) "
Introduction
Morphological and functional patterns of malignant cells are largely determined by specific somatic gene aberrations, which represent the major factor in biology of oncohematological disorders. Meanwhile, some data suggest expression of tumor markers in non-hematopoietic bone marrow cells from the stromal microenvironment [2, 6]. Hence, potential mechanisms of leukemogenesis, like as specific gene mutations, may include certain effects of cellular environment which may promote proliferation and development of hematopoietic stem cells [7, 16]. The so-called bone marrow niches are represented by endothelium, stromal cells, macrophages etc. The role of bone marrow stromal cells (BMSC) in hematopoietic regulation was first described by A.Ya Friedenstein fifty years ago. There are several types of stromal cells, in particular, mesenchymal stem cells (MSC) which are best studied for their potential clinical applications. MSCs are shown to suppress immune conflicts in allogeneic transplantation [10]; to replace and restore functions of the damaged non-hematopoietic tissues [14], support hematopoiesis when co-transplanted with hematopoietic stem cells (HSCs) [11]. These effects are based on favorable actions upon proliferation and differentiation of different hematopoietic lineages, e.g., myeloid and lymphoid progenitors. Hence, studies of stromal cells seem to be quite rational in view of their possible involvement into pathogenesis of oncohematological disorders.
It is known that, despite successful engraftment of donor-derived hematopoietic stem cells after allogeneic stem cell transplantation (alloSCT), the mesenchymal stem cells, are, in general, of host origin, even years post-transplant [15]. Some patients after alloSCT do not recover their stem cells despite receiving large amounts of CD34+ blood progenitors. Possible basis for the graft failure may be due to affection of hematopoietic niche by conditioning chemotherapy which could disturb its supportive (nursing) capacity. A number of researchers have identified the cells of osteoblast lineage to be a key cellular component of the HSC niche. These cells are responsible for bone formation [4, 19]. In addition, some transplant-associated factors might affect the osteoblasts, along with direct driving of hematopoiesis. Thus, it may be assumed that a difference in morphology and functional activity of the bone marrow stromal populations before the transplant could sufficiently influence engraftment kinetics of the donor hematopoietic cells.
The aim of this study was to investigate changes in functional characteristics of bone marrow stromal cells (BMSC) in the patients with pre-treated hematological diseases and evaluate their significance in the post-transplant reconstitution of hematopoiesis.
Materials and Methods
We have studied bone marrow samples from thirty-eight patients with acute leukemia (0 to 47 years old). Twenty healthy donors (13 to 52 years old) served as control group. Bone marrow was obtained by sternal punctures. Separation of mononuclear cells was performed by Ficoll density gradient (1,077 g/l) followed by explantation to culture flasks containing alpha-MEM (Biolot, Russia) supplemented with 20% of fetal calf serum and antibiotics (Penicillin, 100 U/ml, and Streptomycin, 100 U/ml, Gibco). Cell cultures were maintained in sterile closed dishes at 37 °С, 100% humidity and 5% СО2. All the culture variants were studied in duplicate. To evaluate proliferative characteristics of BMSC, we have counted fibroblastic colony-forming units (CFU-F). After 10 days of culture, the cell colonies were fixed with 96% ethanol for 30 min, stained by Romanovsky-Giemsa (1:10 dilution) for another 30 min. The fibroblastic colonies were classified into large (>20 cells) and small ones (<20 cells).
To induce osteogenic and adipogenic differentiation of BMSCs, a standard conditioning medium was discarded at days 7 to 10 of primary cultures, and adherent cells were supplemented with fresh culture medium, or with the same medium supplied with differentiation-inducing agents. Osteogenic differentiation was achieved by addition of-glycerophosphate (7×10-3 М); dexamethasone (1×10-8 М); ascorbic acid (2×10-4 М) (Sigma, St Louis, MO, USA). Adipogenic differentiation of the BMSC was induced by dexamethasone (1×10-7 М); insulin (1×10-9 М). To detect osteocytic differentiation, the adherent cell layer was stained according to von Kossa method, or with Alizarin Red stain (Sigma, St Louis, MO, USA). Osteocytes were identified by typical calcium inclusions (black or red colour, respectively). To visualize adipocytic differentiation in cultured cell populations, the adherent fraction was fixed with formol vapor for 10 min., and incubated in the working Sudan Red O solution (Sigma- Aldrich, St Louis, MO, USA) for 1 hour at room temperature. Adipocytes were identified by orange lipid inclusions. In vitro hematopoietic support produced by BMSCs was evaluated in agar drop liquid culture system [1]. A monolayer of adherent marrow cells was used as a source of colony- stimulating factors. Bone marrow mononuclears from healthy donors were used as target cells. The cell colonies (CFU-GM) were counted at day 7 of culture. Colony-forming activity (CFA) and cluster-forming ability (ClFA) was assessed by colony numbers per culture and their sizes, i.e., small colonies, 20 to 40 cells; medium colonies, 41-100 cells; large colonies, >100 cells. Big cell clusters contained 10 to 19 cells; small clusters, 5-9 cells.
In parallel to the mentioned test system, hemostimulating activity of BMSC co-cultured with bone marrow mononuclears was tested in a liquid phase. The average term of coculture was 10-12 days. In order to analyze differentiation potential of hematopoietic progenitors, the colony-forming assays were performed in 100 μL Iscove’s modified Dulbecco’s medium (IMDM) plus 2% fetal bovine serum (Biolot, Russia), then added to 1 mL methylcellulose medium containing a cocktail of recombinant human stem cell factor, granulocyte-macrophage colony-stimulating factor, interleukin- 3, (MethoCult H4534, Stem Cell Technologies) before and after the co-culture initiation. The cells plated in 35-mm Petri dishes were cultured in a fully humidified environment with 5% CO2 at 37°C for 14 days.
Moreover, we measured expression of several genes responsible for cell adherence (SELECTIN, CXCR4, VCAM and PECAM). To this purpose, we performed reverse transcription of mRNAs from cell lysates. Real-time PCR with gene-specific primers was carried out in a standard reaction mix (Syntol, Russia). The post-PCR amplicon amounts were evaluated by means of a DNA-binding SYBR Green dye. The relative gene expression was normalized for a reference ABL gene.
Statistical evaluation was performed with STATISTICA software, version 6.0. In cases of statistically significant differences, we performed pairwise comparisons of the series using non-parametric Mann-Whitney criterion for two independent samples. The correlation quotients (r) were calculated using Spearman criterion. Statistical significance between experimental series was proven at confidence levels of p<0.05.
Results and Discussion
Comparison of BMSCs functional characteristics in patients and healthy donors A comparative assessment of fibroblast colony-forming units (CFU-F) CM in healthy donors and patients with acute leukemia shows a significant increase in big fibroblastic colonies of AML and ALL patients (p <0.05). (Fig. 1) These changes in proliferative ability of recipient stromal cells may occur due to the influence of malignant cell clones [18], or by previous rounds of chemotherapy [17]. One should discern between these potential causal factors.
Meanwhile, the number of CFU-F capable for adipogenic and osteogenic differentiation in acute leukemia was also significantly higher (p <0.05) (Fig. 2). A typical fibroblastic CFU is shown in Fig. 3. These results are in accordance with data presented by Borojevic et al., who showed similar changes of osteogenic marker expression and alkaline phosphatase activity in stromal cells from MDS patients [3]. Moreover, we find higher numbers of adipogenic progenitors in stroma from leukemia patients (AML and ALL), thus, probably, exerting a negative impact upon normal hematopoiesis. These data were recently described also by Le et al. [9].
When testing hemostimulating activity of stromal cells in soft agar, we observed a significant increase in hematopoiesis-supportive ability by stromal cells in acute myeloid leukemia (AML) (p <0.05), compared with healthy donors, whereas BMSC in patients with acute lymphoblastic leukemia, did not show similar properties (Fig. 4). This finding may suggest a possible role of stromal microenvironment in supporting malignant cell clones, as described elsewhere [13].



We have also revealed significant differences concerning expression of genes controlling adherence of hematopoietic cells to their substrates in bone marrow. We have found that stromal cells from AL patients exhibited higher expression of SELECTIN and VECAM genes in comparison to BMSC from healthy donors (p=0.008 and p=0.04, respectively, data not shown) in 17 patients with acute leukemia and healthy donors. Noteworthy, similar assays of PECAM and CXCR4 expression levels did not reveal such differences between stromal cells from AL patients and normal donors. However,

we have found a trend for decrease in CXCR4 gene expression among older leukemia patients (p=0.05). It should be noted that changed expression of adherence factors may be important over post-transplant period since they determine both engraftment kinetics of donor cells and graft functioning at later terms [5].
Role of BMSC supportive characteristics in hematopoietic engraftment kinetics
When analyzing a potentially predictive role of BMSC functional characteristics for the graft reconstitution, we have revealed a higher in vitro colony-forming ability of recipient BMSCs associated with longer terms of donor cell engraftment. A direct correlation was found between the large and small CFU-F colony numbers and leukocyte recovery terms (>1.0 peripheral leukocytes/μL), i.e., R = 0.34, p = 0.00006 and R = 0.23, p = 0.006, respectively. A similar trend was observed, when assessing connections with blood neutrophil recovery (R= 0.28, p = 0.001 and R = 0.21, p = 0.012, respectively, for large and small CFU-F colonies, data not shown).
Hence, when detecting in vitro hemostimulating activity of recipient stromal cells in comparison to in vivo hematopoietic recovery, we found a direct correlation be tween the number of CFU-GM colonies in soft agar cultures, and the recovery terms for leukocytes and platelets (> 20/L), R = 0.58 and R = 0.48, respectively. Meanwhile, analysis of colony formation in methylcellulose culture has shown an inverse correlation, i.e., shorter engraftment terms for blood leukocytes and platelets, with increasing support of colony-forming ability by stromal BMSCs derived from the recipient. Hence, it may indicate effects of short-distance factors promoting hematopoietic reconstitution after transplantation which may effectively support hematopoiesis in liquid culture but not in soft

agar. At the same time, while the higher hematopoietic progenitor colony formation in methylcellulose correlated with increased probability of post-transplant relapse (p <0.05). In the culture system “drop-agar liquid medium” such patterns have not been identified (Fig. 5), indicating that the contribution relapse in expectation of the same factors.
Moreover, we studied some relationships between quantitative parameters of osteogenic and adipogenic CFU-F, and the rates of hematopoietic engraftment. We have shown that the recovery time for leukocytes and platelets is reduced in the patients with higher in vitro osteogenic

potential of their CFU-F (p = 0,034), as well as a significantly reduced probability of graft hypofunction in cases of a higher number of adipogenic CFU-Fs in recipient stromal populations (p <0.05) (Fig. 6). A number of studies have shown that adipocytes act primarily as a negative regulator of hematopoiesis and are able to lengthen the periods of post-transplant reconstitution of hematopoiesis [12]. In turn, osteoblasts are seen as one of the key components of the hematopoietic microenvironment, regulating adhesion, homing and engraftment of HSCs during post-transplant period. There is the evidence of a leukemic clone inhibition by osteoblasts, thus creating favorable conditions for normal hematopoiesis [8].
When assessing a probability of acute graft-versus-host disease (aGvHD), we have shown that a higher risk of this common HSCT complication was associated with increased proliferative capacity characteristics of the recipient stromal cells, as expressed by CFU-F growth, and higher relative contents of large CFU-F colonies (>100 cells). Interestingly, despite our data, which point to hemostimulating ability of osteoblast progenitors and their role in faster post-transplant recovery, we have not received data which suggest higher risk of relapse in cases of increased osteogenic CFU-F numbers (p >0.05) There is also an association between increased numbers of osteogenic CFU-F and risk of aGvHD development (p >0.05) (Fig. 7).
Another evidence concerns mRNA expression of adhesion molecules (Selectin, Pecam, VCAM and CXCR4) in BMSCs. We have revealed shorter engraftment terms for leukocytes and platelets in the patients with increased mRNA expression of adhesion molecules in patient BMSC (p <0.05) (data not shown). However, the increase of these markers expression was associated with a higher probability of relapse and reduced post-transplant chimerism stability (p <0.05) (Fig. 8).

Conclusions
- Our findigs indicate the presence of higher proliferative activity and hemostimulating ability of BMSC in acute leukemia patients compared with healthy donors
- These biological effects of stroma may be ascribed to preceding chemotherapy and myelosuppression which affected stromal cells, or its interaction with the tumor clone.
- Functional characteristics of patients’ stromal cells may influence on posttransplant hematopoiesis recovery. Thus increase of the osteogenic differentiation is associated with a faster recovery of hematopoiesis, whereas adipogenic differentiation is associated with less probability of graft hypofunction. Some short-range haemostimulating factors may be responsible for such effect.
- Increased pre-transplant expression of some genes controlling HSC-homing molecules in recipient stroma is associated with sooner hematopoietic recovery, however, with higher occurence of post-transplant relapses.
Acknowledgement
The study was supported by The Russian Foundation for Basic Studies, Grant № 15-04-08679
References
- Afanasyev BV, Kulibaba TG, Zabelina TS, Lukasheva TN, Smirnova GA. Agar cloning of hematopoietic cells from the patients with different forms of hematopoietic dysplasia (clinics vs culture). Ter Arkhiv. 1982; 8:97-103.
- Blau O, Hofmann WK, Baldus CD, Thiel G, Serbent V, Schümann E, Thiel E, Blau IW.. Chromosomal aberrations in bone marrow mesenchymal stroma cells from patients with myelodysplastic syndrome and acute myeloblastic leukemia. Exp Hematol. 2007; 35 (2):221-229.
- Borojevic R, Roela RA, Rodarte RS, Thiago LS, Pasini FS, Conti FM, Rossi MI, Reis LF, Lopes LF, Brentani MM. Bone marrow stroma in childhood myelodysplastic syndrome: Composition, ability to sustain hematopoiesis in vitro, and altered gene expression. Leuk Res. 2004; 28 (8):831-844.
- Calvi LM, Adams GB, Weibrecht KW, Weber JM, Olson DP, Knight MC, Martin RP, Schipani E, Divieti P, Bringhurst FR, Milner LA, Kronenberg HM, Scadden DT. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature. 2003; 425 (6960):841-846.
- Chamberlain G, Fox J, Ashton B, Middleton J. Concise review: mesenchymal stem cells: their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells. 2007; 25 (11):2739-2749.
- Flores-Figueroa E, Arana-Trejo RM, Gutiérrez-Espíndola G, Pérez-Cabrera A, Mayani H. Mesenchymal stem cells in myelodysplastic syndromes: Phenotypic and cytogenetic characterization. Leuk Res. 2005; 29 (2):215-224.
- Konopleva M, Tabe Y, Zeng Z, Andreeff M. Therapeutic targeting of microenvironmental interactions in leukemia: Mechanisms and approaches. Drug Resist Updat. 2009; 12 (4-5):103-113.
- Krevvata M, Silva BC, Manavalan JS, Galan-Diez M, Kode A, Matthews BG, Park D, Zhang CA, Galili N, Nickolas TL, Dempster DW, Dougall WJ T-F. Inhibition of leukemia cell engraftment and disease progression in mice by osteoblasts. Blood. 2014; 124 (18):2834-2846.
- Le Y, Fraineau S, Chandran P, Sabloff M, Brand M, Lavoie JR, Gagne R, Rosu-Myles M, Yauk CL, Richardson RB, Allan DS. Adipogenic mesenchymal stromal cells from bone marrow and their hematopoietic supportive role: towards understanding the permissive marrow microenvironment in acute myeloid leukemia. Stem Cell Rev Reports. 2016; 12 (2):235-244.
- Le Blanc K. Mesenchymal stromal cells: Tissue repair and immune modulation. Cytotherapy. 2006; 8 (6):559-561.
- Le Blanc K, Samuelsson H, Gustafsson B, Remberger M, Sundberg B, Arvidson J, Ljungman P, Lönnies H, Nava S, Ringdén O. Transplantation of mesenchymal stem cells to enhance engraftment of hematopoietic stem cells. Leukemia. 2007; 21 (8):1733-1738.
- Naveiras O, Nardi V, Wenzel PL, Hauschka PV, Fahey F, Daley GQ. Bone-marrow adipocytes as negative regulators of the haematopoietic microenvironment. Nature. 2009; 460 (7252):259-263.
- Podar K, Richardson PG, Hideshima T, Chauhan D, Anderson KC. The malignant clone and the bone-marrow environment. Best Pract Res Clin Haematol. 2007; 20 (4):597-612.
- Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;4;276(5309):71-74.
- Rieger K, Marinets O, Fietz T, Körper S, Sommer D, Mücke C, Reufi B, Blau WI, Thiel E, Knauf WU. Mesenchymal stem cells remain of host origin even a long time after allogeneic peripheral blood stem cell or bone marrow transplantation. Exp Hematol. 2005; 33 (5):605-611.
- Scadden DT. The stem cell niche in health and leukemic disease. Best Pr Res Clin Haematol. 2007; 20 (1):19-27.
- Shipounova IN, Petinati NA, Bigildeev AE, Drize NJ, Sorokina TV, Kuzmina LA, Parovichnikova EN, Savchenko VG. Alterations of the bone marrow stromal microenvironment in adult patients with acute myeloid and lymphoblastic leukemias before and after allogeneic hematopoietic stem cell transplantation. Leuk Lymphoma. 2017; 58 (2):408-417.
- Yang G-C, Xu Y-H, Chen H-X, Wang X-J. Acute Lymphoblastic Leukemia Cells Inhibit the Differentiation of Bone Mesenchymal Stem Cells into Osteoblasts In Vitro by Activating Notch Signaling. Stem Cells Int. 2015; 2015:1-11.
- Zhang J, Niu C, Ye L, Huang H, He X, Tong WG, Ross J, Haug J, Johnson T, Feng JQ, Harris S, Wiedemann LM, Mishina Y, Li L. Identification of the haematopoietic stem cell niche and control of the niche size. Nature. 2003; 425:836-841.
" ["DETAIL_TEXT_TYPE"]=> string(4) "html" ["~DETAIL_TEXT_TYPE"]=> string(4) "html" ["PREVIEW_TEXT"]=> string(0) "" ["~PREVIEW_TEXT"]=> string(0) "" ["PREVIEW_TEXT_TYPE"]=> string(4) "text" ["~PREVIEW_TEXT_TYPE"]=> string(4) "text" ["PREVIEW_PICTURE"]=> NULL ["~PREVIEW_PICTURE"]=> NULL ["LANG_DIR"]=> string(4) "/ru/" ["~LANG_DIR"]=> string(4) "/ru/" ["SORT"]=> string(3) "500" ["~SORT"]=> string(3) "500" ["CODE"]=> string(100) "vliyanie-funktsionalnykh-kharakteristik-stromalnykh-kletok-kostnogo-mozga-retsipienta-na-prizhivleni" ["~CODE"]=> string(100) "vliyanie-funktsionalnykh-kharakteristik-stromalnykh-kletok-kostnogo-mozga-retsipienta-na-prizhivleni" ["EXTERNAL_ID"]=> string(3) "700" ["~EXTERNAL_ID"]=> string(3) "700" ["IBLOCK_TYPE_ID"]=> string(7) "journal" ["~IBLOCK_TYPE_ID"]=> string(7) "journal" ["IBLOCK_CODE"]=> string(7) "volumes" ["~IBLOCK_CODE"]=> string(7) "volumes" ["IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["~IBLOCK_EXTERNAL_ID"]=> string(1) "2" ["LID"]=> string(2) "s2" ["~LID"]=> string(2) "s2" ["EDIT_LINK"]=> NULL ["DELETE_LINK"]=> NULL ["DISPLAY_ACTIVE_FROM"]=> string(0) "" ["IPROPERTY_VALUES"]=> array(18) { ["ELEMENT_META_TITLE"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["ELEMENT_META_KEYWORDS"]=> string(189) "Стромальные клетки костного мозга острые лейкозы функциональные характеристики типы дифференцировки" ["ELEMENT_META_DESCRIPTION"]=> string(310) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСКImpact of recipient marrow stromal cells functional characteristics on engraftment after allo-HSCT" ["ELEMENT_PREVIEW_PICTURE_FILE_ALT"]=> string(2500) "Наблюдаемая в ряде случаев гипофункция транс- плантата является нередкой и обусловливает не- обходимость разработки функциональных тестов исследования клеток стромы с целью обоснования показаний к совместной трансплантации гемопоэ- тических и стромальных клеток костного мозга. Це- лью данного исследования являлось изучение роли стромальных клеток костного мозга в процессе при- живления донорского костного мозга и их значения в развитии осложнений после ТГСК. При анализе функциональных характеристик стромальных кле- ток у 38 реципиентов аллогенного трансплантата ге- мопоэтических стволовых клеток было показано уве- личение активности остеогенной дифференцировки на фоне укорочения сроков восстановления кро- ветворения. Вместе с тем, наличие более выражен- ной способности к адипогенной дифференцировке ассоциировано с меньшей вероятностью развития гипофункции трансплантата. При исследовании ге- мостимулирующей активности стромальных клеток реципиента были отмечены факты, указывающие на преимущественное влияние короткодистантных ростовых факторов на посттрансплантационную реконституцию гемопоэза. При этом более выра- женная экспрессия молекул, опосредующих хоу- минг ГСК в стромальных клетках реципиента перед трансплантацией ассоциировано с более быстрым восстановлением гемопоэза у трансплантированных больных." ["ELEMENT_PREVIEW_PICTURE_FILE_TITLE"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["ELEMENT_DETAIL_PICTURE_FILE_ALT"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["ELEMENT_DETAIL_PICTURE_FILE_TITLE"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["SECTION_META_TITLE"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["SECTION_META_KEYWORDS"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["SECTION_META_DESCRIPTION"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["SECTION_PICTURE_FILE_ALT"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["SECTION_PICTURE_FILE_TITLE"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["SECTION_PICTURE_FILE_NAME"]=> string(100) "vliyanie-funktsionalnykh-kharakteristik-stromalnykh-kletok-kostnogo-mozga-retsipienta-na-prizhivleni" ["SECTION_DETAIL_PICTURE_FILE_ALT"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["SECTION_DETAIL_PICTURE_FILE_TITLE"]=> string(212) "Влияние функциональных характеристик стромальных клеток костного мозга реципиента на приживление после алло-ТГСК" ["SECTION_DETAIL_PICTURE_FILE_NAME"]=> string(100) "vliyanie-funktsionalnykh-kharakteristik-stromalnykh-kletok-kostnogo-mozga-retsipienta-na-prizhivleni" ["ELEMENT_PREVIEW_PICTURE_FILE_NAME"]=> string(100) "vliyanie-funktsionalnykh-kharakteristik-stromalnykh-kletok-kostnogo-mozga-retsipienta-na-prizhivleni" ["ELEMENT_DETAIL_PICTURE_FILE_NAME"]=> string(100) "vliyanie-funktsionalnykh-kharakteristik-stromalnykh-kletok-kostnogo-mozga-retsipienta-na-prizhivleni" } ["FIELDS"]=> array(1) { ["IBLOCK_SECTION_ID"]=> string(2) "21" } ["PROPERTIES"]=> array(18) { ["KEYWORDS"]=> array(36) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(4) { [0]=> string(4) "8474" [1]=> string(4) "8475" [2]=> string(4) "8476" [3]=> string(4) "8477" } ["VALUE"]=> array(4) { [0]=> string(3) "696" [1]=> string(3) "697" [2]=> string(3) "698" [3]=> string(3) "699" } ["DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(4) { [0]=> string(3) "696" [1]=> string(3) "697" [2]=> string(3) "698" [3]=> string(3) "699" } ["~DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" } ["SUBMITTED"]=> array(36) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8445" ["VALUE"]=> string(22) "10/10/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "10/10/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL } ["ACCEPTED"]=> array(36) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8446" ["VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL } ["PUBLISHED"]=> array(36) { ["ID"]=> string(2) "22" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Дата публикации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "PUBLISHED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "22" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Дата публикации" ["~DEFAULT_VALUE"]=> NULL } ["CONTACT"]=> array(36) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8447" ["VALUE"]=> string(3) "695" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "695" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHORS"]=> array(36) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(1) { [0]=> string(4) "8478" } ["VALUE"]=> array(1) { [0]=> string(3) "695" } ["DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(1) { [0]=> string(3) "695" } ["~DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_RU"]=> array(36) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8449" ["VALUE"]=> array(2) { ["TEXT"]=> string(327) "Ильдар М. Бархатов, Николай Ю. Цветков, Даниил Е. Ершов, Марина Ю. Лаврухина, Алена И. Шакирова, Алиса Я.<br> Поттер, Анна Г. Смирнова, Людмила С. Зубаровская, Борис В. Афанасьев<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(315) "Ильдар М. Бархатов, Николай Ю. Цветков, Даниил Е. Ершов, Марина Ю. Лаврухина, Алена И. Шакирова, Алиса Я.
Поттер, Анна Г. Смирнова, Людмила С. Зубаровская, Борис В. Афанасьев
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_RU"]=> array(36) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8450" ["VALUE"]=> array(2) { ["TEXT"]=> string(294) "Первый Санкт-Петербургский государственный медицинский Университет им. акад. И. П. Павлова Министерства<br> Здравоохранения РФ, Санкт-Петербург, Россия<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(282) "Первый Санкт-Петербургский государственный медицинский Университет им. акад. И. П. Павлова Министерства
Здравоохранения РФ, Санкт-Петербург, Россия
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_RU"]=> array(36) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8451" ["VALUE"]=> array(2) { ["TEXT"]=> string(2500) "Наблюдаемая в ряде случаев гипофункция транс- плантата является нередкой и обусловливает не- обходимость разработки функциональных тестов исследования клеток стромы с целью обоснования показаний к совместной трансплантации гемопоэ- тических и стромальных клеток костного мозга. Це- лью данного исследования являлось изучение роли стромальных клеток костного мозга в процессе при- живления донорского костного мозга и их значения в развитии осложнений после ТГСК. При анализе функциональных характеристик стромальных кле- ток у 38 реципиентов аллогенного трансплантата ге- мопоэтических стволовых клеток было показано уве- личение активности остеогенной дифференцировки на фоне укорочения сроков восстановления кро- ветворения. Вместе с тем, наличие более выражен- ной способности к адипогенной дифференцировке ассоциировано с меньшей вероятностью развития гипофункции трансплантата. При исследовании ге- мостимулирующей активности стромальных клеток реципиента были отмечены факты, указывающие на преимущественное влияние короткодистантных ростовых факторов на посттрансплантационную реконституцию гемопоэза. При этом более выра- женная экспрессия молекул, опосредующих хоу- минг ГСК в стромальных клетках реципиента перед трансплантацией ассоциировано с более быстрым восстановлением гемопоэза у трансплантированных больных." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2500) "Наблюдаемая в ряде случаев гипофункция транс- плантата является нередкой и обусловливает не- обходимость разработки функциональных тестов исследования клеток стромы с целью обоснования показаний к совместной трансплантации гемопоэ- тических и стромальных клеток костного мозга. Це- лью данного исследования являлось изучение роли стромальных клеток костного мозга в процессе при- живления донорского костного мозга и их значения в развитии осложнений после ТГСК. При анализе функциональных характеристик стромальных кле- ток у 38 реципиентов аллогенного трансплантата ге- мопоэтических стволовых клеток было показано уве- личение активности остеогенной дифференцировки на фоне укорочения сроков восстановления кро- ветворения. Вместе с тем, наличие более выражен- ной способности к адипогенной дифференцировке ассоциировано с меньшей вероятностью развития гипофункции трансплантата. При исследовании ге- мостимулирующей активности стромальных клеток реципиента были отмечены факты, указывающие на преимущественное влияние короткодистантных ростовых факторов на посттрансплантационную реконституцию гемопоэза. При этом более выра- женная экспрессия молекул, опосредующих хоу- минг ГСК в стромальных клетках реципиента перед трансплантацией ассоциировано с более быстрым восстановлением гемопоэза у трансплантированных больных." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["DOI"]=> array(36) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8452" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-71-77" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-71-77" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" } ["AUTHOR_EN"]=> array(36) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8453" ["VALUE"]=> array(2) { ["TEXT"]=> string(202) "Ildar M. Barkhatov, Nikolay Yu Tsvetkov, Daniil E. Ershov, Marina Y. Lavrukhina, Alena I. Shakirova, Alisa Ya. Potter,<br> Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(190) "Ildar M. Barkhatov, Nikolay Yu Tsvetkov, Daniil E. Ershov, Marina Y. Lavrukhina, Alena I. Shakirova, Alisa Ya. Potter,
Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["ORGANIZATION_EN"]=> array(36) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8454" ["VALUE"]=> array(2) { ["TEXT"]=> string(785) "R. M. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, and Chair of Hematology,<br> Transfusiology and Transplantation, The First St. Petersburg State I. Pavlov Medical University, St. Petersburg, Russian<br> Federation<br> <br> Dr. Ildar M.Barkhatov, PhD, Laboratory Head, R. M. Gorbacheva<br> Memorial Research Institute of Children Oncology,<br> Hematology and Transplantation, The First St. Petersburg<br> State I. Pavlov Medical University, L.Tolstoy St. 6/8, 197022,<br> St. Petersburg, Russian Federation<br> <br> Phone: +7(911) 778 2785<br> E-mail: <a href="mailto:i.barkhatov@gmail.com">i.barkhatov@gmail.com</a><br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(691) "R. M. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, and Chair of Hematology,
Transfusiology and Transplantation, The First St. Petersburg State I. Pavlov Medical University, St. Petersburg, Russian
Federation
Dr. Ildar M.Barkhatov, PhD, Laboratory Head, R. M. Gorbacheva
Memorial Research Institute of Children Oncology,
Hematology and Transplantation, The First St. Petersburg
State I. Pavlov Medical University, L.Tolstoy St. 6/8, 197022,
St. Petersburg, Russian Federation
Phone: +7(911) 778 2785
E-mail: i.barkhatov@gmail.com
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["SUMMARY_EN"]=> array(36) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8455" ["VALUE"]=> array(2) { ["TEXT"]=> string(1295) "Functional insufficiency of stem cell grafts is not rare in hematopoietic stem cell transplantation (HSCT), thus requiring further development of functional tests for recipient’s stromal cells, in order to substantiate clinical indications for co-transplantation of hematopoietic and stromal bone marrow cells. The aim of this study was to investigate a role of bone marrow stromal cells (BMSC) in the donor bone marrow engraftment, and their importance for development of complications after HSCT. Functional analysis of BMSC cultures from thirty-eight recipients of allogeneic hematopoietic stem cells has been shown that induced osteogenic differentiation correlated with faster hematopoiesis recovery in transplanted patients. Moreover, a pronounced ability for adipogenic differentiation proved to be associated with lower probability of the graft hypofunction. A pronounced hemostimulatory activity of stromal cells was observed in our study, thus suggesting effects of short-range hematopoietic growth factors from BMSC upon hematopoietic reconstitution. At the same time, a more pronounced pre-transplant expression of molecules which promote donor HSC homing by the recipient stromal cells was associated with faster hematopoietic recovery after transplantation." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(1295) "Functional insufficiency of stem cell grafts is not rare in hematopoietic stem cell transplantation (HSCT), thus requiring further development of functional tests for recipient’s stromal cells, in order to substantiate clinical indications for co-transplantation of hematopoietic and stromal bone marrow cells. The aim of this study was to investigate a role of bone marrow stromal cells (BMSC) in the donor bone marrow engraftment, and their importance for development of complications after HSCT. Functional analysis of BMSC cultures from thirty-eight recipients of allogeneic hematopoietic stem cells has been shown that induced osteogenic differentiation correlated with faster hematopoiesis recovery in transplanted patients. Moreover, a pronounced ability for adipogenic differentiation proved to be associated with lower probability of the graft hypofunction. A pronounced hemostimulatory activity of stromal cells was observed in our study, thus suggesting effects of short-range hematopoietic growth factors from BMSC upon hematopoietic reconstitution. At the same time, a more pronounced pre-transplant expression of molecules which promote donor HSC homing by the recipient stromal cells was associated with faster hematopoietic recovery after transplantation." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["NAME_EN"]=> array(36) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8456" ["VALUE"]=> string(98) "Impact of recipient marrow stromal cells functional characteristics on engraftment after allo-HSCT" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(98) "Impact of recipient marrow stromal cells functional characteristics on engraftment after allo-HSCT" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" } ["FULL_TEXT_RU"]=> array(36) { ["ID"]=> string(2) "42" ["TIMESTAMP_X"]=> string(19) "2015-09-07 20:29:18" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(23) "Полный текст" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(12) "FULL_TEXT_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "42" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(23) "Полный текст" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } } ["PDF_RU"]=> array(36) { ["ID"]=> string(2) "43" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF RUS" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_RU" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "43" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8479" ["VALUE"]=> string(3) "280" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "280" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(7) "PDF RUS" ["~DEFAULT_VALUE"]=> string(0) "" } ["PDF_EN"]=> array(36) { ["ID"]=> string(2) "44" ["TIMESTAMP_X"]=> string(19) "2015-09-09 16:05:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(7) "PDF ENG" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(6) "PDF_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "F" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "44" ["FILE_TYPE"]=> string(18) "doc, txt, rtf, pdf" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8480" ["VALUE"]=> string(3) "281" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "281" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(7) "PDF ENG" ["~DEFAULT_VALUE"]=> string(0) "" } ["NAME_LONG"]=> array(36) { ["ID"]=> string(2) "45" ["TIMESTAMP_X"]=> string(19) "2023-04-13 00:55:00" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(72) "Название (для очень длинных заголовков)" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "NAME_LONG" ["DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "45" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(80) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> NULL ["VALUE"]=> string(0) "" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(0) "" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(72) "Название (для очень длинных заголовков)" ["~DEFAULT_VALUE"]=> array(2) { ["TYPE"]=> string(4) "HTML" ["TEXT"]=> string(0) "" } } } ["DISPLAY_PROPERTIES"]=> array(13) { ["AUTHOR_EN"]=> array(37) { ["ID"]=> string(2) "37" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(6) "Author" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "37" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8453" ["VALUE"]=> array(2) { ["TEXT"]=> string(202) "Ildar M. Barkhatov, Nikolay Yu Tsvetkov, Daniil E. Ershov, Marina Y. Lavrukhina, Alena I. Shakirova, Alisa Ya. Potter,<br> Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(190) "Ildar M. Barkhatov, Nikolay Yu Tsvetkov, Daniil E. Ershov, Marina Y. Lavrukhina, Alena I. Shakirova, Alisa Ya. Potter,
Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(6) "Author" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(190) "Ildar M. Barkhatov, Nikolay Yu Tsvetkov, Daniil E. Ershov, Marina Y. Lavrukhina, Alena I. Shakirova, Alisa Ya. Potter,
Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev
" } ["SUMMARY_EN"]=> array(37) { ["ID"]=> string(2) "39" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Description / Summary" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "39" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8455" ["VALUE"]=> array(2) { ["TEXT"]=> string(1295) "Functional insufficiency of stem cell grafts is not rare in hematopoietic stem cell transplantation (HSCT), thus requiring further development of functional tests for recipient’s stromal cells, in order to substantiate clinical indications for co-transplantation of hematopoietic and stromal bone marrow cells. The aim of this study was to investigate a role of bone marrow stromal cells (BMSC) in the donor bone marrow engraftment, and their importance for development of complications after HSCT. Functional analysis of BMSC cultures from thirty-eight recipients of allogeneic hematopoietic stem cells has been shown that induced osteogenic differentiation correlated with faster hematopoiesis recovery in transplanted patients. Moreover, a pronounced ability for adipogenic differentiation proved to be associated with lower probability of the graft hypofunction. A pronounced hemostimulatory activity of stromal cells was observed in our study, thus suggesting effects of short-range hematopoietic growth factors from BMSC upon hematopoietic reconstitution. At the same time, a more pronounced pre-transplant expression of molecules which promote donor HSC homing by the recipient stromal cells was associated with faster hematopoietic recovery after transplantation." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(1295) "Functional insufficiency of stem cell grafts is not rare in hematopoietic stem cell transplantation (HSCT), thus requiring further development of functional tests for recipient’s stromal cells, in order to substantiate clinical indications for co-transplantation of hematopoietic and stromal bone marrow cells. The aim of this study was to investigate a role of bone marrow stromal cells (BMSC) in the donor bone marrow engraftment, and their importance for development of complications after HSCT. Functional analysis of BMSC cultures from thirty-eight recipients of allogeneic hematopoietic stem cells has been shown that induced osteogenic differentiation correlated with faster hematopoiesis recovery in transplanted patients. Moreover, a pronounced ability for adipogenic differentiation proved to be associated with lower probability of the graft hypofunction. A pronounced hemostimulatory activity of stromal cells was observed in our study, thus suggesting effects of short-range hematopoietic growth factors from BMSC upon hematopoietic reconstitution. At the same time, a more pronounced pre-transplant expression of molecules which promote donor HSC homing by the recipient stromal cells was associated with faster hematopoietic recovery after transplantation." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Description / Summary" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(1295) "Functional insufficiency of stem cell grafts is not rare in hematopoietic stem cell transplantation (HSCT), thus requiring further development of functional tests for recipient’s stromal cells, in order to substantiate clinical indications for co-transplantation of hematopoietic and stromal bone marrow cells. The aim of this study was to investigate a role of bone marrow stromal cells (BMSC) in the donor bone marrow engraftment, and their importance for development of complications after HSCT. Functional analysis of BMSC cultures from thirty-eight recipients of allogeneic hematopoietic stem cells has been shown that induced osteogenic differentiation correlated with faster hematopoiesis recovery in transplanted patients. Moreover, a pronounced ability for adipogenic differentiation proved to be associated with lower probability of the graft hypofunction. A pronounced hemostimulatory activity of stromal cells was observed in our study, thus suggesting effects of short-range hematopoietic growth factors from BMSC upon hematopoietic reconstitution. At the same time, a more pronounced pre-transplant expression of molecules which promote donor HSC homing by the recipient stromal cells was associated with faster hematopoietic recovery after transplantation." } ["DOI"]=> array(37) { ["ID"]=> string(2) "28" ["TIMESTAMP_X"]=> string(19) "2016-04-06 14:11:12" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(3) "DOI" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(3) "DOI" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "28" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8452" ["VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-71-77" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-71-77" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(3) "DOI" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(37) "10.18620/ctt-1866-8836-2016-5-4-71-77" } ["NAME_EN"]=> array(37) { ["ID"]=> string(2) "40" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:49:47" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(4) "Name" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "NAME_EN" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "80" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "40" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> NULL ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8456" ["VALUE"]=> string(98) "Impact of recipient marrow stromal cells functional characteristics on engraftment after allo-HSCT" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(98) "Impact of recipient marrow stromal cells functional characteristics on engraftment after allo-HSCT" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(4) "Name" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(98) "Impact of recipient marrow stromal cells functional characteristics on engraftment after allo-HSCT" } ["ORGANIZATION_EN"]=> array(37) { ["ID"]=> string(2) "38" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:02:59" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Organization" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_EN" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "38" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8454" ["VALUE"]=> array(2) { ["TEXT"]=> string(785) "R. M. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, and Chair of Hematology,<br> Transfusiology and Transplantation, The First St. Petersburg State I. Pavlov Medical University, St. Petersburg, Russian<br> Federation<br> <br> Dr. Ildar M.Barkhatov, PhD, Laboratory Head, R. M. Gorbacheva<br> Memorial Research Institute of Children Oncology,<br> Hematology and Transplantation, The First St. Petersburg<br> State I. Pavlov Medical University, L.Tolstoy St. 6/8, 197022,<br> St. Petersburg, Russian Federation<br> <br> Phone: +7(911) 778 2785<br> E-mail: <a href="mailto:i.barkhatov@gmail.com">i.barkhatov@gmail.com</a><br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(691) "R. M. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, and Chair of Hematology,
Transfusiology and Transplantation, The First St. Petersburg State I. Pavlov Medical University, St. Petersburg, Russian
Federation
Dr. Ildar M.Barkhatov, PhD, Laboratory Head, R. M. Gorbacheva
Memorial Research Institute of Children Oncology,
Hematology and Transplantation, The First St. Petersburg
State I. Pavlov Medical University, L.Tolstoy St. 6/8, 197022,
St. Petersburg, Russian Federation
Phone: +7(911) 778 2785
E-mail: i.barkhatov@gmail.com
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Organization" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(691) "R. M. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, and Chair of Hematology,
Transfusiology and Transplantation, The First St. Petersburg State I. Pavlov Medical University, St. Petersburg, Russian
Federation
Dr. Ildar M.Barkhatov, PhD, Laboratory Head, R. M. Gorbacheva
Memorial Research Institute of Children Oncology,
Hematology and Transplantation, The First St. Petersburg
State I. Pavlov Medical University, L.Tolstoy St. 6/8, 197022,
St. Petersburg, Russian Federation
Phone: +7(911) 778 2785
E-mail: i.barkhatov@gmail.com
" } ["AUTHORS"]=> array(38) { ["ID"]=> string(2) "24" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:45:07" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "AUTHORS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "24" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(1) { [0]=> string(4) "8478" } ["VALUE"]=> array(1) { [0]=> string(3) "695" } ["DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(1) { [0]=> string(3) "695" } ["~DESCRIPTION"]=> array(1) { [0]=> string(0) "" } ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(61) "Ildar M. Barkhatov" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["AUTHOR_RU"]=> array(37) { ["ID"]=> string(2) "25" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(12) "Авторы" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "AUTHOR_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "25" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8449" ["VALUE"]=> array(2) { ["TEXT"]=> string(327) "Ильдар М. Бархатов, Николай Ю. Цветков, Даниил Е. Ершов, Марина Ю. Лаврухина, Алена И. Шакирова, Алиса Я.<br> Поттер, Анна Г. Смирнова, Людмила С. Зубаровская, Борис В. Афанасьев<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(315) "Ильдар М. Бархатов, Николай Ю. Цветков, Даниил Е. Ершов, Марина Ю. Лаврухина, Алена И. Шакирова, Алиса Я.
Поттер, Анна Г. Смирнова, Людмила С. Зубаровская, Борис В. Афанасьев
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(12) "Авторы" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(315) "Ильдар М. Бархатов, Николай Ю. Цветков, Даниил Е. Ершов, Марина Ю. Лаврухина, Алена И. Шакирова, Алиса Я.
Поттер, Анна Г. Смирнова, Людмила С. Зубаровская, Борис В. Афанасьев
" } ["SUBMITTED"]=> array(37) { ["ID"]=> string(2) "20" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(21) "Дата подачи" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(9) "SUBMITTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "20" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8445" ["VALUE"]=> string(22) "10/10/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "10/10/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(21) "Дата подачи" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "10/10/2016 12:00:00 am" } ["ACCEPTED"]=> array(37) { ["ID"]=> string(2) "21" ["TIMESTAMP_X"]=> string(19) "2015-09-02 17:21:42" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(25) "Дата принятия" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "ACCEPTED" ["DEFAULT_VALUE"]=> NULL ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "21" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(8) "DateTime" ["USER_TYPE_SETTINGS"]=> NULL ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8446" ["VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(22) "11/25/2016 12:00:00 am" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(25) "Дата принятия" ["~DEFAULT_VALUE"]=> NULL ["DISPLAY_VALUE"]=> string(32) "11/25/2016 12:00:00 am" } ["KEYWORDS"]=> array(38) { ["ID"]=> string(2) "19" ["TIMESTAMP_X"]=> string(19) "2015-09-03 10:46:01" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(27) "Ключевые слова" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(8) "KEYWORDS" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "Y" ["XML_ID"]=> string(2) "19" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "4" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "Y" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "Y" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> array(4) { [0]=> string(4) "8474" [1]=> string(4) "8475" [2]=> string(4) "8476" [3]=> string(4) "8477" } ["VALUE"]=> array(4) { [0]=> string(3) "696" [1]=> string(3) "697" [2]=> string(3) "698" [3]=> string(3) "699" } ["DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(4) { [0]=> string(3) "696" [1]=> string(3) "697" [2]=> string(3) "698" [3]=> string(3) "699" } ["~DESCRIPTION"]=> array(4) { [0]=> string(0) "" [1]=> string(0) "" [2]=> string(0) "" [3]=> string(0) "" } ["~NAME"]=> string(27) "Ключевые слова" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> array(4) { [0]=> string(107) "Стромальные клетки костного мозга" [1]=> string(71) "острые лейкозы" [2]=> string(101) "функциональные характеристики" [3]=> string(83) "типы дифференцировки" } ["LINK_ELEMENT_VALUE"]=> bool(false) } ["CONTACT"]=> array(38) { ["ID"]=> string(2) "23" ["TIMESTAMP_X"]=> string(19) "2015-09-03 14:43:05" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(14) "Контакт" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(7) "CONTACT" ["DEFAULT_VALUE"]=> string(0) "" ["PROPERTY_TYPE"]=> string(1) "E" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "23" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "3" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "Y" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(13) "EAutocomplete" ["USER_TYPE_SETTINGS"]=> array(9) { ["VIEW"]=> string(1) "E" ["SHOW_ADD"]=> string(1) "Y" ["MAX_WIDTH"]=> int(0) ["MIN_HEIGHT"]=> int(24) ["MAX_HEIGHT"]=> int(1000) ["BAN_SYM"]=> string(2) ",;" ["REP_SYM"]=> string(1) " " ["OTHER_REP_SYM"]=> string(0) "" ["IBLOCK_MESS"]=> string(1) "N" } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8447" ["VALUE"]=> string(3) "695" ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> string(3) "695" ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(14) "Контакт" ["~DEFAULT_VALUE"]=> string(0) "" ["DISPLAY_VALUE"]=> string(61) "Ildar M. Barkhatov" ["LINK_ELEMENT_VALUE"]=> bool(false) } ["SUMMARY_RU"]=> array(37) { ["ID"]=> string(2) "27" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(29) "Описание/Резюме" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(10) "SUMMARY_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "27" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8451" ["VALUE"]=> array(2) { ["TEXT"]=> string(2500) "Наблюдаемая в ряде случаев гипофункция транс- плантата является нередкой и обусловливает не- обходимость разработки функциональных тестов исследования клеток стромы с целью обоснования показаний к совместной трансплантации гемопоэ- тических и стромальных клеток костного мозга. Це- лью данного исследования являлось изучение роли стромальных клеток костного мозга в процессе при- живления донорского костного мозга и их значения в развитии осложнений после ТГСК. При анализе функциональных характеристик стромальных кле- ток у 38 реципиентов аллогенного трансплантата ге- мопоэтических стволовых клеток было показано уве- личение активности остеогенной дифференцировки на фоне укорочения сроков восстановления кро- ветворения. Вместе с тем, наличие более выражен- ной способности к адипогенной дифференцировке ассоциировано с меньшей вероятностью развития гипофункции трансплантата. При исследовании ге- мостимулирующей активности стромальных клеток реципиента были отмечены факты, указывающие на преимущественное влияние короткодистантных ростовых факторов на посттрансплантационную реконституцию гемопоэза. При этом более выра- женная экспрессия молекул, опосредующих хоу- минг ГСК в стромальных клетках реципиента перед трансплантацией ассоциировано с более быстрым восстановлением гемопоэза у трансплантированных больных." ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(2500) "Наблюдаемая в ряде случаев гипофункция транс- плантата является нередкой и обусловливает не- обходимость разработки функциональных тестов исследования клеток стромы с целью обоснования показаний к совместной трансплантации гемопоэ- тических и стромальных клеток костного мозга. Це- лью данного исследования являлось изучение роли стромальных клеток костного мозга в процессе при- живления донорского костного мозга и их значения в развитии осложнений после ТГСК. При анализе функциональных характеристик стромальных кле- ток у 38 реципиентов аллогенного трансплантата ге- мопоэтических стволовых клеток было показано уве- личение активности остеогенной дифференцировки на фоне укорочения сроков восстановления кро- ветворения. Вместе с тем, наличие более выражен- ной способности к адипогенной дифференцировке ассоциировано с меньшей вероятностью развития гипофункции трансплантата. При исследовании ге- мостимулирующей активности стромальных клеток реципиента были отмечены факты, указывающие на преимущественное влияние короткодистантных ростовых факторов на посттрансплантационную реконституцию гемопоэза. При этом более выра- женная экспрессия молекул, опосредующих хоу- минг ГСК в стромальных клетках реципиента перед трансплантацией ассоциировано с более быстрым восстановлением гемопоэза у трансплантированных больных." ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(29) "Описание/Резюме" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(2500) "Наблюдаемая в ряде случаев гипофункция транс- плантата является нередкой и обусловливает не- обходимость разработки функциональных тестов исследования клеток стромы с целью обоснования показаний к совместной трансплантации гемопоэ- тических и стромальных клеток костного мозга. Це- лью данного исследования являлось изучение роли стромальных клеток костного мозга в процессе при- живления донорского костного мозга и их значения в развитии осложнений после ТГСК. При анализе функциональных характеристик стромальных кле- ток у 38 реципиентов аллогенного трансплантата ге- мопоэтических стволовых клеток было показано уве- личение активности остеогенной дифференцировки на фоне укорочения сроков восстановления кро- ветворения. Вместе с тем, наличие более выражен- ной способности к адипогенной дифференцировке ассоциировано с меньшей вероятностью развития гипофункции трансплантата. При исследовании ге- мостимулирующей активности стромальных клеток реципиента были отмечены факты, указывающие на преимущественное влияние короткодистантных ростовых факторов на посттрансплантационную реконституцию гемопоэза. При этом более выра- женная экспрессия молекул, опосредующих хоу- минг ГСК в стромальных клетках реципиента перед трансплантацией ассоциировано с более быстрым восстановлением гемопоэза у трансплантированных больных." } ["ORGANIZATION_RU"]=> array(37) { ["ID"]=> string(2) "26" ["TIMESTAMP_X"]=> string(19) "2015-09-02 18:01:20" ["IBLOCK_ID"]=> string(1) "2" ["NAME"]=> string(22) "Организации" ["ACTIVE"]=> string(1) "Y" ["SORT"]=> string(3) "500" ["CODE"]=> string(15) "ORGANIZATION_RU" ["DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["PROPERTY_TYPE"]=> string(1) "S" ["ROW_COUNT"]=> string(1) "1" ["COL_COUNT"]=> string(2) "30" ["LIST_TYPE"]=> string(1) "L" ["MULTIPLE"]=> string(1) "N" ["XML_ID"]=> string(2) "26" ["FILE_TYPE"]=> string(0) "" ["MULTIPLE_CNT"]=> string(1) "5" ["TMP_ID"]=> NULL ["LINK_IBLOCK_ID"]=> string(1) "0" ["WITH_DESCRIPTION"]=> string(1) "N" ["SEARCHABLE"]=> string(1) "N" ["FILTRABLE"]=> string(1) "N" ["IS_REQUIRED"]=> string(1) "N" ["VERSION"]=> string(1) "1" ["USER_TYPE"]=> string(4) "HTML" ["USER_TYPE_SETTINGS"]=> array(1) { ["height"]=> int(200) } ["HINT"]=> string(0) "" ["PROPERTY_VALUE_ID"]=> string(4) "8450" ["VALUE"]=> array(2) { ["TEXT"]=> string(294) "Первый Санкт-Петербургский государственный медицинский Университет им. акад. И. П. Павлова Министерства<br> Здравоохранения РФ, Санкт-Петербург, Россия<br>" ["TYPE"]=> string(4) "HTML" } ["DESCRIPTION"]=> string(0) "" ["VALUE_ENUM"]=> NULL ["VALUE_XML_ID"]=> NULL ["VALUE_SORT"]=> NULL ["~VALUE"]=> array(2) { ["TEXT"]=> string(282) "Первый Санкт-Петербургский государственный медицинский Университет им. акад. И. П. Павлова Министерства
Здравоохранения РФ, Санкт-Петербург, Россия
" ["TYPE"]=> string(4) "HTML" } ["~DESCRIPTION"]=> string(0) "" ["~NAME"]=> string(22) "Организации" ["~DEFAULT_VALUE"]=> array(2) { ["TEXT"]=> string(0) "" ["TYPE"]=> string(4) "HTML" } ["DISPLAY_VALUE"]=> string(282) "Первый Санкт-Петербургский государственный медицинский Университет им. акад. И. П. Павлова Министерства
Здравоохранения РФ, Санкт-Петербург, Россия
" } } } }
12/30/2016 02:22:00 pm

Afanasyev B. V. (St. Petersburg, Russia)
Wagemaker G. (Rotterdam, Netherlands)
Zander A.R. (Hamburg, Germany)
Fehse B. (Hamburg, Germany)
Chukhlovin A. B. (St. Petersburg, Russia)
Aleynikova O. V. (Minsk, Belarus)
Borset M. (Trondheim, Norway)
Chechetkin A. V. (St. Petersburg, Russia)
Fibbe W. (Leiden, Netherlands)
Galibin O. V. (St. Petersburg, Russia)
Hölzer D. (Frankfurt a.M., Germany)
Klimko N. N. (St. Petersburg, Russia)
Kolb H.-J. (München, Germany)
Kröger N. (Hamburg, Germany)
Kulagin A. D. (St. Petersburg, Russia)
Lange C. (Hamburg, Germany)
Mamaev N. N. (St. Petersburg, Russia)
Mikhailova N. B. (St. Petersburg, Russia)
Moiseev I. S. (St. Petersburg, Russia)
Nagler A. (Tel-Aviv, Israel)
Nemkov A. S. (St. Petersburg, Russia)
Paramonov I. V. (Kirov, Russia)
Roumiantsev A. G. (Moscow, Russia)
Savchenko V. G. (Moscow, Russia)
Smirnov A. V. (St. Petersburg, Russia)
Uss A. L. (Minsk, Belarus)
Zubarovskaya L. S. (St.Petersburg, Russia)
In the past few years there is a rising opinion in the scientific circles that the prospects of hematopoietic stem cell transplantation (HSCT) are diminishing due to approval of novel target and immunomodulating agents.
These targeted anticancer drugs have drawn attention of clinicians, scientists and social media. For instance, imatinib and other tyrosine kinase inhibitors (TKI’s) proved to be the first ‘magic’ bullet applied for treatment of chronic myeloid leukemia and other Ph-positive malignancies. Development of JAK-2 inhibitors (Ruxolitinib) paved further ways in therapy of some chronic myeloproliferative neoplasms.
A less specific approach includes eradication of malignant cell populations expressing a typical surface marker which could be attacked by the cytolytic drug. Clinical experience brings great expectations for Blinatumomab, an anti-CD-19/ CD3 bi-specific antibody applied for treatment of B-cell acute lymphoblastic leukemia (B-ALL). Hodgkin’s disease, another common disorder, is now more curable with cytolytic drug-antibody conjugate, Brentuximab, an anti-CD30+ monoclonal antibody.
The most encouraging results in clinical oncology have been quite recently obtained with the so-called immune checkpoint inhibitors which proved to be effective in a variety of solid tumors, as well as in hematological malignancies. Nivolumab, a PD-1 inhibitor, is the most advanced drug developed in the field. Specific action of these drugs on the target cells is based on blockage of a molecular checkpoint which regulates T-cell switch towards apoptosis. However, precise therapeutic effects of PD1-inhibitors should be further elucidated. In particular, we are only in the beginning of the path to understanding the mechanisms, facilitating the sensitivity and resistance to this group of agents. Therefore, several years are required in order to specify the place of PD-inhibitors in the treatment of different tumors.
Meanwhile, with accumulation of clinical data, it becomes clear that novel agents do not substitute HSCT, but rather significantly extend its curative potential through achievement of deeper remissions before the transplant and prevention of relapse as post-transplant therapy. This is particularly important for the patients resistant to conventional chemotherapy, and novel agents are applied as a ‘bridge’ to the subsequent hematopoietic transplantation. In this respect, further clinical studies are required for TKI’s, Blinatumumab, Brentuximab, immune checkpoint inhibitors, and other targeted drugs. On the other hand, there is emerging data that some of the target agents, like TKIs or Ruxolitinib, bear the significant potential for modulating graft-versus-host and graft-versus-leukemia effects, thus further improving the results of HSCT.
We are awaiting new reviews and original articles on the targeted drugs designed for treatment of oncological disorders. The CTT Journal is becoming popular among Russian- speaking specialists in the field of hematopoietic stem cell transplantation, due to broader coverage of different topics related to cellular therapy. The Journal is indexed in Scopus, thus increasing its visibility in the World Web. Moreover, the articles published in CTT are now adopted for Doctor’s Thesis by the Russian Higher Attestation Commission. We hope that the number of our authors and readership will also increase with time.
Clinical articles
Experimental studies
Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev
Clinical articles
Array
(
[KEYWORDS] => Array
(
[ID] => 19
[TIMESTAMP_X] => 2015-09-03 10:46:01
[IBLOCK_ID] => 2
[NAME] => Ключевые слова
[ACTIVE] => Y
[SORT] => 500
[CODE] => KEYWORDS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 19
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 4
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => Y
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => Y
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8260
[1] => 8261
[2] => 8262
[3] => 8263
[4] => 8264
[5] => 8265
)
[VALUE] => Array
(
[0] => 94
[1] => 344
[2] => 669
[3] => 670
[4] => 671
[5] => 672
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
[5] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 94
[1] => 344
[2] => 669
[3] => 670
[4] => 671
[5] => 672
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
[5] =>
)
[~NAME] => Ключевые слова
[~DEFAULT_VALUE] =>
)
[SUBMITTED] => Array
(
[ID] => 20
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата подачи
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUBMITTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 20
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8205
[VALUE] => 06/17/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 06/17/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата подачи
[~DEFAULT_VALUE] =>
)
[ACCEPTED] => Array
(
[ID] => 21
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата принятия
[ACTIVE] => Y
[SORT] => 500
[CODE] => ACCEPTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 21
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8206
[VALUE] => 11/25/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 11/25/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата принятия
[~DEFAULT_VALUE] =>
)
[PUBLISHED] => Array
(
[ID] => 22
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата публикации
[ACTIVE] => Y
[SORT] => 500
[CODE] => PUBLISHED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 22
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Дата публикации
[~DEFAULT_VALUE] =>
)
[CONTACT] => Array
(
[ID] => 23
[TIMESTAMP_X] => 2015-09-03 14:43:05
[IBLOCK_ID] => 2
[NAME] => Контакт
[ACTIVE] => Y
[SORT] => 500
[CODE] => CONTACT
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 23
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8225
[VALUE] => 663
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 663
[~DESCRIPTION] =>
[~NAME] => Контакт
[~DEFAULT_VALUE] =>
)
[AUTHORS] => Array
(
[ID] => 24
[TIMESTAMP_X] => 2015-09-03 10:45:07
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHORS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 24
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8266
[1] => 8267
[2] => 8268
[3] => 8269
[4] => 8270
[5] => 8271
)
[VALUE] => Array
(
[0] => 663
[1] => 664
[2] => 665
[3] => 666
[4] => 667
[5] => 668
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
[5] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 663
[1] => 664
[2] => 665
[3] => 666
[4] => 667
[5] => 668
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
[5] =>
)
[~NAME] => Авторы
[~DEFAULT_VALUE] =>
)
[AUTHOR_RU] => Array
(
[ID] => 25
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 25
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8213
[VALUE] => Array
(
[TEXT] => Виталий Н. Чеботкевич, Станислав С. Бессмельцев, Екатерина Е. Киселева, Наталья П. Стижак,
Елена И. Кайтанджан, Виталий В. Бурылев
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Виталий Н. Чеботкевич, Станислав С. Бессмельцев, Екатерина Е. Киселева, Наталья П. Стижак,
Елена И. Кайтанджан, Виталий В. Бурылев
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_RU] => Array
(
[ID] => 26
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Организации
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 26
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8214
[VALUE] => Array
(
[TEXT] => Российский научно-исследовательский институт гематологии и трансфузиологии, Санкт-Петербург, Россия
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Российский научно-исследовательский институт гематологии и трансфузиологии, Санкт-Петербург, Россия
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Организации
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_RU] => Array
(
[ID] => 27
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Описание/Резюме
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 27
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8258
[VALUE] => Array
(
[TEXT] => <p>
Интенсивная цитостатическая химиотерапия является стандартной стратегией лечения лейкозов и лимфом. В то же время такое лечение вызывает негативные эффекты, в том числе лимфопению, гранулоцитопению и повреждение тканевых барьеров, что ассоциировано с существенными рисками инфекционных осложнений, особенно – бактериальным сепсисом и виремией. Целью нашей работы было выявление бактериемии и фунгемии у онкогематологических больных после интенсивной химиотерапии и оценка потенциальной модифицирующей роли герпесвирусных инфекций.
</p>
<p>
Мы определяли частоту развития инфекционных осложнений и соответствующих этиологических факторов в 2 группах пациентов онкогематологического профиля, проходивших лечение в Российском институте гематологии и трансфузиологии, учитывая, в частности, случаи смешанных инфекций. Мазки из зева, венозная кровь, а также образцы мочи и мокроты отбирали для рутинных бактериологических и вирусологических исследований. Лейкоциты цельной крови тестировали на вирусы посредством ПЦР с применением стандартных протоколов.
</p>
<p>
В первой группе пациентов (85 случаев) проводили исследования на респираторные вирусные инфекции. Вирусы гриппа и парагриппа, респираторно-синцитиальный вирус (РСВ), риновирус, аденовирус определялись в крови у единичных пациентов. В то же время герпесвирусы выявлялись, в целом, в 42% случаев. В частности, ДНК вирусов простого герпеса, Эпштейна-Барр (ВЭБ) и цитомегаловируса (ЦМВ) в лейкоцитах крови обнаружены в 5.2%, 26.3%, и 10.5%, соответственно. Вирусные инфекции в этой группе пациентов не были достоверно ассоциированы с положительными бактериологическими пробами.
</p>
<p>
Отдельно анализировали результаты в группе из 33 случаев доказанного сепсиса, которые выявили среди 64 пациентов. В целом, частота выявления Грам-позитивных микроорганизмов преобладала над Грам-негативными культурами (69,2% и 30,8%, соответственно). Однако соотношение выявленной Грам-негативной флоры повысилось с 23% до 40% в период с 2002 по 2013 гг. (p < 0,05). Коагулазо-негативные стафилококки (КНС) преобладали среди Грам-позитивных микроорганизмов, в частности, S. epidermidis и S. aureus, тогда как Enterobacteriaceae, в особенности, E.coli, были наиболее частыми Грам-негативными бактериями.
</p>
<p>
Представляет интерес, что у 4 из 11 пациентов с КНС-сепсисом отмечены сопутствующие герпесвирусные инфекции, а именно 2 случая были ассоциированы с ВЭБ; 1 – с ЦМВ и 1 – с герпесвирусом человека 6 типа и ВЭБ. Частая реактивация герпесвирусов может вызвать иммуносупрессию или быть дополнительным прогностическим признаком иммунодефицита в плане риска бактериальных инфекций в более поздние сроки. Поэтому в таких случаях следует учитывать предрасположенность к тяжелым инфекционным осложнениям при планировании трансплантацию гемопоэтических клеток этим больным.
</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] =>
Интенсивная цитостатическая химиотерапия является стандартной стратегией лечения лейкозов и лимфом. В то же время такое лечение вызывает негативные эффекты, в том числе лимфопению, гранулоцитопению и повреждение тканевых барьеров, что ассоциировано с существенными рисками инфекционных осложнений, особенно – бактериальным сепсисом и виремией. Целью нашей работы было выявление бактериемии и фунгемии у онкогематологических больных после интенсивной химиотерапии и оценка потенциальной модифицирующей роли герпесвирусных инфекций.
Мы определяли частоту развития инфекционных осложнений и соответствующих этиологических факторов в 2 группах пациентов онкогематологического профиля, проходивших лечение в Российском институте гематологии и трансфузиологии, учитывая, в частности, случаи смешанных инфекций. Мазки из зева, венозная кровь, а также образцы мочи и мокроты отбирали для рутинных бактериологических и вирусологических исследований. Лейкоциты цельной крови тестировали на вирусы посредством ПЦР с применением стандартных протоколов.
В первой группе пациентов (85 случаев) проводили исследования на респираторные вирусные инфекции. Вирусы гриппа и парагриппа, респираторно-синцитиальный вирус (РСВ), риновирус, аденовирус определялись в крови у единичных пациентов. В то же время герпесвирусы выявлялись, в целом, в 42% случаев. В частности, ДНК вирусов простого герпеса, Эпштейна-Барр (ВЭБ) и цитомегаловируса (ЦМВ) в лейкоцитах крови обнаружены в 5.2%, 26.3%, и 10.5%, соответственно. Вирусные инфекции в этой группе пациентов не были достоверно ассоциированы с положительными бактериологическими пробами.
Отдельно анализировали результаты в группе из 33 случаев доказанного сепсиса, которые выявили среди 64 пациентов. В целом, частота выявления Грам-позитивных микроорганизмов преобладала над Грам-негативными культурами (69,2% и 30,8%, соответственно). Однако соотношение выявленной Грам-негативной флоры повысилось с 23% до 40% в период с 2002 по 2013 гг. (p < 0,05). Коагулазо-негативные стафилококки (КНС) преобладали среди Грам-позитивных микроорганизмов, в частности, S. epidermidis и S. aureus, тогда как Enterobacteriaceae, в особенности, E.coli, были наиболее частыми Грам-негативными бактериями.
Представляет интерес, что у 4 из 11 пациентов с КНС-сепсисом отмечены сопутствующие герпесвирусные инфекции, а именно 2 случая были ассоциированы с ВЭБ; 1 – с ЦМВ и 1 – с герпесвирусом человека 6 типа и ВЭБ. Частая реактивация герпесвирусов может вызвать иммуносупрессию или быть дополнительным прогностическим признаком иммунодефицита в плане риска бактериальных инфекций в более поздние сроки. Поэтому в таких случаях следует учитывать предрасположенность к тяжелым инфекционным осложнениям при планировании трансплантацию гемопоэтических клеток этим больным.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Описание/Резюме
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[DOI] => Array
(
[ID] => 28
[TIMESTAMP_X] => 2016-04-06 14:11:12
[IBLOCK_ID] => 2
[NAME] => DOI
[ACTIVE] => Y
[SORT] => 500
[CODE] => DOI
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 28
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8215
[VALUE] => 10.18620/ctt-1866-8836-2016-5-4-21-31
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10.18620/ctt-1866-8836-2016-5-4-21-31
[~DESCRIPTION] =>
[~NAME] => DOI
[~DEFAULT_VALUE] =>
)
[AUTHOR_EN] => Array
(
[ID] => 37
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Author
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 37
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8216
[VALUE] => Array
(
[TEXT] => Vitaly N. Chebotkevich, Stanislav S. Bessmeltsev, Ekaterina E. Kiseleva, Natalya P. Stizhak, Elena I. Kaytandzhan, Vitaly V. Burylev
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Vitaly N. Chebotkevich, Stanislav S. Bessmeltsev, Ekaterina E. Kiseleva, Natalya P. Stizhak, Elena I. Kaytandzhan, Vitaly V. Burylev
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Author
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_EN] => Array
(
[ID] => 38
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Organization
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 38
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8217
[VALUE] => Array
(
[TEXT] => Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St.16, 191024 St. Petersburg, Russia.<br>
<br>
Professor Vitaly N. Chebotkevich, Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St. 16, 191024 St. Petersburg, Russia.<br>
<br>
Phone: +7 (812) 717-29-58<br>
Fax: +7 (812) 717-25-50<br>
E-mail: <a href="mailto:vitnikcheb@mail.ru">vitnikcheb@mail.ru</a>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St.16, 191024 St. Petersburg, Russia.
Professor Vitaly N. Chebotkevich, Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St. 16, 191024 St. Petersburg, Russia.
Phone: +7 (812) 717-29-58
Fax: +7 (812) 717-25-50
E-mail: vitnikcheb@mail.ru
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Organization
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_EN] => Array
(
[ID] => 39
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Description / Summary
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 39
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8259
[VALUE] => Array
(
[TEXT] => <p>
Intensive cytostatic chemotherapy is a standard strategy for leukemia treatment. Meanwhile, such treatment causes negative effects, i.e., including lymphopenia, granulocytopenia and damage to tissue barriers associated with significant risks of infectious complications, especially, bacterial sepsis and viremia. Our study was aimed for identification of bacteremia and fungemia in oncohematological patients following intensive chemotherapy, and assessment of potential modifying role of herpesvirus infections.
</p>
<p>
We assessed frequency of infectious complications and their etiological agents in two groups of oncohematological patients treated at the Russian Institute of Hematology and Transfusion, especially focusing on mixed infections. Throat smears, venous blood, as well as urine and sputum specimens were taken for routine bacteriological cultures. Whole blood leukocytes were virologically tested by PCR, using standard examination protocol.
</p>
<p>
Our first virological study was performed for respiratory infections and included 85 randomly chosen patients. Influenza and parainfluenza viruses, respiratory syncytial virus, rhinovirus, adenovirus were detected in blood of single patients. Meanwhile, herpesviruses were detectable in 42% proportion of cases, i.e., HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. Viral infections were not associated with positive bacteriological findings in this group. Our group of 33 clinical cases with proven sepsis observed among 64 patients. Generally, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, the ratio of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p<0.05). Coagulase- negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria.
</p>
<p>
Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: 2 cases were associated with EBV; 1, with CMV, and 1, with HHV6/EBV coinfection. Therefore, a high ratio of viral and bacterial co-infections may be revealed in a number of patients with proven sepsis. Moreover, common reactivation of herpesviruses may cause immunosuppression, or represent additional immunodeficiency markers predictive for bacterial infections at later terms. Therefore, one should take into account their predisposal for severe infectious complications when planning hematopoietic stem cell transplantation (HSCT) for these patients.
</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] =>
Intensive cytostatic chemotherapy is a standard strategy for leukemia treatment. Meanwhile, such treatment causes negative effects, i.e., including lymphopenia, granulocytopenia and damage to tissue barriers associated with significant risks of infectious complications, especially, bacterial sepsis and viremia. Our study was aimed for identification of bacteremia and fungemia in oncohematological patients following intensive chemotherapy, and assessment of potential modifying role of herpesvirus infections.
We assessed frequency of infectious complications and their etiological agents in two groups of oncohematological patients treated at the Russian Institute of Hematology and Transfusion, especially focusing on mixed infections. Throat smears, venous blood, as well as urine and sputum specimens were taken for routine bacteriological cultures. Whole blood leukocytes were virologically tested by PCR, using standard examination protocol.
Our first virological study was performed for respiratory infections and included 85 randomly chosen patients. Influenza and parainfluenza viruses, respiratory syncytial virus, rhinovirus, adenovirus were detected in blood of single patients. Meanwhile, herpesviruses were detectable in 42% proportion of cases, i.e., HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. Viral infections were not associated with positive bacteriological findings in this group. Our group of 33 clinical cases with proven sepsis observed among 64 patients. Generally, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, the ratio of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p<0.05). Coagulase- negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria.
Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: 2 cases were associated with EBV; 1, with CMV, and 1, with HHV6/EBV coinfection. Therefore, a high ratio of viral and bacterial co-infections may be revealed in a number of patients with proven sepsis. Moreover, common reactivation of herpesviruses may cause immunosuppression, or represent additional immunodeficiency markers predictive for bacterial infections at later terms. Therefore, one should take into account their predisposal for severe infectious complications when planning hematopoietic stem cell transplantation (HSCT) for these patients.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Description / Summary
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[NAME_EN] => Array
(
[ID] => 40
[TIMESTAMP_X] => 2015-09-03 10:49:47
[IBLOCK_ID] => 2
[NAME] => Name
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 40
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8218
[VALUE] => Bloodstream infections and herpesvirus activation following intensive chemotherapy of adult oncohematological patients
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Bloodstream infections and herpesvirus activation following intensive chemotherapy of adult oncohematological patients
[~DESCRIPTION] =>
[~NAME] => Name
[~DEFAULT_VALUE] =>
)
[FULL_TEXT_RU] => Array
(
[ID] => 42
[TIMESTAMP_X] => 2015-09-07 20:29:18
[IBLOCK_ID] => 2
[NAME] => Полный текст
[ACTIVE] => Y
[SORT] => 500
[CODE] => FULL_TEXT_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 42
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Полный текст
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[PDF_RU] => Array
(
[ID] => 43
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF RUS
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_RU
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 43
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8232
[VALUE] => 248
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 248
[~DESCRIPTION] =>
[~NAME] => PDF RUS
[~DEFAULT_VALUE] =>
)
[PDF_EN] => Array
(
[ID] => 44
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF ENG
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 44
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8233
[VALUE] => 249
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 249
[~DESCRIPTION] =>
[~NAME] => PDF ENG
[~DEFAULT_VALUE] =>
)
[NAME_LONG] => Array
(
[ID] => 45
[TIMESTAMP_X] => 2023-04-13 00:55:00
[IBLOCK_ID] => 2
[NAME] => Название (для очень длинных заголовков)
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_LONG
[DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 45
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 80
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Название (для очень длинных заголовков)
[~DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
)
)
Vitaly N. Chebotkevich, Stanislav S. Bessmeltsev, Ekaterina E. Kiseleva, Natalya P. Stizhak, Elena I. Kaytandzhan, Vitaly V. Burylev
Professor Vitaly N. Chebotkevich, Russian Research Institute of Hematology and Transfusiology, St. Petersburg; 2nd Sovetskaya St. 16, 191024 St. Petersburg, Russia.
Phone: +7 (812) 717-29-58
Fax: +7 (812) 717-25-50
E-mail: vitnikcheb@mail.ru
Intensive cytostatic chemotherapy is a standard strategy for leukemia treatment. Meanwhile, such treatment causes negative effects, i.e., including lymphopenia, granulocytopenia and damage to tissue barriers associated with significant risks of infectious complications, especially, bacterial sepsis and viremia. Our study was aimed for identification of bacteremia and fungemia in oncohematological patients following intensive chemotherapy, and assessment of potential modifying role of herpesvirus infections.
We assessed frequency of infectious complications and their etiological agents in two groups of oncohematological patients treated at the Russian Institute of Hematology and Transfusion, especially focusing on mixed infections. Throat smears, venous blood, as well as urine and sputum specimens were taken for routine bacteriological cultures. Whole blood leukocytes were virologically tested by PCR, using standard examination protocol.
Our first virological study was performed for respiratory infections and included 85 randomly chosen patients. Influenza and parainfluenza viruses, respiratory syncytial virus, rhinovirus, adenovirus were detected in blood of single patients. Meanwhile, herpesviruses were detectable in 42% proportion of cases, i.e., HSV, EBV, and CMV DNA in blood cells were revealed in 5.2%, 26.3%, and 10.5%, respectively. Viral infections were not associated with positive bacteriological findings in this group. Our group of 33 clinical cases with proven sepsis observed among 64 patients. Generally, Gram-positive species prevailed over Gram-negative bacteria (69.2% versus 30.8%). However, the ratio of detectable Gram-negative flora was found to be increased from 23.1% to 40.2% between 2002 and 2013 (p<0.05). Coagulase- negative staphylococci (CoNS) prevailed among Gram-positive microorganisms, in particular, S. epidermidis and S. aureus), whereas Enterobacteriaceae, especially, E.coli, dominated among the Gram-negative bacteria.
Interestingly, 4 of 11 patients with coagulase-negative bacteremia had a concomitant herpesvirus infection: 2 cases were associated with EBV; 1, with CMV, and 1, with HHV6/EBV coinfection. Therefore, a high ratio of viral and bacterial co-infections may be revealed in a number of patients with proven sepsis. Moreover, common reactivation of herpesviruses may cause immunosuppression, or represent additional immunodeficiency markers predictive for bacterial infections at later terms. Therefore, one should take into account their predisposal for severe infectious complications when planning hematopoietic stem cell transplantation (HSCT) for these patients.
Clinical articles
Array
(
[KEYWORDS] => Array
(
[ID] => 19
[TIMESTAMP_X] => 2015-09-03 10:46:01
[IBLOCK_ID] => 2
[NAME] => Ключевые слова
[ACTIVE] => Y
[SORT] => 500
[CODE] => KEYWORDS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 19
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 4
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => Y
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => Y
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8152
[1] => 8153
[2] => 8154
[3] => 8155
[4] => 8156
[5] => 8157
)
[VALUE] => Array
(
[0] => 105
[1] => 298
[2] => 659
[3] => 660
[4] => 18
[5] => 661
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
[5] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 105
[1] => 298
[2] => 659
[3] => 660
[4] => 18
[5] => 661
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
[5] =>
)
[~NAME] => Ключевые слова
[~DEFAULT_VALUE] =>
)
[SUBMITTED] => Array
(
[ID] => 20
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата подачи
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUBMITTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 20
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8138
[VALUE] => 09/21/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 09/21/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата подачи
[~DEFAULT_VALUE] =>
)
[ACCEPTED] => Array
(
[ID] => 21
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата принятия
[ACTIVE] => Y
[SORT] => 500
[CODE] => ACCEPTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 21
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8139
[VALUE] => 10/28/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10/28/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата принятия
[~DEFAULT_VALUE] =>
)
[PUBLISHED] => Array
(
[ID] => 22
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата публикации
[ACTIVE] => Y
[SORT] => 500
[CODE] => PUBLISHED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 22
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Дата публикации
[~DEFAULT_VALUE] =>
)
[CONTACT] => Array
(
[ID] => 23
[TIMESTAMP_X] => 2015-09-03 14:43:05
[IBLOCK_ID] => 2
[NAME] => Контакт
[ACTIVE] => Y
[SORT] => 500
[CODE] => CONTACT
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 23
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8140
[VALUE] => 7
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 7
[~DESCRIPTION] =>
[~NAME] => Контакт
[~DEFAULT_VALUE] =>
)
[AUTHORS] => Array
(
[ID] => 24
[TIMESTAMP_X] => 2015-09-03 10:45:07
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHORS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 24
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8158
)
[VALUE] => Array
(
[0] => 7
)
[DESCRIPTION] => Array
(
[0] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 7
)
[~DESCRIPTION] => Array
(
[0] =>
)
[~NAME] => Авторы
[~DEFAULT_VALUE] =>
)
[AUTHOR_RU] => Array
(
[ID] => 25
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 25
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8142
[VALUE] => Array
(
[TEXT] => Тапани Рууту
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Тапани Рууту
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_RU] => Array
(
[ID] => 26
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Организации
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 26
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8143
[VALUE] => Array
(
[TEXT] => Институт клинических исследований, Университетский госпиталь, Хельсинки, Финляндия
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Институт клинических исследований, Университетский госпиталь, Хельсинки, Финляндия
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Организации
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_RU] => Array
(
[ID] => 27
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Описание/Резюме
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 27
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8144
[VALUE] => Array
(
[TEXT] => <p>Трансплантация органов (Tx) широко применяется для лечения тяжелой органной недостаточности. Аллогенная трансплантация гемопоэтических клеток (алло-ТГСК), как таковая, часто вызывает повреждение органов, что может быть показанием к Тх. В связи с возрастающим числом обоих видов лечения растет вероятность комбинированных состояний, требующих, как Тх, так и ТГСК у одного и того же пациента. Эти ситуации приводят к ряду клинических проблем. Так, факторами риска, способными повлиять на исходы Тх после ТГСК, могут быть: токсичность процедуры ТГСК, последующее иммуносупрессивное лечение, не адекватное режиму после Тх, возможные проблемы, связанные с тканевой несовместимостью больного, гемопоэтических клеток донора и органного трансплантата, а также развитие инфекций. В то же время иммуносупрессивная терапия больных после Тх может нарушать функции органа и повышать риск инфекций. Кроме того, Тх иногда ассоциирована с развитием гематологических заболеваний, как, например, апластической анемии, посттрансплантационных лимфопролиферативных заболеваний, острых лейкозов и др. Цель настоящей статьи состоит в анализе существующего клинического опыта в области алло-ТГСК и Тх у одних и тех же пациентов. В частности, рассматривается европейское исследование с участием 107 центров EBMT и клинические данные о 45 Тх, проведенных пациентам, которым ранее выполнялась ТГСК. Им проводили, главным образом трансплантацию почек из-за повреждений после химио- или радиотерапии, тогда как пересадки печени делали в ранние сроки после ТГСК в связи с тяжелой вено-окклюзионной болезнью или оРТПХб или позже, по поводу хронической РТПХ или цирроза. Выживаемость и клинические исходы анализировали для каждой клинической ситуации. Особое внимание уделяли больным, получавшим трансплантаты легких при облитерирующем альвеолите после алло-ТГСК, где наблюдались худшие клинические исходы и меньшие сроки выживания.</p>
<p>Ряд свежих работ касается алло-ТГСК, проведенных после Тх. Частота гематологических рецидивов составила 22%, что позволило длительно наблюдать пациентов. Пятилетняя выживаемость пересаженных органов была около 30%, в основном – из-за отторжения трансплантата.</p>
<p>Таким образом, Тх может быть ценной стратегией при лечении реципиентов гемопоэтических клеток в случае развития органной недостаточности. Инфекции, отторжение трансплантата и другие осложнения довольно часты, но обычно поддаются лечению. Кроме того, некоторые реципиенты органов, у которых развились гематологические заболевания, могут выиграть от алло-ТГСК.</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Трансплантация органов (Tx) широко применяется для лечения тяжелой органной недостаточности. Аллогенная трансплантация гемопоэтических клеток (алло-ТГСК), как таковая, часто вызывает повреждение органов, что может быть показанием к Тх. В связи с возрастающим числом обоих видов лечения растет вероятность комбинированных состояний, требующих, как Тх, так и ТГСК у одного и того же пациента. Эти ситуации приводят к ряду клинических проблем. Так, факторами риска, способными повлиять на исходы Тх после ТГСК, могут быть: токсичность процедуры ТГСК, последующее иммуносупрессивное лечение, не адекватное режиму после Тх, возможные проблемы, связанные с тканевой несовместимостью больного, гемопоэтических клеток донора и органного трансплантата, а также развитие инфекций. В то же время иммуносупрессивная терапия больных после Тх может нарушать функции органа и повышать риск инфекций. Кроме того, Тх иногда ассоциирована с развитием гематологических заболеваний, как, например, апластической анемии, посттрансплантационных лимфопролиферативных заболеваний, острых лейкозов и др. Цель настоящей статьи состоит в анализе существующего клинического опыта в области алло-ТГСК и Тх у одних и тех же пациентов. В частности, рассматривается европейское исследование с участием 107 центров EBMT и клинические данные о 45 Тх, проведенных пациентам, которым ранее выполнялась ТГСК. Им проводили, главным образом трансплантацию почек из-за повреждений после химио- или радиотерапии, тогда как пересадки печени делали в ранние сроки после ТГСК в связи с тяжелой вено-окклюзионной болезнью или оРТПХб или позже, по поводу хронической РТПХ или цирроза. Выживаемость и клинические исходы анализировали для каждой клинической ситуации. Особое внимание уделяли больным, получавшим трансплантаты легких при облитерирующем альвеолите после алло-ТГСК, где наблюдались худшие клинические исходы и меньшие сроки выживания.
Ряд свежих работ касается алло-ТГСК, проведенных после Тх. Частота гематологических рецидивов составила 22%, что позволило длительно наблюдать пациентов. Пятилетняя выживаемость пересаженных органов была около 30%, в основном – из-за отторжения трансплантата.
Таким образом, Тх может быть ценной стратегией при лечении реципиентов гемопоэтических клеток в случае развития органной недостаточности. Инфекции, отторжение трансплантата и другие осложнения довольно часты, но обычно поддаются лечению. Кроме того, некоторые реципиенты органов, у которых развились гематологические заболевания, могут выиграть от алло-ТГСК.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Описание/Резюме
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[DOI] => Array
(
[ID] => 28
[TIMESTAMP_X] => 2016-04-06 14:11:12
[IBLOCK_ID] => 2
[NAME] => DOI
[ACTIVE] => Y
[SORT] => 500
[CODE] => DOI
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 28
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8145
[VALUE] => 10.18620/ctt-1866-8836-2016-5-4-15-20
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10.18620/ctt-1866-8836-2016-5-4-15-20
[~DESCRIPTION] =>
[~NAME] => DOI
[~DEFAULT_VALUE] =>
)
[AUTHOR_EN] => Array
(
[ID] => 37
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Author
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 37
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8146
[VALUE] => Array
(
[TEXT] => Tapani Ruutu
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Tapani Ruutu
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Author
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_EN] => Array
(
[ID] => 38
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Organization
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 38
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8147
[VALUE] => Array
(
[TEXT] => Tapani Ruutu, Helsinki University Hospital, Biomedicum<br>
2C, POB 705, FIN-00029 HUS, Helsinki, Finland<br>
<br>
Phone: +358 50 555 1279<br>
Fax: +358 9 4717 1506<br>
E-mail: <a href="mailto:tapani.ruutu@hus.fi">tapani.ruutu@hus.fi</a><br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Tapani Ruutu, Helsinki University Hospital, Biomedicum
2C, POB 705, FIN-00029 HUS, Helsinki, Finland
Phone: +358 50 555 1279
Fax: +358 9 4717 1506
E-mail: tapani.ruutu@hus.fi
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Organization
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_EN] => Array
(
[ID] => 39
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Description / Summary
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 39
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8148
[VALUE] => Array
(
[TEXT] => <p>
Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. Meanwhile, allogeneic hematopoietic stem cell transplantation (HSCT), by itself, causes frequent organ injury, thus being a potential indication for SOT. Due to increasing numbers of these interventions worldwide, a probability of combined disorders requiring both SOT and HSCT in the same patient is increasing. These transplantation combinations represent some clinical problems. Appropriate risk factors that may affect subsequent SOT outcomes post-HSCT include toxicity of HSCT procedure, immunosuppressive treatment for HSCT patients which may not be optimal for the SOT, potential problems caused by tissue incompatibility between host tissues, hematopoietic graft, and the solid organ graft, and emerging infections. Vice versa, in SOT recipients, the immunosuppressive medication may affect the organ functions, and increase the risk of infections. Moreover, SOT can be associated with development of hematological disorders, such as aplastic anemia, post-transplant lymphoproliferative disease, acute leukemias, etc. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients. Here we discuss the survey which enrolled patients from 107 EBMT centres and clinical data on 45 SOTs carried out in the patients who previously underwent allo-HSCT. Kidney transplantations were performed, mainly, because of the drug-induced organ affection, or radiotherapy, whereas liver transplantations were made either early, for severe VOD or aGvHD, or later, due to chronic liver GVHD or cirrhosis. Survival rates and clinical outcomes are analyzed for each clinical situation in the groups. A special attention is given to the patients who received lung transplants in cases of bronchiolitis obliterans following allo-HSCT, characterized by inferior clinical outcomes and shorter survival time.
</p>
<p>
Some recent data concern allo-HSCT carried out after SOT. The hematological relapse rate was 22 %, thus allowing long-term observations. The 5-year rate for the solid organ failure was about 30%, mostly, due to graft rejection.
</p>
<p>
In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. Infections, graft rejection and other complications are frequent but usually manageable. Also, selected SOT recipients developing hematological disorders may benefit from allo-HSCT.
</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] =>
Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. Meanwhile, allogeneic hematopoietic stem cell transplantation (HSCT), by itself, causes frequent organ injury, thus being a potential indication for SOT. Due to increasing numbers of these interventions worldwide, a probability of combined disorders requiring both SOT and HSCT in the same patient is increasing. These transplantation combinations represent some clinical problems. Appropriate risk factors that may affect subsequent SOT outcomes post-HSCT include toxicity of HSCT procedure, immunosuppressive treatment for HSCT patients which may not be optimal for the SOT, potential problems caused by tissue incompatibility between host tissues, hematopoietic graft, and the solid organ graft, and emerging infections. Vice versa, in SOT recipients, the immunosuppressive medication may affect the organ functions, and increase the risk of infections. Moreover, SOT can be associated with development of hematological disorders, such as aplastic anemia, post-transplant lymphoproliferative disease, acute leukemias, etc. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients. Here we discuss the survey which enrolled patients from 107 EBMT centres and clinical data on 45 SOTs carried out in the patients who previously underwent allo-HSCT. Kidney transplantations were performed, mainly, because of the drug-induced organ affection, or radiotherapy, whereas liver transplantations were made either early, for severe VOD or aGvHD, or later, due to chronic liver GVHD or cirrhosis. Survival rates and clinical outcomes are analyzed for each clinical situation in the groups. A special attention is given to the patients who received lung transplants in cases of bronchiolitis obliterans following allo-HSCT, characterized by inferior clinical outcomes and shorter survival time.
Some recent data concern allo-HSCT carried out after SOT. The hematological relapse rate was 22 %, thus allowing long-term observations. The 5-year rate for the solid organ failure was about 30%, mostly, due to graft rejection.
In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. Infections, graft rejection and other complications are frequent but usually manageable. Also, selected SOT recipients developing hematological disorders may benefit from allo-HSCT.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Description / Summary
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[NAME_EN] => Array
(
[ID] => 40
[TIMESTAMP_X] => 2015-09-03 10:49:47
[IBLOCK_ID] => 2
[NAME] => Name
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 40
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8149
[VALUE] => Allogeneic transplantation of hematopoietic stem cells and solid organ in the same patient: an update
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Allogeneic transplantation of hematopoietic stem cells and solid organ in the same patient: an update
[~DESCRIPTION] =>
[~NAME] => Name
[~DEFAULT_VALUE] =>
)
[FULL_TEXT_RU] => Array
(
[ID] => 42
[TIMESTAMP_X] => 2015-09-07 20:29:18
[IBLOCK_ID] => 2
[NAME] => Полный текст
[ACTIVE] => Y
[SORT] => 500
[CODE] => FULL_TEXT_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 42
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Полный текст
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[PDF_RU] => Array
(
[ID] => 43
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF RUS
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_RU
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 43
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8150
[VALUE] => 246
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 246
[~DESCRIPTION] =>
[~NAME] => PDF RUS
[~DEFAULT_VALUE] =>
)
[PDF_EN] => Array
(
[ID] => 44
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF ENG
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 44
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8151
[VALUE] => 247
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 247
[~DESCRIPTION] =>
[~NAME] => PDF ENG
[~DEFAULT_VALUE] =>
)
[NAME_LONG] => Array
(
[ID] => 45
[TIMESTAMP_X] => 2023-04-13 00:55:00
[IBLOCK_ID] => 2
[NAME] => Название (для очень длинных заголовков)
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_LONG
[DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 45
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 80
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Название (для очень длинных заголовков)
[~DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
)
)
Tapani Ruutu
2C, POB 705, FIN-00029 HUS, Helsinki, Finland
Phone: +358 50 555 1279
Fax: +358 9 4717 1506
E-mail: tapani.ruutu@hus.fi
Solid organ transplantation (SOT) is widely used for the treatment of end-stage organ insufficiency. Meanwhile, allogeneic hematopoietic stem cell transplantation (HSCT), by itself, causes frequent organ injury, thus being a potential indication for SOT. Due to increasing numbers of these interventions worldwide, a probability of combined disorders requiring both SOT and HSCT in the same patient is increasing. These transplantation combinations represent some clinical problems. Appropriate risk factors that may affect subsequent SOT outcomes post-HSCT include toxicity of HSCT procedure, immunosuppressive treatment for HSCT patients which may not be optimal for the SOT, potential problems caused by tissue incompatibility between host tissues, hematopoietic graft, and the solid organ graft, and emerging infections. Vice versa, in SOT recipients, the immunosuppressive medication may affect the organ functions, and increase the risk of infections. Moreover, SOT can be associated with development of hematological disorders, such as aplastic anemia, post-transplant lymphoproliferative disease, acute leukemias, etc. This paper aims at updating recent clinical experience with HSCT and SOT in the same patients. Here we discuss the survey which enrolled patients from 107 EBMT centres and clinical data on 45 SOTs carried out in the patients who previously underwent allo-HSCT. Kidney transplantations were performed, mainly, because of the drug-induced organ affection, or radiotherapy, whereas liver transplantations were made either early, for severe VOD or aGvHD, or later, due to chronic liver GVHD or cirrhosis. Survival rates and clinical outcomes are analyzed for each clinical situation in the groups. A special attention is given to the patients who received lung transplants in cases of bronchiolitis obliterans following allo-HSCT, characterized by inferior clinical outcomes and shorter survival time.
Some recent data concern allo-HSCT carried out after SOT. The hematological relapse rate was 22 %, thus allowing long-term observations. The 5-year rate for the solid organ failure was about 30%, mostly, due to graft rejection.
In summary, SOT can represent a valuable treatment strategy in HSCT recipients who develop an organ failure. Infections, graft rejection and other complications are frequent but usually manageable. Also, selected SOT recipients developing hematological disorders may benefit from allo-HSCT.
Clinical articles
Array
(
[KEYWORDS] => Array
(
[ID] => 19
[TIMESTAMP_X] => 2015-09-03 10:46:01
[IBLOCK_ID] => 2
[NAME] => Ключевые слова
[ACTIVE] => Y
[SORT] => 500
[CODE] => KEYWORDS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 19
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 4
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => Y
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => Y
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8120
[1] => 8121
[2] => 8122
)
[VALUE] => Array
(
[0] => 655
[1] => 15
[2] => 657
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 655
[1] => 15
[2] => 657
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
)
[~NAME] => Ключевые слова
[~DEFAULT_VALUE] =>
)
[SUBMITTED] => Array
(
[ID] => 20
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата подачи
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUBMITTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 20
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8000
[VALUE] => 11/10/2016 02:55:00 pm
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 11/10/2016 02:55:00 pm
[~DESCRIPTION] =>
[~NAME] => Дата подачи
[~DEFAULT_VALUE] =>
)
[ACCEPTED] => Array
(
[ID] => 21
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата принятия
[ACTIVE] => Y
[SORT] => 500
[CODE] => ACCEPTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 21
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8001
[VALUE] => 12/09/2016 02:55:00 pm
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 12/09/2016 02:55:00 pm
[~DESCRIPTION] =>
[~NAME] => Дата принятия
[~DEFAULT_VALUE] =>
)
[PUBLISHED] => Array
(
[ID] => 22
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата публикации
[ACTIVE] => Y
[SORT] => 500
[CODE] => PUBLISHED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 22
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Дата публикации
[~DEFAULT_VALUE] =>
)
[CONTACT] => Array
(
[ID] => 23
[TIMESTAMP_X] => 2015-09-03 14:43:05
[IBLOCK_ID] => 2
[NAME] => Контакт
[ACTIVE] => Y
[SORT] => 500
[CODE] => CONTACT
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 23
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8002
[VALUE] => 390
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 390
[~DESCRIPTION] =>
[~NAME] => Контакт
[~DEFAULT_VALUE] =>
)
[AUTHORS] => Array
(
[ID] => 24
[TIMESTAMP_X] => 2015-09-03 10:45:07
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHORS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 24
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8123
[1] => 8124
[2] => 8125
[3] => 8126
)
[VALUE] => Array
(
[0] => 390
[1] => 389
[2] => 654
[3] => 391
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 390
[1] => 389
[2] => 654
[3] => 391
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
)
[~NAME] => Авторы
[~DEFAULT_VALUE] =>
)
[AUTHOR_RU] => Array
(
[ID] => 25
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 25
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8007
[VALUE] => Array
(
[TEXT] => Владимир А. Добронравов<sup>1,2</sup>, Кирилл А. Смирнов<sup>1,2</sup>, Борис В. Афанасьев<sup>1,3</sup>, Ольга В. Галкина<sup>1,2</sup>, Алексей В. Смирнов<sup>1</sup>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Владимир А. Добронравов1,2, Кирилл А. Смирнов1,2, Борис В. Афанасьев1,3, Ольга В. Галкина1,2, Алексей В. Смирнов1
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_RU] => Array
(
[ID] => 26
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Организации
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 26
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8008
[VALUE] => Array
(
[TEXT] => <sup>1</sup>Первый Санкт-Петербургский государственный медицинский университет им. акад. И. П. Павлова, Россия <br>
<sup>2</sup>Научно-исследовательский институт нефрологии <br>
<sup>3</sup>Научно-исследовательский институт детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => 1Первый Санкт-Петербургский государственный медицинский университет им. акад. И. П. Павлова, Россия
2Научно-исследовательский институт нефрологии
3Научно-исследовательский институт детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Организации
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_RU] => Array
(
[ID] => 27
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Описание/Резюме
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 27
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8009
[VALUE] => Array
(
[TEXT] => <h3>ЦЕЛЬ ИССЛЕДОВАНИЯ</h3>
<p>определение значения молекулярных биомаркеров (МБМ), ассоциированных с альтерацией клеток тубулярного эпителия, в прогнозировании острого повреждения почек после трансплантации гемопоэтических стволовых клеток (ТГСК).</p>
<h3>ПАЦИЕНТЫ И МЕТОДЫ </h3>
<p>В открытое обзервационное проспективное исследование включены 90 больных (46 мужчин и 44 женщины), которым была выполнена ТГСК. В образцах мочи до ТГСК и на пяти первых неделях раннего посттрансплантационного периода определены концентрации МБМ (кальбиндина, кластерина, интерлейкина-18 (IL-18), молекулы повреждения почек-1 (KIM-1), глютатион-S-трансферазы (π-класс) (GST-π), протеина хемотаксиса моноцитов-1 (MCP-1). Параллельно мониторировали основные клинические показатели. Диагностику и стратификацию тяжести острого повреждения почек (ОПП) проводили согласно рекомендациям KDIGO (Kidney Disease Improving Global Outcomes).</p>
<h3>РЕЗУЛЬТАТЫ</h3>
<p>Доля случаев с ОПП прогрессивно увеличивалась в раннем посттрансплантационном периоде, достигнув максимума к неделе 5 после ТГСК. Повышение содержания МБМ выявляли существенно чаще случаев ОПП, причем повышение МБМ предшествовало формированию дисфункции почек (ОПП). При множественном регрессионном анализе кластерин, MCP-1 и KIM-1 имели прямую, достоверную, независимую от других анализируемых факторов связь с креатинином сыворотки крови на неделе, следующей за определением МБМ. Повышение мочевой экскреции KIM-1 и (или) MCP-1 было независимо от других клинических факторов ассоциировано с увеличением относительного риска (ОР) развития ОПП.</p>
<h3>ЗАКЛЮЧЕНИЕ</h3>
<p>Множественные ренотоксичные воздействия при ТГСК приводят к существенному и одновременному повышению экскреции с мочой БМ тубулярного повреждения, отражающему субклиническое повреждение клеток тубулярного эпителия в отсутствии критериев ОПП; оценка мочевой экскреции KIM-1 и MCP-1 представляется наиболее подходящим методом предиктивной диагностики ОПП ассоциированного с ТГСК.</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => ЦЕЛЬ ИССЛЕДОВАНИЯ
определение значения молекулярных биомаркеров (МБМ), ассоциированных с альтерацией клеток тубулярного эпителия, в прогнозировании острого повреждения почек после трансплантации гемопоэтических стволовых клеток (ТГСК).
ПАЦИЕНТЫ И МЕТОДЫ
В открытое обзервационное проспективное исследование включены 90 больных (46 мужчин и 44 женщины), которым была выполнена ТГСК. В образцах мочи до ТГСК и на пяти первых неделях раннего посттрансплантационного периода определены концентрации МБМ (кальбиндина, кластерина, интерлейкина-18 (IL-18), молекулы повреждения почек-1 (KIM-1), глютатион-S-трансферазы (π-класс) (GST-π), протеина хемотаксиса моноцитов-1 (MCP-1). Параллельно мониторировали основные клинические показатели. Диагностику и стратификацию тяжести острого повреждения почек (ОПП) проводили согласно рекомендациям KDIGO (Kidney Disease Improving Global Outcomes).
РЕЗУЛЬТАТЫ
Доля случаев с ОПП прогрессивно увеличивалась в раннем посттрансплантационном периоде, достигнув максимума к неделе 5 после ТГСК. Повышение содержания МБМ выявляли существенно чаще случаев ОПП, причем повышение МБМ предшествовало формированию дисфункции почек (ОПП). При множественном регрессионном анализе кластерин, MCP-1 и KIM-1 имели прямую, достоверную, независимую от других анализируемых факторов связь с креатинином сыворотки крови на неделе, следующей за определением МБМ. Повышение мочевой экскреции KIM-1 и (или) MCP-1 было независимо от других клинических факторов ассоциировано с увеличением относительного риска (ОР) развития ОПП.
ЗАКЛЮЧЕНИЕ
Множественные ренотоксичные воздействия при ТГСК приводят к существенному и одновременному повышению экскреции с мочой БМ тубулярного повреждения, отражающему субклиническое повреждение клеток тубулярного эпителия в отсутствии критериев ОПП; оценка мочевой экскреции KIM-1 и MCP-1 представляется наиболее подходящим методом предиктивной диагностики ОПП ассоциированного с ТГСК.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Описание/Резюме
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[DOI] => Array
(
[ID] => 28
[TIMESTAMP_X] => 2016-04-06 14:11:12
[IBLOCK_ID] => 2
[NAME] => DOI
[ACTIVE] => Y
[SORT] => 500
[CODE] => DOI
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 28
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8010
[VALUE] => 10.18620/ctt-1866-8836-2016-5-4-8-14
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10.18620/ctt-1866-8836-2016-5-4-8-14
[~DESCRIPTION] =>
[~NAME] => DOI
[~DEFAULT_VALUE] =>
)
[AUTHOR_EN] => Array
(
[ID] => 37
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Author
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 37
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8011
[VALUE] => Array
(
[TEXT] => Vladimir A. Dobronravov <sup>1,2</sup>, Kirill A. Smirnov <sup>1,2</sup>, Boris V. Afanasiev <sup>1,3</sup>, Olga V. Galkina <sup>1,2</sup>, Alexey V. Smirnov <sup>1,2</sup>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Vladimir A. Dobronravov 1,2, Kirill A. Smirnov 1,2, Boris V. Afanasiev 1,3, Olga V. Galkina 1,2, Alexey V. Smirnov 1,2
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Author
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_EN] => Array
(
[ID] => 38
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Organization
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 38
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8012
[VALUE] => Array
(
[TEXT] => Prof. Vladimir A. Dobronravov, MD, PhD, DSc, Research Institute of Nephrology, The First St. Petersburg I. Pavlov State<br>
Medical University, Russia, L.Tolstoy St. 17, 197089, St. Petersburg, Russia<br>
<br>
<sup>1</sup>First St. Petersburg I. Pavlov State Medical University, Russia,<br>
<sup>2</sup>Nephrology Research Institute,<br>
<sup>3</sup>Raisa Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia<br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Prof. Vladimir A. Dobronravov, MD, PhD, DSc, Research Institute of Nephrology, The First St. Petersburg I. Pavlov State
Medical University, Russia, L.Tolstoy St. 17, 197089, St. Petersburg, Russia
1First St. Petersburg I. Pavlov State Medical University, Russia,
2Nephrology Research Institute,
3Raisa Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Organization
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_EN] => Array
(
[ID] => 39
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Description / Summary
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 39
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8049
[VALUE] => Array
(
[TEXT] => <p>
The aim of this study was to determine clinical value of molecular biomarkers (MBM), associated with tubular epithelium damage, for the prediction of acute kidney injury (AKI) in the setting of hematopoietic stem cell transplantation (HSCT).
</p>
<h3>Patients and methods</h3>
<p>
Ninety HSCT recipients (46 males, 44 females) were enrolled into the observational prospective study. Urinary concentrations of calbindin, clusterin, IL-18 (interleukin- 18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase-π) and MCP-1 (monocyte chemoattractant protein-1) were measured in all patients before HSCT and at 5 consequent time points during early post-transplant period, along with routine clinical monitoring. AKI was defined according to the KDIGO (Kidney Disease Improving Global Outcomes) Guidelines.
</p>
<h3>Results</h3>
<p>
The incidence of AKI cases constantly increased during the observation period and reached the maximum level by the week 5 following HSCT. MBM elevation was observed more frequently than AKI and preceded the latter. Clusterin, MCP-1 and KIM-1 levels significantly correlated with subsequent serum creatinine values, measured a week after the MBM’s analysis according to multivariate linear regression models adjusted for other confounders. An increase in KIM-1 and/or MCP-1 urinary excretion was independently associated with a relative risk of AKI development. In summary, multiple renotoxic events early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation.
</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] =>
The aim of this study was to determine clinical value of molecular biomarkers (MBM), associated with tubular epithelium damage, for the prediction of acute kidney injury (AKI) in the setting of hematopoietic stem cell transplantation (HSCT).
Patients and methods
Ninety HSCT recipients (46 males, 44 females) were enrolled into the observational prospective study. Urinary concentrations of calbindin, clusterin, IL-18 (interleukin- 18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase-π) and MCP-1 (monocyte chemoattractant protein-1) were measured in all patients before HSCT and at 5 consequent time points during early post-transplant period, along with routine clinical monitoring. AKI was defined according to the KDIGO (Kidney Disease Improving Global Outcomes) Guidelines.
Results
The incidence of AKI cases constantly increased during the observation period and reached the maximum level by the week 5 following HSCT. MBM elevation was observed more frequently than AKI and preceded the latter. Clusterin, MCP-1 and KIM-1 levels significantly correlated with subsequent serum creatinine values, measured a week after the MBM’s analysis according to multivariate linear regression models adjusted for other confounders. An increase in KIM-1 and/or MCP-1 urinary excretion was independently associated with a relative risk of AKI development. In summary, multiple renotoxic events early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Description / Summary
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[NAME_EN] => Array
(
[ID] => 40
[TIMESTAMP_X] => 2015-09-03 10:49:47
[IBLOCK_ID] => 2
[NAME] => Name
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 40
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8013
[VALUE] => Subclinical tubular epithelium damage and acute kidney injury following allogeneic hematopoietic stem cell transplantation
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Subclinical tubular epithelium damage and acute kidney injury following allogeneic hematopoietic stem cell transplantation
[~DESCRIPTION] =>
[~NAME] => Name
[~DEFAULT_VALUE] =>
)
[FULL_TEXT_RU] => Array
(
[ID] => 42
[TIMESTAMP_X] => 2015-09-07 20:29:18
[IBLOCK_ID] => 2
[NAME] => Полный текст
[ACTIVE] => Y
[SORT] => 500
[CODE] => FULL_TEXT_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 42
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Полный текст
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[PDF_RU] => Array
(
[ID] => 43
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF RUS
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_RU
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 43
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8127
[VALUE] => 244
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 244
[~DESCRIPTION] =>
[~NAME] => PDF RUS
[~DEFAULT_VALUE] =>
)
[PDF_EN] => Array
(
[ID] => 44
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF ENG
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 44
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8128
[VALUE] => 245
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 245
[~DESCRIPTION] =>
[~NAME] => PDF ENG
[~DEFAULT_VALUE] =>
)
[NAME_LONG] => Array
(
[ID] => 45
[TIMESTAMP_X] => 2023-04-13 00:55:00
[IBLOCK_ID] => 2
[NAME] => Название (для очень длинных заголовков)
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_LONG
[DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 45
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 80
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Название (для очень длинных заголовков)
[~DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
)
)
Vladimir A. Dobronravov 1,2, Kirill A. Smirnov 1,2, Boris V. Afanasiev 1,3, Olga V. Galkina 1,2, Alexey V. Smirnov 1,2
Medical University, Russia, L.Tolstoy St. 17, 197089, St. Petersburg, Russia
1First St. Petersburg I. Pavlov State Medical University, Russia,
2Nephrology Research Institute,
3Raisa Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
The aim of this study was to determine clinical value of molecular biomarkers (MBM), associated with tubular epithelium damage, for the prediction of acute kidney injury (AKI) in the setting of hematopoietic stem cell transplantation (HSCT).
Patients and methods
Ninety HSCT recipients (46 males, 44 females) were enrolled into the observational prospective study. Urinary concentrations of calbindin, clusterin, IL-18 (interleukin- 18), KIM-1 (kidney injury molecule-1), GST-π (glutathione S-transferase-π) and MCP-1 (monocyte chemoattractant protein-1) were measured in all patients before HSCT and at 5 consequent time points during early post-transplant period, along with routine clinical monitoring. AKI was defined according to the KDIGO (Kidney Disease Improving Global Outcomes) Guidelines.
Results
The incidence of AKI cases constantly increased during the observation period and reached the maximum level by the week 5 following HSCT. MBM elevation was observed more frequently than AKI and preceded the latter. Clusterin, MCP-1 and KIM-1 levels significantly correlated with subsequent serum creatinine values, measured a week after the MBM’s analysis according to multivariate linear regression models adjusted for other confounders. An increase in KIM-1 and/or MCP-1 urinary excretion was independently associated with a relative risk of AKI development. In summary, multiple renotoxic events early after HSCT commonly result in markedly increased urinary excretion of distinct molecular biomarkers, reflecting subclinical tubular injury in the absence of AKI criteria. The subsequent development of AKI can be predicted by means of KIM-1 and MCP-1 urinary excretion evaluation.
Clinical articles
Array
(
[KEYWORDS] => Array
(
[ID] => 19
[TIMESTAMP_X] => 2015-09-03 10:46:01
[IBLOCK_ID] => 2
[NAME] => Ключевые слова
[ACTIVE] => Y
[SORT] => 500
[CODE] => KEYWORDS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 19
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 4
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => Y
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => Y
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8318
[1] => 8319
[2] => 8320
[3] => 8321
[4] => 8322
[5] => 8323
)
[VALUE] => Array
(
[0] => 15
[1] => 674
[2] => 675
[3] => 676
[4] => 677
[5] => 647
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
[5] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 15
[1] => 674
[2] => 675
[3] => 676
[4] => 677
[5] => 647
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
[5] =>
)
[~NAME] => Ключевые слова
[~DEFAULT_VALUE] =>
)
[SUBMITTED] => Array
(
[ID] => 20
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата подачи
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUBMITTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 20
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8282
[VALUE] => 11/02/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 11/02/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата подачи
[~DEFAULT_VALUE] =>
)
[ACCEPTED] => Array
(
[ID] => 21
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата принятия
[ACTIVE] => Y
[SORT] => 500
[CODE] => ACCEPTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 21
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8283
[VALUE] => 12/09/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 12/09/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата принятия
[~DEFAULT_VALUE] =>
)
[PUBLISHED] => Array
(
[ID] => 22
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата публикации
[ACTIVE] => Y
[SORT] => 500
[CODE] => PUBLISHED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 22
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Дата публикации
[~DEFAULT_VALUE] =>
)
[CONTACT] => Array
(
[ID] => 23
[TIMESTAMP_X] => 2015-09-03 14:43:05
[IBLOCK_ID] => 2
[NAME] => Контакт
[ACTIVE] => Y
[SORT] => 500
[CODE] => CONTACT
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 23
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Контакт
[~DEFAULT_VALUE] =>
)
[AUTHORS] => Array
(
[ID] => 24
[TIMESTAMP_X] => 2015-09-03 10:45:07
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHORS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 24
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] =>
)
[AUTHOR_RU] => Array
(
[ID] => 25
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 25
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8284
[VALUE] => Array
(
[TEXT] => Ольга С. Панкратова<sup>1</sup>, Алексей Б. Чухловин<sup>2</sup>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Ольга С. Панкратова1, Алексей Б. Чухловин2
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_RU] => Array
(
[ID] => 26
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Организации
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 26
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8285
[VALUE] => Array
(
[TEXT] => <sup>1</sup>Госпиталь Университета г. Тампере, Финляндия <br>
<sup>2</sup>НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский<br>
государственный медицинский университет им. акад. И. П.Павлова, Санкт-Петербург, Россия
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => 1Госпиталь Университета г. Тампере, Финляндия
2НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. акад. И. П.Павлова, Санкт-Петербург, Россия
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Организации
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_RU] => Array
(
[ID] => 27
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Описание/Резюме
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 27
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8286
[VALUE] => Array
(
[TEXT] => <p>
Полное истощение клеточных популяций врожденного и адаптивного иммунитета возникает после интенсивной химиотерапии и трансплантации гемопоэтических стволовых клеток (ТГСК), за чем следует постепенное восстановление иммунной системы за счет предшественников, образующихся из донорских гемопоэтических клеток, которые дифференцируются в миелоидный и лимфоидные ростки. Временная динамика восстановления иммунной системы и, в частности, отдельных клеточных популяций изучена лишь отчасти, особенно в ранние сроки после ТГСК. Например, популяции клеток врожденного иммунитета восстанавливаются в течение 1-го месяца после ТГСК, в связи с быстрым обновлением популяций гранулоцитов, моноцитов и НК-клеток. В то же время функциональное созревание субпопуляций НК-клеток и моноцитов крови может длиться до 3 мес.
</p>
<p>
Как Т-, так и В-лимфоциты восстанавливаются гораздо медленнее, чем миеломоноцитарные популяции. Есть лишь ограниченная информация об их посттрансплантационном обновлении, так как большинство исследований проводились в поздние сроки (>1 мес. после ТГСК). Так, абсолютное число CD8+ T-клеток нормализуется примерно через 4 мес., при суженном спектре Т-лимфоцитов памяти. Темпы восстановления и созревания CD4+ T во многом зависят от остаточной функции тимуса, особенно в младших возрастах. Поэтому у младших пациентов преобладает популяция «наивных» CD4+ T cell в сроки >6 мес. после ТГСК. У лиц старшего возраста с инактивацией функции тимуса общее число CD4 клеток остается низким годами после ТГСК. В то же время антивирусный клеточный иммунитет активен, начиная с ранних сроков после трансплантации. Так, цитотоксические CD8+ клетки, специфичные в отношении ЦМВ или ВЭБ, быстро размножаются в случаях реактивации этих вирусов после ТГСК.
</p>
<p>
Несмотря на восстановление абсолютных количеств В-клеток к 30-му дню после ТГСК, их функции (в т.ч. - антиген-специфическая продукция антител) снижены многие месяцы и годы после ТГСК из-за медленного восстановления зрелых популяций иммунных клеток, что напоминает постепенную эволюцию в иерархии В-клеток в организме человека.
</p>
<p>
Реактивация герпесвирусов (в основном –ЦМВ, ВЭБ и ВПГ) является известным признаком иммунодефицита. Периоды максимального риска реактивации герпесвирусов (2-3 мес. после ТГСК) соответствуют периодам функционального дефицита CD8+ и CD4+ T-клеток, а также незрелости В-клеток, что отражает их недостаточную способность к устранению лейкоцитов, пораженных герпесвирусами. Индивидуальные сроки иммунного восстановления после ТГСК зависят от возраста больного, источника донорских клеток, развития острой РТПХ после трансплантации и др. Ответ на вакцинирование – важный критерии состояния иммунитета после ТГСК – также зависит от возраста больного и сохранности функций В-клеток. Временная динамика специфического антительного ответа показывает, что больные с латентной реактивацией ЦМВ могут позже проявить сильный гуморальный иммунитет, что ведет к ограничению инфекционного процесса.
</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] =>
Полное истощение клеточных популяций врожденного и адаптивного иммунитета возникает после интенсивной химиотерапии и трансплантации гемопоэтических стволовых клеток (ТГСК), за чем следует постепенное восстановление иммунной системы за счет предшественников, образующихся из донорских гемопоэтических клеток, которые дифференцируются в миелоидный и лимфоидные ростки. Временная динамика восстановления иммунной системы и, в частности, отдельных клеточных популяций изучена лишь отчасти, особенно в ранние сроки после ТГСК. Например, популяции клеток врожденного иммунитета восстанавливаются в течение 1-го месяца после ТГСК, в связи с быстрым обновлением популяций гранулоцитов, моноцитов и НК-клеток. В то же время функциональное созревание субпопуляций НК-клеток и моноцитов крови может длиться до 3 мес.
Как Т-, так и В-лимфоциты восстанавливаются гораздо медленнее, чем миеломоноцитарные популяции. Есть лишь ограниченная информация об их посттрансплантационном обновлении, так как большинство исследований проводились в поздние сроки (>1 мес. после ТГСК). Так, абсолютное число CD8+ T-клеток нормализуется примерно через 4 мес., при суженном спектре Т-лимфоцитов памяти. Темпы восстановления и созревания CD4+ T во многом зависят от остаточной функции тимуса, особенно в младших возрастах. Поэтому у младших пациентов преобладает популяция «наивных» CD4+ T cell в сроки >6 мес. после ТГСК. У лиц старшего возраста с инактивацией функции тимуса общее число CD4 клеток остается низким годами после ТГСК. В то же время антивирусный клеточный иммунитет активен, начиная с ранних сроков после трансплантации. Так, цитотоксические CD8+ клетки, специфичные в отношении ЦМВ или ВЭБ, быстро размножаются в случаях реактивации этих вирусов после ТГСК.
Несмотря на восстановление абсолютных количеств В-клеток к 30-му дню после ТГСК, их функции (в т.ч. - антиген-специфическая продукция антител) снижены многие месяцы и годы после ТГСК из-за медленного восстановления зрелых популяций иммунных клеток, что напоминает постепенную эволюцию в иерархии В-клеток в организме человека.
Реактивация герпесвирусов (в основном –ЦМВ, ВЭБ и ВПГ) является известным признаком иммунодефицита. Периоды максимального риска реактивации герпесвирусов (2-3 мес. после ТГСК) соответствуют периодам функционального дефицита CD8+ и CD4+ T-клеток, а также незрелости В-клеток, что отражает их недостаточную способность к устранению лейкоцитов, пораженных герпесвирусами. Индивидуальные сроки иммунного восстановления после ТГСК зависят от возраста больного, источника донорских клеток, развития острой РТПХ после трансплантации и др. Ответ на вакцинирование – важный критерии состояния иммунитета после ТГСК – также зависит от возраста больного и сохранности функций В-клеток. Временная динамика специфического антительного ответа показывает, что больные с латентной реактивацией ЦМВ могут позже проявить сильный гуморальный иммунитет, что ведет к ограничению инфекционного процесса.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Описание/Резюме
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[DOI] => Array
(
[ID] => 28
[TIMESTAMP_X] => 2016-04-06 14:11:12
[IBLOCK_ID] => 2
[NAME] => DOI
[ACTIVE] => Y
[SORT] => 500
[CODE] => DOI
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 28
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8287
[VALUE] => 10.18620/ctt-1866-8836-2016-5-4-32-43
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10.18620/ctt-1866-8836-2016-5-4-32-43
[~DESCRIPTION] =>
[~NAME] => DOI
[~DEFAULT_VALUE] =>
)
[AUTHOR_EN] => Array
(
[ID] => 37
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Author
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 37
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8288
[VALUE] => Array
(
[TEXT] => <sup>1</sup>Olga S. Pankratova, <sup>2</sup>Alexei B. Chukhlovin
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => 1Olga S. Pankratova, 2Alexei B. Chukhlovin
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Author
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_EN] => Array
(
[ID] => 38
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Organization
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 38
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8289
[VALUE] => Array
(
[TEXT] => <sup>1</sup>Tampere University Hospital, Tampere, Finland <br>
<sup>2</sup>R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => 1Tampere University Hospital, Tampere, Finland
2R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Organization
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_EN] => Array
(
[ID] => 39
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Description / Summary
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 39
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8290
[VALUE] => Array
(
[TEXT] => <p>
Total depletion of innate and adaptive immune cell populations occurs after intensive chemotherapy and hematopoietic stem cell transplantation (HSCT) then followed by gradual recovery of immune populations, due to progenitors derived from donor hematopoietic cells which differentiate to myeloid and lymphoid lineages. Time dynamics of immune reconstitution and differential maturation of distinct immune populations is only partially evaluated, especially, at early terms post-transplant. E.g., innate immunity is restored within 1st month after HSCT, due to rapid reconstitution of granulocytes, monocytes, and natural killer (NK) cells. Meanwhile, functional recovery of mature NK-cell subsets and blood monocytes may continue for up to 3 months.
</p>
<p>
Both T- and B-lymphocyte pools are restored much slower than myelomonocytic populations. The available information on their post-HSCT immune recovery is limited, since most studies are performed at later terms (>1 month post-transplant). Absolute numbers of CD8+ T cells return to control values ca. 4 months post-HSCT, however, exhibiting a skewed repertoire of memory T lymphocytes. Recovery and maturation rates of CD4+ T cells largely depend on residual thymus function, especially, in young subjects. Hence, a naïve CD4+ T cell population in pediatric patients predominates over 6 months post-HSCT. In older persons with inactive thymopoiesis the total CD4 cell counts remain low for years after HSCT. Meanwhile, antiviral cellular immunity is active since early terms post-transplant. E.g., cytotoxic CD8+ cells specific for cytomegalovirus (CMV), or Epstein- Barr virus (EBV) rapidly expand in cases of CMV or EBV activation.
</p>
<p>
Despite recovery of absolute B-cell counts by day 30 post-HSCT, their functions, i.e., antigen-specific antibody production, are reduced for months and years after HSCT, due to slow restoration of mature immune cell populations, thus resemling normal evolution of B cell hierarchy in human organism.
</p>
<p>
Reactivation of herpesviruses (mostly, CMV, EBV and Herpes Simplex) is a known feature of immune deficiency. Timing of maximal herpesvirus incidence (2-3 months post-HSCT) corresponds to the period of CD8+ and CD4+ T cell functional deficiency and B cell immaturity, thus reflecting their suboptimal ability to eliminate herpesvirus-affected leukocytes. Individual terms of immune recovery after allo-HSCT depend on the patients’ age, source of donor cells, acute GvHD post-HSCT etc. Vaccination response, being a potent in vivo criterion of immune recovery in post-transplant patients, is also dependent on the subjects’ age and restored B cell functions. Time dynamics of specific antibody response shows that the patients with latent CMV reactivation may later exhibit a strong humoral immune response, thus making the infection self-limiting.
</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] =>
Total depletion of innate and adaptive immune cell populations occurs after intensive chemotherapy and hematopoietic stem cell transplantation (HSCT) then followed by gradual recovery of immune populations, due to progenitors derived from donor hematopoietic cells which differentiate to myeloid and lymphoid lineages. Time dynamics of immune reconstitution and differential maturation of distinct immune populations is only partially evaluated, especially, at early terms post-transplant. E.g., innate immunity is restored within 1st month after HSCT, due to rapid reconstitution of granulocytes, monocytes, and natural killer (NK) cells. Meanwhile, functional recovery of mature NK-cell subsets and blood monocytes may continue for up to 3 months.
Both T- and B-lymphocyte pools are restored much slower than myelomonocytic populations. The available information on their post-HSCT immune recovery is limited, since most studies are performed at later terms (>1 month post-transplant). Absolute numbers of CD8+ T cells return to control values ca. 4 months post-HSCT, however, exhibiting a skewed repertoire of memory T lymphocytes. Recovery and maturation rates of CD4+ T cells largely depend on residual thymus function, especially, in young subjects. Hence, a naïve CD4+ T cell population in pediatric patients predominates over 6 months post-HSCT. In older persons with inactive thymopoiesis the total CD4 cell counts remain low for years after HSCT. Meanwhile, antiviral cellular immunity is active since early terms post-transplant. E.g., cytotoxic CD8+ cells specific for cytomegalovirus (CMV), or Epstein- Barr virus (EBV) rapidly expand in cases of CMV or EBV activation.
Despite recovery of absolute B-cell counts by day 30 post-HSCT, their functions, i.e., antigen-specific antibody production, are reduced for months and years after HSCT, due to slow restoration of mature immune cell populations, thus resemling normal evolution of B cell hierarchy in human organism.
Reactivation of herpesviruses (mostly, CMV, EBV and Herpes Simplex) is a known feature of immune deficiency. Timing of maximal herpesvirus incidence (2-3 months post-HSCT) corresponds to the period of CD8+ and CD4+ T cell functional deficiency and B cell immaturity, thus reflecting their suboptimal ability to eliminate herpesvirus-affected leukocytes. Individual terms of immune recovery after allo-HSCT depend on the patients’ age, source of donor cells, acute GvHD post-HSCT etc. Vaccination response, being a potent in vivo criterion of immune recovery in post-transplant patients, is also dependent on the subjects’ age and restored B cell functions. Time dynamics of specific antibody response shows that the patients with latent CMV reactivation may later exhibit a strong humoral immune response, thus making the infection self-limiting.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Description / Summary
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[NAME_EN] => Array
(
[ID] => 40
[TIMESTAMP_X] => 2015-09-03 10:49:47
[IBLOCK_ID] => 2
[NAME] => Name
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 40
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8291
[VALUE] => Time course of immune recovery and viral reactivation following hematopoietic stem cell transplantation
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Time course of immune recovery and viral reactivation following hematopoietic stem cell transplantation
[~DESCRIPTION] =>
[~NAME] => Name
[~DEFAULT_VALUE] =>
)
[FULL_TEXT_RU] => Array
(
[ID] => 42
[TIMESTAMP_X] => 2015-09-07 20:29:18
[IBLOCK_ID] => 2
[NAME] => Полный текст
[ACTIVE] => Y
[SORT] => 500
[CODE] => FULL_TEXT_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 42
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Полный текст
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[PDF_RU] => Array
(
[ID] => 43
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF RUS
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_RU
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 43
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8292
[VALUE] => 257
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 257
[~DESCRIPTION] =>
[~NAME] => PDF RUS
[~DEFAULT_VALUE] =>
)
[PDF_EN] => Array
(
[ID] => 44
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF ENG
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 44
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8293
[VALUE] => 258
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 258
[~DESCRIPTION] =>
[~NAME] => PDF ENG
[~DEFAULT_VALUE] =>
)
[NAME_LONG] => Array
(
[ID] => 45
[TIMESTAMP_X] => 2023-04-13 00:55:00
[IBLOCK_ID] => 2
[NAME] => Название (для очень длинных заголовков)
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_LONG
[DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 45
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 80
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Название (для очень длинных заголовков)
[~DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
)
)
1Olga S. Pankratova, 2Alexei B. Chukhlovin
2R. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical University
Total depletion of innate and adaptive immune cell populations occurs after intensive chemotherapy and hematopoietic stem cell transplantation (HSCT) then followed by gradual recovery of immune populations, due to progenitors derived from donor hematopoietic cells which differentiate to myeloid and lymphoid lineages. Time dynamics of immune reconstitution and differential maturation of distinct immune populations is only partially evaluated, especially, at early terms post-transplant. E.g., innate immunity is restored within 1st month after HSCT, due to rapid reconstitution of granulocytes, monocytes, and natural killer (NK) cells. Meanwhile, functional recovery of mature NK-cell subsets and blood monocytes may continue for up to 3 months.
Both T- and B-lymphocyte pools are restored much slower than myelomonocytic populations. The available information on their post-HSCT immune recovery is limited, since most studies are performed at later terms (>1 month post-transplant). Absolute numbers of CD8+ T cells return to control values ca. 4 months post-HSCT, however, exhibiting a skewed repertoire of memory T lymphocytes. Recovery and maturation rates of CD4+ T cells largely depend on residual thymus function, especially, in young subjects. Hence, a naïve CD4+ T cell population in pediatric patients predominates over 6 months post-HSCT. In older persons with inactive thymopoiesis the total CD4 cell counts remain low for years after HSCT. Meanwhile, antiviral cellular immunity is active since early terms post-transplant. E.g., cytotoxic CD8+ cells specific for cytomegalovirus (CMV), or Epstein- Barr virus (EBV) rapidly expand in cases of CMV or EBV activation.
Despite recovery of absolute B-cell counts by day 30 post-HSCT, their functions, i.e., antigen-specific antibody production, are reduced for months and years after HSCT, due to slow restoration of mature immune cell populations, thus resemling normal evolution of B cell hierarchy in human organism.
Reactivation of herpesviruses (mostly, CMV, EBV and Herpes Simplex) is a known feature of immune deficiency. Timing of maximal herpesvirus incidence (2-3 months post-HSCT) corresponds to the period of CD8+ and CD4+ T cell functional deficiency and B cell immaturity, thus reflecting their suboptimal ability to eliminate herpesvirus-affected leukocytes. Individual terms of immune recovery after allo-HSCT depend on the patients’ age, source of donor cells, acute GvHD post-HSCT etc. Vaccination response, being a potent in vivo criterion of immune recovery in post-transplant patients, is also dependent on the subjects’ age and restored B cell functions. Time dynamics of specific antibody response shows that the patients with latent CMV reactivation may later exhibit a strong humoral immune response, thus making the infection self-limiting.
Clinical articles
Array
(
[KEYWORDS] => Array
(
[ID] => 19
[TIMESTAMP_X] => 2015-09-03 10:46:01
[IBLOCK_ID] => 2
[NAME] => Ключевые слова
[ACTIVE] => Y
[SORT] => 500
[CODE] => KEYWORDS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 19
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 4
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => Y
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => Y
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8365
[1] => 8366
[2] => 8367
[3] => 8368
[4] => 8369
)
[VALUE] => Array
(
[0] => 83
[1] => 680
[2] => 681
[3] => 682
[4] => 683
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 83
[1] => 680
[2] => 681
[3] => 682
[4] => 683
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
)
[~NAME] => Ключевые слова
[~DEFAULT_VALUE] =>
)
[SUBMITTED] => Array
(
[ID] => 20
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата подачи
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUBMITTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 20
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8339
[VALUE] => 10/18/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10/18/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата подачи
[~DEFAULT_VALUE] =>
)
[ACCEPTED] => Array
(
[ID] => 21
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата принятия
[ACTIVE] => Y
[SORT] => 500
[CODE] => ACCEPTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 21
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8340
[VALUE] => 11/25/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 11/25/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата принятия
[~DEFAULT_VALUE] =>
)
[PUBLISHED] => Array
(
[ID] => 22
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата публикации
[ACTIVE] => Y
[SORT] => 500
[CODE] => PUBLISHED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 22
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Дата публикации
[~DEFAULT_VALUE] =>
)
[CONTACT] => Array
(
[ID] => 23
[TIMESTAMP_X] => 2015-09-03 14:43:05
[IBLOCK_ID] => 2
[NAME] => Контакт
[ACTIVE] => Y
[SORT] => 500
[CODE] => CONTACT
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 23
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8341
[VALUE] => 679
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 679
[~DESCRIPTION] =>
[~NAME] => Контакт
[~DEFAULT_VALUE] =>
)
[AUTHORS] => Array
(
[ID] => 24
[TIMESTAMP_X] => 2015-09-03 10:45:07
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHORS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 24
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8370
)
[VALUE] => Array
(
[0] => 679
)
[DESCRIPTION] => Array
(
[0] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 679
)
[~DESCRIPTION] => Array
(
[0] =>
)
[~NAME] => Авторы
[~DEFAULT_VALUE] =>
)
[AUTHOR_RU] => Array
(
[ID] => 25
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 25
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8343
[VALUE] => Array
(
[TEXT] => Владислав С. Сергеев
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Владислав С. Сергеев
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_RU] => Array
(
[ID] => 26
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Организации
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 26
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8344
[VALUE] => Array
(
[TEXT] => НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский<br>
государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Организации
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_RU] => Array
(
[ID] => 27
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Описание/Резюме
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 27
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8345
[VALUE] => Array
(
[TEXT] => <p>
Костный мозг (КМ) содержит популяцию резидентных клеток, именуемую «мультипотентными мезенхимными стволовыми клетками» (МСК). Эти адгезивные клетки могут быть выделены и культивированы в простых питательных средах, и способны к дифференцировке в жировые или костные клетки. До сих пор не доказана возможность массовой дифференцировки МСК в гепатоциты, клетки почек или головного мозга. В то же время рассматриваются два возможных варианта их клинического применения: (1) в качестве средства иммунной модуляции при реакции «трансплантат против хозяина» (РТПХ) и аутоиммунных болезнях или (2) как потенциального источника рост-стимулирующих факторов в специализированных тканях. Эта гетерогенная популяция может поддерживать гемопоэз путем секреции факторов роста, цитокинов и других биологически активных веществ. После введения МСК способны мигрировать в поврежденные ткани, тем самым способствуя их регенерации. Однако только малая часть МСК может достичь костномозговых ниш после внутривенной инфузии. Многочисленные эксперименты с МСК на различных моделях тканевых повреждений показали их способность подавлять апоптоз, вызванный гипоксией, химическими агентами и другими разрушительными факторами. Этот защитный эффект опосредован рядом секретируемых факторов роста, как, например, ГМ-КСФ. В большом числе клинических исследований показана безопасность терапии МСК. Как клинические, так и экспериментальные исследования доказали слабую иммуногенность аллогенных МСК при их введении иммунокомпетентным реципиентам. К настоящему времени проводились инъекции культивированных МСК больным с острой РТПХ после трансплантации гемопоэтических клеток (ТГСК) и при некоторых аутоиммунных заболеваниях.
</p>
<p>
За последнее десятилетие несколько работ кались потенциальной эффективности МСК при введении в пораженные участки костей у пациентов с несовершенным остеогенезом (ОИ) – тяжелым наследственным заболеванием с нарушением структуры коллагена, приводящим к повышенной ломкости костей. Здесь представлены сведения о клиническом протоколе для оценки безопасности, иммуногенности и клиническ их эффектов МСК, вводимых больным ОИ в ходе корригирующей остеотомии. Можно допустить, что небольшая субпопуляция МСК сможет мигрировать в поврежденные участки, дифференцироваться в хондроциты и остеобласты и, тем самым, участвовать в процессах регенерации кости.
</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] =>
Костный мозг (КМ) содержит популяцию резидентных клеток, именуемую «мультипотентными мезенхимными стволовыми клетками» (МСК). Эти адгезивные клетки могут быть выделены и культивированы в простых питательных средах, и способны к дифференцировке в жировые или костные клетки. До сих пор не доказана возможность массовой дифференцировки МСК в гепатоциты, клетки почек или головного мозга. В то же время рассматриваются два возможных варианта их клинического применения: (1) в качестве средства иммунной модуляции при реакции «трансплантат против хозяина» (РТПХ) и аутоиммунных болезнях или (2) как потенциального источника рост-стимулирующих факторов в специализированных тканях. Эта гетерогенная популяция может поддерживать гемопоэз путем секреции факторов роста, цитокинов и других биологически активных веществ. После введения МСК способны мигрировать в поврежденные ткани, тем самым способствуя их регенерации. Однако только малая часть МСК может достичь костномозговых ниш после внутривенной инфузии. Многочисленные эксперименты с МСК на различных моделях тканевых повреждений показали их способность подавлять апоптоз, вызванный гипоксией, химическими агентами и другими разрушительными факторами. Этот защитный эффект опосредован рядом секретируемых факторов роста, как, например, ГМ-КСФ. В большом числе клинических исследований показана безопасность терапии МСК. Как клинические, так и экспериментальные исследования доказали слабую иммуногенность аллогенных МСК при их введении иммунокомпетентным реципиентам. К настоящему времени проводились инъекции культивированных МСК больным с острой РТПХ после трансплантации гемопоэтических клеток (ТГСК) и при некоторых аутоиммунных заболеваниях.
За последнее десятилетие несколько работ кались потенциальной эффективности МСК при введении в пораженные участки костей у пациентов с несовершенным остеогенезом (ОИ) – тяжелым наследственным заболеванием с нарушением структуры коллагена, приводящим к повышенной ломкости костей. Здесь представлены сведения о клиническом протоколе для оценки безопасности, иммуногенности и клиническ их эффектов МСК, вводимых больным ОИ в ходе корригирующей остеотомии. Можно допустить, что небольшая субпопуляция МСК сможет мигрировать в поврежденные участки, дифференцироваться в хондроциты и остеобласты и, тем самым, участвовать в процессах регенерации кости.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Описание/Резюме
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[DOI] => Array
(
[ID] => 28
[TIMESTAMP_X] => 2016-04-06 14:11:12
[IBLOCK_ID] => 2
[NAME] => DOI
[ACTIVE] => Y
[SORT] => 500
[CODE] => DOI
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 28
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8346
[VALUE] => 10.18620/ctt-1866-8836-2016-5-4-44-55
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10.18620/ctt-1866-8836-2016-5-4-44-55
[~DESCRIPTION] =>
[~NAME] => DOI
[~DEFAULT_VALUE] =>
)
[AUTHOR_EN] => Array
(
[ID] => 37
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Author
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 37
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8347
[VALUE] => Array
(
[TEXT] => Vladislav S. Sergeev
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Vladislav S. Sergeev
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Author
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_EN] => Array
(
[ID] => 38
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Organization
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 38
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8348
[VALUE] => Array
(
[TEXT] => R. Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical<br>
University, St. Petersburg, Russia<br>
<br>
Dr. V. S. Sergeev, St. Petersburg State I. Pavlov Medical University,<br>
L. Tolstoy St. 6-8, 197022, St. Petersburg, Russia<br>
<br>
Phone: +7 (911) 129 7580<br>
E-mail: <a href="mailto:sergeev.vlad@gmail.com">sergeev.vlad@gmail.com</a><br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => R. Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, The St. Petersburg State I. Pavlov Medical
University, St. Petersburg, Russia
Dr. V. S. Sergeev, St. Petersburg State I. Pavlov Medical University,
L. Tolstoy St. 6-8, 197022, St. Petersburg, Russia
Phone: +7 (911) 129 7580
E-mail: sergeev.vlad@gmail.com
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Organization
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_EN] => Array
(
[ID] => 39
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Description / Summary
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 39
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8349
[VALUE] => Array
(
[TEXT] => <p>
Bone marrow (BM) contains a small resident cell population referred to as ‘multipotent mesenchymal stem cells’ (MSC). These adherent cells could be isolated and expanded in simple culture media and may differentiate in adipogenic or osteogenic pathway. So far an opportunity of MSC differentiation to hepatocytes, brain, or renal cells is not proven yet. Meanwhile, two potential clinical applications are considered for MSC: (1) as a tool for immune modulation in graft-versus-host disease (GVHD) and autoimmune diseases, or, (2) as a potential source of growth-promoting factors in specialized tissues. This heterogenous population may support hematopoiesis by secreting growth factors, cytokines and other biologically active substances. Upon injection, MSCs are able to migrate into damaged tissues, thus promoting their repair. However, only small MSC fraction may reach bone marrow niches following intravenous infusion. Multiple experiments with MSCs in different injury models show their ability to suppress apoptosis initiated by hypoxia, chemical agents/acidity and other deteriorating factors. This protective effect is mediated by a number of secreted growth factors, e.g., granulocyte-macrophage colony- stimulating growth factor (GM-CSF). A big number of clinical trials show high level of safety for the MSC therapy. Both clinical and experimental studies demonstrated only weak immunogenic effects of allogeneic MSC upon injection into immunocompetent recipients. At the present time, injections of in vitro expanded MSCs were performed in the patients developing acute GVHD after hematopoietic stem cell transplantation (HSCT), and in some autoimmune disorders.
</p>
<p>
Over last decade, several studies concerned potentially curative effects of MSCs injected into affected bone areas in the patients with osteogenesis imperfecta (OI), a severe inherited disease with altered collagen structure resulting into increased bone fragility. Here we present a synopsis of clinical protocol aimed for assessing safety, immunogenicity, and clinical effects of MSC injected to the OI patients during corrective osteotomy. One may suggest that a minor MSC subpopulation may migrate to the damaged areas differentiating to chondrocytes and osteoblasts, and, hence, contributing to the bone repair.
</p>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] =>
Bone marrow (BM) contains a small resident cell population referred to as ‘multipotent mesenchymal stem cells’ (MSC). These adherent cells could be isolated and expanded in simple culture media and may differentiate in adipogenic or osteogenic pathway. So far an opportunity of MSC differentiation to hepatocytes, brain, or renal cells is not proven yet. Meanwhile, two potential clinical applications are considered for MSC: (1) as a tool for immune modulation in graft-versus-host disease (GVHD) and autoimmune diseases, or, (2) as a potential source of growth-promoting factors in specialized tissues. This heterogenous population may support hematopoiesis by secreting growth factors, cytokines and other biologically active substances. Upon injection, MSCs are able to migrate into damaged tissues, thus promoting their repair. However, only small MSC fraction may reach bone marrow niches following intravenous infusion. Multiple experiments with MSCs in different injury models show their ability to suppress apoptosis initiated by hypoxia, chemical agents/acidity and other deteriorating factors. This protective effect is mediated by a number of secreted growth factors, e.g., granulocyte-macrophage colony- stimulating growth factor (GM-CSF). A big number of clinical trials show high level of safety for the MSC therapy. Both clinical and experimental studies demonstrated only weak immunogenic effects of allogeneic MSC upon injection into immunocompetent recipients. At the present time, injections of in vitro expanded MSCs were performed in the patients developing acute GVHD after hematopoietic stem cell transplantation (HSCT), and in some autoimmune disorders.
Over last decade, several studies concerned potentially curative effects of MSCs injected into affected bone areas in the patients with osteogenesis imperfecta (OI), a severe inherited disease with altered collagen structure resulting into increased bone fragility. Here we present a synopsis of clinical protocol aimed for assessing safety, immunogenicity, and clinical effects of MSC injected to the OI patients during corrective osteotomy. One may suggest that a minor MSC subpopulation may migrate to the damaged areas differentiating to chondrocytes and osteoblasts, and, hence, contributing to the bone repair.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Description / Summary
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[NAME_EN] => Array
(
[ID] => 40
[TIMESTAMP_X] => 2015-09-03 10:49:47
[IBLOCK_ID] => 2
[NAME] => Name
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 40
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8350
[VALUE] => Potential clinical applications of bone marrow-derived mesenchymal stem cells in bone regeneration
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Potential clinical applications of bone marrow-derived mesenchymal stem cells in bone regeneration
[~DESCRIPTION] =>
[~NAME] => Name
[~DEFAULT_VALUE] =>
)
[FULL_TEXT_RU] => Array
(
[ID] => 42
[TIMESTAMP_X] => 2015-09-07 20:29:18
[IBLOCK_ID] => 2
[NAME] => Полный текст
[ACTIVE] => Y
[SORT] => 500
[CODE] => FULL_TEXT_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 42
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Полный текст
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[PDF_RU] => Array
(
[ID] => 43
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF RUS
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_RU
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 43
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8357
[VALUE] => 263
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 263
[~DESCRIPTION] =>
[~NAME] => PDF RUS
[~DEFAULT_VALUE] =>
)
[PDF_EN] => Array
(
[ID] => 44
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF ENG
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 44
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8358
[VALUE] => 264
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 264
[~DESCRIPTION] =>
[~NAME] => PDF ENG
[~DEFAULT_VALUE] =>
)
[NAME_LONG] => Array
(
[ID] => 45
[TIMESTAMP_X] => 2023-04-13 00:55:00
[IBLOCK_ID] => 2
[NAME] => Название (для очень длинных заголовков)
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_LONG
[DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 45
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 80
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Название (для очень длинных заголовков)
[~DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
)
)
Vladislav S. Sergeev
University, St. Petersburg, Russia
Dr. V. S. Sergeev, St. Petersburg State I. Pavlov Medical University,
L. Tolstoy St. 6-8, 197022, St. Petersburg, Russia
Phone: +7 (911) 129 7580
E-mail: sergeev.vlad@gmail.com
Bone marrow (BM) contains a small resident cell population referred to as ‘multipotent mesenchymal stem cells’ (MSC). These adherent cells could be isolated and expanded in simple culture media and may differentiate in adipogenic or osteogenic pathway. So far an opportunity of MSC differentiation to hepatocytes, brain, or renal cells is not proven yet. Meanwhile, two potential clinical applications are considered for MSC: (1) as a tool for immune modulation in graft-versus-host disease (GVHD) and autoimmune diseases, or, (2) as a potential source of growth-promoting factors in specialized tissues. This heterogenous population may support hematopoiesis by secreting growth factors, cytokines and other biologically active substances. Upon injection, MSCs are able to migrate into damaged tissues, thus promoting their repair. However, only small MSC fraction may reach bone marrow niches following intravenous infusion. Multiple experiments with MSCs in different injury models show their ability to suppress apoptosis initiated by hypoxia, chemical agents/acidity and other deteriorating factors. This protective effect is mediated by a number of secreted growth factors, e.g., granulocyte-macrophage colony- stimulating growth factor (GM-CSF). A big number of clinical trials show high level of safety for the MSC therapy. Both clinical and experimental studies demonstrated only weak immunogenic effects of allogeneic MSC upon injection into immunocompetent recipients. At the present time, injections of in vitro expanded MSCs were performed in the patients developing acute GVHD after hematopoietic stem cell transplantation (HSCT), and in some autoimmune disorders.
Over last decade, several studies concerned potentially curative effects of MSCs injected into affected bone areas in the patients with osteogenesis imperfecta (OI), a severe inherited disease with altered collagen structure resulting into increased bone fragility. Here we present a synopsis of clinical protocol aimed for assessing safety, immunogenicity, and clinical effects of MSC injected to the OI patients during corrective osteotomy. One may suggest that a minor MSC subpopulation may migrate to the damaged areas differentiating to chondrocytes and osteoblasts, and, hence, contributing to the bone repair.
Experimental studies
Array
(
[KEYWORDS] => Array
(
[ID] => 19
[TIMESTAMP_X] => 2015-09-03 10:46:01
[IBLOCK_ID] => 2
[NAME] => Ключевые слова
[ACTIVE] => Y
[SORT] => 500
[CODE] => KEYWORDS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 19
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 4
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => Y
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => Y
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8467
[1] => 8468
[2] => 8469
[3] => 8470
)
[VALUE] => Array
(
[0] => 692
[1] => 693
[2] => 607
[3] => 282
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 692
[1] => 693
[2] => 607
[3] => 282
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
)
[~NAME] => Ключевые слова
[~DEFAULT_VALUE] =>
)
[SUBMITTED] => Array
(
[ID] => 20
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата подачи
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUBMITTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 20
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8414
[VALUE] => 09/14/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 09/14/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата подачи
[~DEFAULT_VALUE] =>
)
[ACCEPTED] => Array
(
[ID] => 21
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата принятия
[ACTIVE] => Y
[SORT] => 500
[CODE] => ACCEPTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 21
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8415
[VALUE] => 11/25/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 11/25/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата принятия
[~DEFAULT_VALUE] =>
)
[PUBLISHED] => Array
(
[ID] => 22
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата публикации
[ACTIVE] => Y
[SORT] => 500
[CODE] => PUBLISHED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 22
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Дата публикации
[~DEFAULT_VALUE] =>
)
[CONTACT] => Array
(
[ID] => 23
[TIMESTAMP_X] => 2015-09-03 14:43:05
[IBLOCK_ID] => 2
[NAME] => Контакт
[ACTIVE] => Y
[SORT] => 500
[CODE] => CONTACT
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 23
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8416
[VALUE] => 691
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 691
[~DESCRIPTION] =>
[~NAME] => Контакт
[~DEFAULT_VALUE] =>
)
[AUTHORS] => Array
(
[ID] => 24
[TIMESTAMP_X] => 2015-09-03 10:45:07
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHORS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 24
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8471
)
[VALUE] => Array
(
[0] => 691
)
[DESCRIPTION] => Array
(
[0] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 691
)
[~DESCRIPTION] => Array
(
[0] =>
)
[~NAME] => Авторы
[~DEFAULT_VALUE] =>
)
[AUTHOR_RU] => Array
(
[ID] => 25
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 25
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8418
[VALUE] => Array
(
[TEXT] => <sup>1</sup>Юрий А. Серов, <sup>1</sup>Ильдар M. Бархатов, <sup>1</sup>Антон С. Климов, <sup>2</sup>Андрей С. Беркос
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => 1Юрий А. Серов, 1Ильдар M. Бархатов, 1Антон С. Климов, 2Андрей С. Беркос
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_RU] => Array
(
[ID] => 26
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Организации
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 26
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8419
[VALUE] => Array
(
[TEXT] => <sup>1</sup>НИИ детской онкологии, гематологии и трансплантации им. Р. М. Горбачевой, Первый Санкт-Петербургский<br>
государственный медицинский университет им. И. П. Павлова, Санкт-Петебург, Россия;<br>
<sup>2</sup>Российский НИИ гематологии и трансфузиологии, Санкт-Петербург, Россия<br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => 1НИИ детской онкологии, гематологии и трансплантации им. Р. М. Горбачевой, Первый Санкт-Петербургский
государственный медицинский университет им. И. П. Павлова, Санкт-Петебург, Россия;
2Российский НИИ гематологии и трансфузиологии, Санкт-Петербург, Россия
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Организации
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_RU] => Array
(
[ID] => 27
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Описание/Резюме
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 27
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8420
[VALUE] => Array
(
[TEXT] => Область генома, кодирующая лейкоциты антигенов человека (HLA) – это сегмент длиной 3,6 миллиона пар оснований на хромосоме 6(p21). Сложная по антигенному составу система HLA содержит важные молекулы, участвующие в трансплантационном иммунитете. Специфические генные последовательности генов HLA довольно сложно расшифровать с помощью классических методов прямого секвенирования в связи с их техническими ограничениями, сложным составом генов HLA, и возрастающим числом новых аллелей, что требует новых методов генотипирования. Секвенирование следуюшего поколения (NGS) является методом, который может обеспечить полное решение проблемы HLA-типирования. За последнее десятилетие разработка NGS обеспечила более легкий способ полногеномного анализа у человека, в том числе для типирования генов HLA. Несколько вариантов высокопроизводительных методов оценки HLA, основанные на мультиплексных аналитических технологиях NGS были разработаны на основе различных технологических платформ. В этом обзоре мы обсуждаем возможные области применения и прогресс в оборудовании для NGS в плане глубокого типирования системы HLA, с особым вниманием к будущим аспектам его клинического использования. Общеизвестный полиморфизм генов HLA и минимальные межаллельные различия представляют собой особую проблему. Поэтому некоторые вопросы, связанные с биоинформатикой и углубленной обработкой данных для более эффективного анализа HLA в контексте оценки совместимости донора и реципиента. В этом плане различными производителями предложены специализированные компьютерные программы для анализа больших баз данных, получаемых посредством технологий NGS. Наконец, эти высокопроизводительные подходы позволяют минимизировать финансовые затраты на 1 образец, особенно в крупных HLA-лабораториях, где соответствующие расходы на NGS-исследования стали теперь значительно затрат, возникающих при использовании классических технологий секвенирования по Сэнгеру.
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Область генома, кодирующая лейкоциты антигенов человека (HLA) – это сегмент длиной 3,6 миллиона пар оснований на хромосоме 6(p21). Сложная по антигенному составу система HLA содержит важные молекулы, участвующие в трансплантационном иммунитете. Специфические генные последовательности генов HLA довольно сложно расшифровать с помощью классических методов прямого секвенирования в связи с их техническими ограничениями, сложным составом генов HLA, и возрастающим числом новых аллелей, что требует новых методов генотипирования. Секвенирование следуюшего поколения (NGS) является методом, который может обеспечить полное решение проблемы HLA-типирования. За последнее десятилетие разработка NGS обеспечила более легкий способ полногеномного анализа у человека, в том числе для типирования генов HLA. Несколько вариантов высокопроизводительных методов оценки HLA, основанные на мультиплексных аналитических технологиях NGS были разработаны на основе различных технологических платформ. В этом обзоре мы обсуждаем возможные области применения и прогресс в оборудовании для NGS в плане глубокого типирования системы HLA, с особым вниманием к будущим аспектам его клинического использования. Общеизвестный полиморфизм генов HLA и минимальные межаллельные различия представляют собой особую проблему. Поэтому некоторые вопросы, связанные с биоинформатикой и углубленной обработкой данных для более эффективного анализа HLA в контексте оценки совместимости донора и реципиента. В этом плане различными производителями предложены специализированные компьютерные программы для анализа больших баз данных, получаемых посредством технологий NGS. Наконец, эти высокопроизводительные подходы позволяют минимизировать финансовые затраты на 1 образец, особенно в крупных HLA-лабораториях, где соответствующие расходы на NGS-исследования стали теперь значительно затрат, возникающих при использовании классических технологий секвенирования по Сэнгеру.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Описание/Резюме
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[DOI] => Array
(
[ID] => 28
[TIMESTAMP_X] => 2016-04-06 14:11:12
[IBLOCK_ID] => 2
[NAME] => DOI
[ACTIVE] => Y
[SORT] => 500
[CODE] => DOI
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 28
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8421
[VALUE] => 10.18620/ctt-1866-8836-2016-5-4-63-70
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10.18620/ctt-1866-8836-2016-5-4-63-70
[~DESCRIPTION] =>
[~NAME] => DOI
[~DEFAULT_VALUE] =>
)
[AUTHOR_EN] => Array
(
[ID] => 37
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Author
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 37
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8422
[VALUE] => Array
(
[TEXT] => <sup>1</sup>Youri A. Serov, <sup>1</sup>Ildar M. Barkhatov, <sup>1</sup>Anton S. Klimov, <sup>2</sup>Andrey S. Berkos
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => 1Youri A. Serov, 1Ildar M. Barkhatov, 1Anton S. Klimov, 2Andrey S. Berkos
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Author
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_EN] => Array
(
[ID] => 38
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Organization
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 38
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8423
[VALUE] => Array
(
[TEXT] => <sup>1</sup>Research Institute of Children Oncology, Hematology and Transplantation, The First State I. Pavlov Medical University,<br>
St. Petersburg, Russia<br>
<sup>2</sup>Russian Research Institute of Hematology and Transfusiology, Federal Medical-Biological Agency, St. Petersburg, Russia<br>
<br>
Youri A. Serov, MD, Ph. D., CHS(ABHI), The Head of Biobank<br>
Research Unit, Raisa Gorbacheva Memorial Institute of Children’s<br>
Hematology, Oncology and Transplantation, The First<br>
St. Petersburg I. P. Pavlov State Medical University, Leo Tolstoy<br>
str. 6-8, 197022, St. Petersburg, Russia<br>
<br>
Phone: +7(812) 338-6260 (office); +7(981) 973-8041 (mob.)<br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => 1Research Institute of Children Oncology, Hematology and Transplantation, The First State I. Pavlov Medical University,
St. Petersburg, Russia
2Russian Research Institute of Hematology and Transfusiology, Federal Medical-Biological Agency, St. Petersburg, Russia
Youri A. Serov, MD, Ph. D., CHS(ABHI), The Head of Biobank
Research Unit, Raisa Gorbacheva Memorial Institute of Children’s
Hematology, Oncology and Transplantation, The First
St. Petersburg I. P. Pavlov State Medical University, Leo Tolstoy
str. 6-8, 197022, St. Petersburg, Russia
Phone: +7(812) 338-6260 (office); +7(981) 973-8041 (mob.)
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Organization
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_EN] => Array
(
[ID] => 39
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Description / Summary
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 39
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8424
[VALUE] => Array
(
[TEXT] => The region encoding human leukocyte antigens (HLA) is a 3.6-Mb segment at chromosome 6p21. A highly complex HLA system comprises important molecules involved in transplantation immunity, classical direct sequencing methods making it very complicating to resolve the genomic specificities of HLA genes, due of the complex nature of them. Current limitations of existing methods and the increasing rate of new alleles, strongly demanding for a new methodology approach for the HLA genotyping. Next-Generation Sequencing (NGS) is a method that can provide a complete solution to the HLA typing problem. Last decade, the development of (NGS) has shown the easy way for whole-genome analysis in individuals. HLA typing has also benefited from NGS technologies. Several high-throughput HLA- NGS-based typing methods were developed using different technological platforms. In this review, we summarize the possible applications and instrumentation developments of NGS in the in-depth HLA typing, with a focus on future clinical applications. However, some issues of bioinformatics and data mining remain an obstacle to more effective analysis of HLA analysis in the context of donor/recipient compatibility assessment.
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => The region encoding human leukocyte antigens (HLA) is a 3.6-Mb segment at chromosome 6p21. A highly complex HLA system comprises important molecules involved in transplantation immunity, classical direct sequencing methods making it very complicating to resolve the genomic specificities of HLA genes, due of the complex nature of them. Current limitations of existing methods and the increasing rate of new alleles, strongly demanding for a new methodology approach for the HLA genotyping. Next-Generation Sequencing (NGS) is a method that can provide a complete solution to the HLA typing problem. Last decade, the development of (NGS) has shown the easy way for whole-genome analysis in individuals. HLA typing has also benefited from NGS technologies. Several high-throughput HLA- NGS-based typing methods were developed using different technological platforms. In this review, we summarize the possible applications and instrumentation developments of NGS in the in-depth HLA typing, with a focus on future clinical applications. However, some issues of bioinformatics and data mining remain an obstacle to more effective analysis of HLA analysis in the context of donor/recipient compatibility assessment.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Description / Summary
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[NAME_EN] => Array
(
[ID] => 40
[TIMESTAMP_X] => 2015-09-03 10:49:47
[IBLOCK_ID] => 2
[NAME] => Name
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 40
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8425
[VALUE] => Current methods and opportunities of next-generation sequencing (NGS) for HLA typing
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Current methods and opportunities of next-generation sequencing (NGS) for HLA typing
[~DESCRIPTION] =>
[~NAME] => Name
[~DEFAULT_VALUE] =>
)
[FULL_TEXT_RU] => Array
(
[ID] => 42
[TIMESTAMP_X] => 2015-09-07 20:29:18
[IBLOCK_ID] => 2
[NAME] => Полный текст
[ACTIVE] => Y
[SORT] => 500
[CODE] => FULL_TEXT_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 42
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Полный текст
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[PDF_RU] => Array
(
[ID] => 43
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF RUS
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_RU
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 43
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8472
[VALUE] => 278
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 278
[~DESCRIPTION] =>
[~NAME] => PDF RUS
[~DEFAULT_VALUE] =>
)
[PDF_EN] => Array
(
[ID] => 44
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF ENG
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 44
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8473
[VALUE] => 279
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 279
[~DESCRIPTION] =>
[~NAME] => PDF ENG
[~DEFAULT_VALUE] =>
)
[NAME_LONG] => Array
(
[ID] => 45
[TIMESTAMP_X] => 2023-04-13 00:55:00
[IBLOCK_ID] => 2
[NAME] => Название (для очень длинных заголовков)
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_LONG
[DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 45
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 80
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Название (для очень длинных заголовков)
[~DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
)
)
1Youri A. Serov, 1Ildar M. Barkhatov, 1Anton S. Klimov, 2Andrey S. Berkos
St. Petersburg, Russia
2Russian Research Institute of Hematology and Transfusiology, Federal Medical-Biological Agency, St. Petersburg, Russia
Youri A. Serov, MD, Ph. D., CHS(ABHI), The Head of Biobank
Research Unit, Raisa Gorbacheva Memorial Institute of Children’s
Hematology, Oncology and Transplantation, The First
St. Petersburg I. P. Pavlov State Medical University, Leo Tolstoy
str. 6-8, 197022, St. Petersburg, Russia
Phone: +7(812) 338-6260 (office); +7(981) 973-8041 (mob.)
Experimental studies
Array
(
[KEYWORDS] => Array
(
[ID] => 19
[TIMESTAMP_X] => 2015-09-03 10:46:01
[IBLOCK_ID] => 2
[NAME] => Ключевые слова
[ACTIVE] => Y
[SORT] => 500
[CODE] => KEYWORDS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 19
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 4
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => Y
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => Y
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8396
[1] => 8397
[2] => 8398
[3] => 8399
[4] => 8400
)
[VALUE] => Array
(
[0] => 686
[1] => 687
[2] => 688
[3] => 689
[4] => 690
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 686
[1] => 687
[2] => 688
[3] => 689
[4] => 690
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
[4] =>
)
[~NAME] => Ключевые слова
[~DEFAULT_VALUE] =>
)
[SUBMITTED] => Array
(
[ID] => 20
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата подачи
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUBMITTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 20
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8371
[VALUE] => 11/28/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 11/28/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата подачи
[~DEFAULT_VALUE] =>
)
[ACCEPTED] => Array
(
[ID] => 21
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата принятия
[ACTIVE] => Y
[SORT] => 500
[CODE] => ACCEPTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 21
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8372
[VALUE] => 12/09/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 12/09/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата принятия
[~DEFAULT_VALUE] =>
)
[PUBLISHED] => Array
(
[ID] => 22
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата публикации
[ACTIVE] => Y
[SORT] => 500
[CODE] => PUBLISHED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 22
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Дата публикации
[~DEFAULT_VALUE] =>
)
[CONTACT] => Array
(
[ID] => 23
[TIMESTAMP_X] => 2015-09-03 14:43:05
[IBLOCK_ID] => 2
[NAME] => Контакт
[ACTIVE] => Y
[SORT] => 500
[CODE] => CONTACT
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 23
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8373
[VALUE] => 433
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 433
[~DESCRIPTION] =>
[~NAME] => Контакт
[~DEFAULT_VALUE] =>
)
[AUTHORS] => Array
(
[ID] => 24
[TIMESTAMP_X] => 2015-09-03 10:45:07
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHORS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 24
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8401
)
[VALUE] => Array
(
[0] => 433
)
[DESCRIPTION] => Array
(
[0] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 433
)
[~DESCRIPTION] => Array
(
[0] =>
)
[~NAME] => Авторы
[~DEFAULT_VALUE] =>
)
[AUTHOR_RU] => Array
(
[ID] => 25
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 25
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8388
[VALUE] => Array
(
[TEXT] => Герард Вагемакер
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Герард Вагемакер
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_RU] => Array
(
[ID] => 26
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Организации
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 26
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8389
[VALUE] => Array
(
[TEXT] => Университет Эразмус, Роттердам, Нидерланды; Центр исследования и разработки стволовых клеток, Университет<br>
Хашеттепе, Анкара, Турция; НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый<br>
Санкт-Петербургский государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия<br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Университет Эразмус, Роттердам, Нидерланды; Центр исследования и разработки стволовых клеток, Университет
Хашеттепе, Анкара, Турция; НИИ детской онкологии, гематологии и трансплантологии им. Р. М. Горбачевой, Первый
Санкт-Петербургский государственный медицинский университет им. И. П. Павлова, Санкт-Петербург, Россия
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Организации
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_RU] => Array
(
[ID] => 27
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Описание/Резюме
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 27
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8390
[VALUE] => Array
(
[TEXT] => Редкие болезни поражают, в целом, миллионы людей во всем мире. Многие из этих являются наследственными заболеваниями, ведущими к инвалидности и требующими дорогостоящего ухода. Генная терапия гемопоэтическими стволовыми клетками (ГТГСК) разработана за последние 20 лет. На современном уровне генная терапия выполнима при заболеваниях, для которых (1) есть идентифицированный генетический дефект, (2) диагноз ставится достаточно рано для действенного терапевтического вмешательства, (3) имеется специфическая экспериментальная модель для оценки эффективности и безопасности лечения. Соответствующие терапевтические трансгены должны также отвечать определенным биологическим критериям. Лентивирусные векторы третьего поколения выполнены самоинактивирующимися (SIN), путем делеции энхансерных участков из LTR-последовательностей, тем самым снижая риск воздействия на соседние гены, что приводит к достаточным уровням безопасности. В настоящее время лентивирусная ГТГСК вступила в фазу начального клинического внедрения для лечения иммунодефицитов и лизосомных болезней накопления. Мы обсуждаем начальные клинические испытания с применением этих векторов для некоторых метаболических болезней накопления, которые включают адренолейкодистрофию, метахроматическую лейкодистрофию, синдром Гурлер (MPS I), Помпе (GSD II), и болезнь Фабри. Данный краткий обзор обобщает развитие и современное клиническое внедрение этих подходов.
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Редкие болезни поражают, в целом, миллионы людей во всем мире. Многие из этих являются наследственными заболеваниями, ведущими к инвалидности и требующими дорогостоящего ухода. Генная терапия гемопоэтическими стволовыми клетками (ГТГСК) разработана за последние 20 лет. На современном уровне генная терапия выполнима при заболеваниях, для которых (1) есть идентифицированный генетический дефект, (2) диагноз ставится достаточно рано для действенного терапевтического вмешательства, (3) имеется специфическая экспериментальная модель для оценки эффективности и безопасности лечения. Соответствующие терапевтические трансгены должны также отвечать определенным биологическим критериям. Лентивирусные векторы третьего поколения выполнены самоинактивирующимися (SIN), путем делеции энхансерных участков из LTR-последовательностей, тем самым снижая риск воздействия на соседние гены, что приводит к достаточным уровням безопасности. В настоящее время лентивирусная ГТГСК вступила в фазу начального клинического внедрения для лечения иммунодефицитов и лизосомных болезней накопления. Мы обсуждаем начальные клинические испытания с применением этих векторов для некоторых метаболических болезней накопления, которые включают адренолейкодистрофию, метахроматическую лейкодистрофию, синдром Гурлер (MPS I), Помпе (GSD II), и болезнь Фабри. Данный краткий обзор обобщает развитие и современное клиническое внедрение этих подходов.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Описание/Резюме
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[DOI] => Array
(
[ID] => 28
[TIMESTAMP_X] => 2016-04-06 14:11:12
[IBLOCK_ID] => 2
[NAME] => DOI
[ACTIVE] => Y
[SORT] => 500
[CODE] => DOI
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 28
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8375
[VALUE] => 10.18620/ctt-1866-8836-2016-5-4-56-62
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10.18620/ctt-1866-8836-2016-5-4-56-62
[~DESCRIPTION] =>
[~NAME] => DOI
[~DEFAULT_VALUE] =>
)
[AUTHOR_EN] => Array
(
[ID] => 37
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Author
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 37
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8391
[VALUE] => Array
(
[TEXT] => Gerard Wagemaker*
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Gerard Wagemaker*
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Author
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_EN] => Array
(
[ID] => 38
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Organization
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 38
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8392
[VALUE] => Array
(
[TEXT] => Erasmus University Rotterdam, The Netherlands; Center for Stem Cell Research and Development, Hacettepe University, Ankara, Turkey;<br>
Raisa Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia<br>
<br>
Gerard Wagemaker, PhD, Professor, Erasmus University Rotterdam,<br>
c/o PO Box 37048<br>
3005 LA Rotterdam, The Netherlands<br>
<br>
Phone: +31-6-51619585<br>
E-mail: <a href="mailto:g.wagemaker@genetherapy.nl">g.wagemaker@genetherapy.nl</a><br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Erasmus University Rotterdam, The Netherlands; Center for Stem Cell Research and Development, Hacettepe University, Ankara, Turkey;
Raisa Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
Gerard Wagemaker, PhD, Professor, Erasmus University Rotterdam,
c/o PO Box 37048
3005 LA Rotterdam, The Netherlands
Phone: +31-6-51619585
E-mail: g.wagemaker@genetherapy.nl
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Organization
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_EN] => Array
(
[ID] => 39
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Description / Summary
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 39
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8393
[VALUE] => Array
(
[TEXT] => Rare diseases affect millions of people worldwide. Many of those are inherited disorders resulting in chronic disability and requiring cost-intensive care. Hematopoietic stem cell gene therapy has been developed over more than 20 years. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation. Appropriate therapeutic transgenes should also comply with certain biological criteria. Third-generation lentiviral vectors have been made self-inactivating (SIN) by deletion of enhancer regions from the LTR sequences thus reducing the risk of influencing nearby genes, resulting in favorable safety profiles. At the present time, lentiviral hematopoietic stem cell gene therapy has entered the stage of initial clinical implementation for immune deficiencies and lysosomal storage disorders. We discuss initial clinical trials using these vectors for selected metabolic storage disorders, which include adrenoleukodystrophy, metachromatic leukodystrophy, Hurler (MPS I), Pompe (GSD II), and Fabry diseases. This brief review summarizes the development and current clinical implementation of these approaches.
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Rare diseases affect millions of people worldwide. Many of those are inherited disorders resulting in chronic disability and requiring cost-intensive care. Hematopoietic stem cell gene therapy has been developed over more than 20 years. At the state of the art, gene therapy is within reach for diseases in which (i) the genetic defect is identified, (ii) the diagnosis is made sufficiently early for a meaningful therapeutic intervention, (iii) a specific animal model is available for efficacy and safety evaluation. Appropriate therapeutic transgenes should also comply with certain biological criteria. Third-generation lentiviral vectors have been made self-inactivating (SIN) by deletion of enhancer regions from the LTR sequences thus reducing the risk of influencing nearby genes, resulting in favorable safety profiles. At the present time, lentiviral hematopoietic stem cell gene therapy has entered the stage of initial clinical implementation for immune deficiencies and lysosomal storage disorders. We discuss initial clinical trials using these vectors for selected metabolic storage disorders, which include adrenoleukodystrophy, metachromatic leukodystrophy, Hurler (MPS I), Pompe (GSD II), and Fabry diseases. This brief review summarizes the development and current clinical implementation of these approaches.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Description / Summary
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[NAME_EN] => Array
(
[ID] => 40
[TIMESTAMP_X] => 2015-09-03 10:49:47
[IBLOCK_ID] => 2
[NAME] => Name
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 40
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8376
[VALUE] => Lentiviral Hematopoietic Stem Cell Gene Therapy in Inherited Immune and Lysosomal Enzyme Deficiencies
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Lentiviral Hematopoietic Stem Cell Gene Therapy in Inherited Immune and Lysosomal Enzyme Deficiencies
[~DESCRIPTION] =>
[~NAME] => Name
[~DEFAULT_VALUE] =>
)
[FULL_TEXT_RU] => Array
(
[ID] => 42
[TIMESTAMP_X] => 2015-09-07 20:29:18
[IBLOCK_ID] => 2
[NAME] => Полный текст
[ACTIVE] => Y
[SORT] => 500
[CODE] => FULL_TEXT_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 42
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Полный текст
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[PDF_RU] => Array
(
[ID] => 43
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF RUS
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_RU
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 43
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8394
[VALUE] => 266
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 266
[~DESCRIPTION] =>
[~NAME] => PDF RUS
[~DEFAULT_VALUE] =>
)
[PDF_EN] => Array
(
[ID] => 44
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF ENG
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 44
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8395
[VALUE] => 267
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 267
[~DESCRIPTION] =>
[~NAME] => PDF ENG
[~DEFAULT_VALUE] =>
)
[NAME_LONG] => Array
(
[ID] => 45
[TIMESTAMP_X] => 2023-04-13 00:55:00
[IBLOCK_ID] => 2
[NAME] => Название (для очень длинных заголовков)
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_LONG
[DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 45
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 80
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Название (для очень длинных заголовков)
[~DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
)
)
Gerard Wagemaker*
Raisa Gorbacheva Memorial Institute of Children Oncology, Hematology and Transplantation, St. Petersburg, Russia
Gerard Wagemaker, PhD, Professor, Erasmus University Rotterdam,
c/o PO Box 37048
3005 LA Rotterdam, The Netherlands
Phone: +31-6-51619585
E-mail: g.wagemaker@genetherapy.nl
Experimental studies
Array
(
[KEYWORDS] => Array
(
[ID] => 19
[TIMESTAMP_X] => 2015-09-03 10:46:01
[IBLOCK_ID] => 2
[NAME] => Ключевые слова
[ACTIVE] => Y
[SORT] => 500
[CODE] => KEYWORDS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 19
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 4
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => Y
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => Y
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8474
[1] => 8475
[2] => 8476
[3] => 8477
)
[VALUE] => Array
(
[0] => 696
[1] => 697
[2] => 698
[3] => 699
)
[DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 696
[1] => 697
[2] => 698
[3] => 699
)
[~DESCRIPTION] => Array
(
[0] =>
[1] =>
[2] =>
[3] =>
)
[~NAME] => Ключевые слова
[~DEFAULT_VALUE] =>
)
[SUBMITTED] => Array
(
[ID] => 20
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата подачи
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUBMITTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 20
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8445
[VALUE] => 10/10/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10/10/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата подачи
[~DEFAULT_VALUE] =>
)
[ACCEPTED] => Array
(
[ID] => 21
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата принятия
[ACTIVE] => Y
[SORT] => 500
[CODE] => ACCEPTED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 21
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8446
[VALUE] => 11/25/2016 12:00:00 am
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 11/25/2016 12:00:00 am
[~DESCRIPTION] =>
[~NAME] => Дата принятия
[~DEFAULT_VALUE] =>
)
[PUBLISHED] => Array
(
[ID] => 22
[TIMESTAMP_X] => 2015-09-02 17:21:42
[IBLOCK_ID] => 2
[NAME] => Дата публикации
[ACTIVE] => Y
[SORT] => 500
[CODE] => PUBLISHED
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 22
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => DateTime
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Дата публикации
[~DEFAULT_VALUE] =>
)
[CONTACT] => Array
(
[ID] => 23
[TIMESTAMP_X] => 2015-09-03 14:43:05
[IBLOCK_ID] => 2
[NAME] => Контакт
[ACTIVE] => Y
[SORT] => 500
[CODE] => CONTACT
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 23
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8447
[VALUE] => 695
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 695
[~DESCRIPTION] =>
[~NAME] => Контакт
[~DEFAULT_VALUE] =>
)
[AUTHORS] => Array
(
[ID] => 24
[TIMESTAMP_X] => 2015-09-03 10:45:07
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHORS
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => E
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => Y
[XML_ID] => 24
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 3
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] => EAutocomplete
[USER_TYPE_SETTINGS] => Array
(
[VIEW] => E
[SHOW_ADD] => Y
[MAX_WIDTH] => 0
[MIN_HEIGHT] => 24
[MAX_HEIGHT] => 1000
[BAN_SYM] => ,;
[REP_SYM] =>
[OTHER_REP_SYM] =>
[IBLOCK_MESS] => N
)
[HINT] =>
[PROPERTY_VALUE_ID] => Array
(
[0] => 8478
)
[VALUE] => Array
(
[0] => 695
)
[DESCRIPTION] => Array
(
[0] =>
)
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[0] => 695
)
[~DESCRIPTION] => Array
(
[0] =>
)
[~NAME] => Авторы
[~DEFAULT_VALUE] =>
)
[AUTHOR_RU] => Array
(
[ID] => 25
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Авторы
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 25
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8449
[VALUE] => Array
(
[TEXT] => Ильдар М. Бархатов, Николай Ю. Цветков, Даниил Е. Ершов, Марина Ю. Лаврухина, Алена И. Шакирова, Алиса Я.<br>
Поттер, Анна Г. Смирнова, Людмила С. Зубаровская, Борис В. Афанасьев<br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Ильдар М. Бархатов, Николай Ю. Цветков, Даниил Е. Ершов, Марина Ю. Лаврухина, Алена И. Шакирова, Алиса Я.
Поттер, Анна Г. Смирнова, Людмила С. Зубаровская, Борис В. Афанасьев
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Авторы
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_RU] => Array
(
[ID] => 26
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Организации
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 26
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8450
[VALUE] => Array
(
[TEXT] => Первый Санкт-Петербургский государственный медицинский Университет им. акад. И. П. Павлова Министерства<br>
Здравоохранения РФ, Санкт-Петербург, Россия<br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Первый Санкт-Петербургский государственный медицинский Университет им. акад. И. П. Павлова Министерства
Здравоохранения РФ, Санкт-Петербург, Россия
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Организации
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_RU] => Array
(
[ID] => 27
[TIMESTAMP_X] => 2015-09-02 18:01:20
[IBLOCK_ID] => 2
[NAME] => Описание/Резюме
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 27
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8451
[VALUE] => Array
(
[TEXT] => Наблюдаемая в ряде случаев гипофункция транс-
плантата является нередкой и обусловливает не-
обходимость разработки функциональных тестов
исследования клеток стромы с целью обоснования
показаний к совместной трансплантации гемопоэ-
тических и стромальных клеток костного мозга. Це-
лью данного исследования являлось изучение роли
стромальных клеток костного мозга в процессе при-
живления донорского костного мозга и их значения
в развитии осложнений после ТГСК. При анализе
функциональных характеристик стромальных кле-
ток у 38 реципиентов аллогенного трансплантата ге-
мопоэтических стволовых клеток было показано уве-
личение активности остеогенной дифференцировки
на фоне укорочения сроков восстановления кро-
ветворения. Вместе с тем, наличие более выражен-
ной способности к адипогенной дифференцировке
ассоциировано с меньшей вероятностью развития
гипофункции трансплантата. При исследовании ге-
мостимулирующей активности стромальных клеток
реципиента были отмечены факты, указывающие
на преимущественное влияние короткодистантных
ростовых факторов на посттрансплантационную
реконституцию гемопоэза. При этом более выра-
женная экспрессия молекул, опосредующих хоу-
минг ГСК в стромальных клетках реципиента перед
трансплантацией ассоциировано с более быстрым
восстановлением гемопоэза у трансплантированных
больных.
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Наблюдаемая в ряде случаев гипофункция транс-
плантата является нередкой и обусловливает не-
обходимость разработки функциональных тестов
исследования клеток стромы с целью обоснования
показаний к совместной трансплантации гемопоэ-
тических и стромальных клеток костного мозга. Це-
лью данного исследования являлось изучение роли
стромальных клеток костного мозга в процессе при-
живления донорского костного мозга и их значения
в развитии осложнений после ТГСК. При анализе
функциональных характеристик стромальных кле-
ток у 38 реципиентов аллогенного трансплантата ге-
мопоэтических стволовых клеток было показано уве-
личение активности остеогенной дифференцировки
на фоне укорочения сроков восстановления кро-
ветворения. Вместе с тем, наличие более выражен-
ной способности к адипогенной дифференцировке
ассоциировано с меньшей вероятностью развития
гипофункции трансплантата. При исследовании ге-
мостимулирующей активности стромальных клеток
реципиента были отмечены факты, указывающие
на преимущественное влияние короткодистантных
ростовых факторов на посттрансплантационную
реконституцию гемопоэза. При этом более выра-
женная экспрессия молекул, опосредующих хоу-
минг ГСК в стромальных клетках реципиента перед
трансплантацией ассоциировано с более быстрым
восстановлением гемопоэза у трансплантированных
больных.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Описание/Резюме
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[DOI] => Array
(
[ID] => 28
[TIMESTAMP_X] => 2016-04-06 14:11:12
[IBLOCK_ID] => 2
[NAME] => DOI
[ACTIVE] => Y
[SORT] => 500
[CODE] => DOI
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 28
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8452
[VALUE] => 10.18620/ctt-1866-8836-2016-5-4-71-77
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 10.18620/ctt-1866-8836-2016-5-4-71-77
[~DESCRIPTION] =>
[~NAME] => DOI
[~DEFAULT_VALUE] =>
)
[AUTHOR_EN] => Array
(
[ID] => 37
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Author
[ACTIVE] => Y
[SORT] => 500
[CODE] => AUTHOR_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 37
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8453
[VALUE] => Array
(
[TEXT] => Ildar M. Barkhatov, Nikolay Yu Tsvetkov, Daniil E. Ershov, Marina Y. Lavrukhina, Alena I. Shakirova, Alisa Ya. Potter,<br>
Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev<br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Ildar M. Barkhatov, Nikolay Yu Tsvetkov, Daniil E. Ershov, Marina Y. Lavrukhina, Alena I. Shakirova, Alisa Ya. Potter,
Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Author
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[ORGANIZATION_EN] => Array
(
[ID] => 38
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Organization
[ACTIVE] => Y
[SORT] => 500
[CODE] => ORGANIZATION_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 38
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8454
[VALUE] => Array
(
[TEXT] => R. M. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, and Chair of Hematology,<br>
Transfusiology and Transplantation, The First St. Petersburg State I. Pavlov Medical University, St. Petersburg, Russian<br>
Federation<br>
<br>
Dr. Ildar M.Barkhatov, PhD, Laboratory Head, R. M. Gorbacheva<br>
Memorial Research Institute of Children Oncology,<br>
Hematology and Transplantation, The First St. Petersburg<br>
State I. Pavlov Medical University, L.Tolstoy St. 6/8, 197022,<br>
St. Petersburg, Russian Federation<br>
<br>
Phone: +7(911) 778 2785<br>
E-mail: <a href="mailto:i.barkhatov@gmail.com">i.barkhatov@gmail.com</a><br>
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => R. M. Gorbacheva Memorial Research Institute of Children Oncology, Hematology and Transplantation, and Chair of Hematology,
Transfusiology and Transplantation, The First St. Petersburg State I. Pavlov Medical University, St. Petersburg, Russian
Federation
Dr. Ildar M.Barkhatov, PhD, Laboratory Head, R. M. Gorbacheva
Memorial Research Institute of Children Oncology,
Hematology and Transplantation, The First St. Petersburg
State I. Pavlov Medical University, L.Tolstoy St. 6/8, 197022,
St. Petersburg, Russian Federation
Phone: +7(911) 778 2785
E-mail: i.barkhatov@gmail.com
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Organization
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[SUMMARY_EN] => Array
(
[ID] => 39
[TIMESTAMP_X] => 2015-09-02 18:02:59
[IBLOCK_ID] => 2
[NAME] => Description / Summary
[ACTIVE] => Y
[SORT] => 500
[CODE] => SUMMARY_EN
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 39
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] => 8455
[VALUE] => Array
(
[TEXT] => Functional insufficiency of stem cell grafts is not rare in
hematopoietic stem cell transplantation (HSCT), thus
requiring further development of functional tests for recipient’s
stromal cells, in order to substantiate clinical indications
for co-transplantation of hematopoietic and
stromal bone marrow cells. The aim of this study was to
investigate a role of bone marrow stromal cells (BMSC) in
the donor bone marrow engraftment, and their importance
for development of complications after HSCT. Functional
analysis of BMSC cultures from thirty-eight recipients of
allogeneic hematopoietic stem cells has been shown that
induced osteogenic differentiation correlated with faster
hematopoiesis recovery in transplanted patients. Moreover,
a pronounced ability for adipogenic differentiation proved
to be associated with lower probability of the graft hypofunction.
A pronounced hemostimulatory activity of stromal
cells was observed in our study, thus suggesting effects
of short-range hematopoietic growth factors from BMSC
upon hematopoietic reconstitution. At the same time, a
more pronounced pre-transplant expression of molecules
which promote donor HSC homing by the recipient stromal
cells was associated with faster hematopoietic recovery
after transplantation.
[TYPE] => HTML
)
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Array
(
[TEXT] => Functional insufficiency of stem cell grafts is not rare in
hematopoietic stem cell transplantation (HSCT), thus
requiring further development of functional tests for recipient’s
stromal cells, in order to substantiate clinical indications
for co-transplantation of hematopoietic and
stromal bone marrow cells. The aim of this study was to
investigate a role of bone marrow stromal cells (BMSC) in
the donor bone marrow engraftment, and their importance
for development of complications after HSCT. Functional
analysis of BMSC cultures from thirty-eight recipients of
allogeneic hematopoietic stem cells has been shown that
induced osteogenic differentiation correlated with faster
hematopoiesis recovery in transplanted patients. Moreover,
a pronounced ability for adipogenic differentiation proved
to be associated with lower probability of the graft hypofunction.
A pronounced hemostimulatory activity of stromal
cells was observed in our study, thus suggesting effects
of short-range hematopoietic growth factors from BMSC
upon hematopoietic reconstitution. At the same time, a
more pronounced pre-transplant expression of molecules
which promote donor HSC homing by the recipient stromal
cells was associated with faster hematopoietic recovery
after transplantation.
[TYPE] => HTML
)
[~DESCRIPTION] =>
[~NAME] => Description / Summary
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[NAME_EN] => Array
(
[ID] => 40
[TIMESTAMP_X] => 2015-09-03 10:49:47
[IBLOCK_ID] => 2
[NAME] => Name
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 80
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 40
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => Y
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8456
[VALUE] => Impact of recipient marrow stromal cells functional characteristics on engraftment after allo-HSCT
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => Impact of recipient marrow stromal cells functional characteristics on engraftment after allo-HSCT
[~DESCRIPTION] =>
[~NAME] => Name
[~DEFAULT_VALUE] =>
)
[FULL_TEXT_RU] => Array
(
[ID] => 42
[TIMESTAMP_X] => 2015-09-07 20:29:18
[IBLOCK_ID] => 2
[NAME] => Полный текст
[ACTIVE] => Y
[SORT] => 500
[CODE] => FULL_TEXT_RU
[DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 42
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 200
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Полный текст
[~DEFAULT_VALUE] => Array
(
[TEXT] =>
[TYPE] => HTML
)
)
[PDF_RU] => Array
(
[ID] => 43
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF RUS
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_RU
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 43
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8479
[VALUE] => 280
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 280
[~DESCRIPTION] =>
[~NAME] => PDF RUS
[~DEFAULT_VALUE] =>
)
[PDF_EN] => Array
(
[ID] => 44
[TIMESTAMP_X] => 2015-09-09 16:05:20
[IBLOCK_ID] => 2
[NAME] => PDF ENG
[ACTIVE] => Y
[SORT] => 500
[CODE] => PDF_EN
[DEFAULT_VALUE] =>
[PROPERTY_TYPE] => F
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 44
[FILE_TYPE] => doc, txt, rtf, pdf
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] =>
[USER_TYPE_SETTINGS] =>
[HINT] =>
[PROPERTY_VALUE_ID] => 8480
[VALUE] => 281
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] => 281
[~DESCRIPTION] =>
[~NAME] => PDF ENG
[~DEFAULT_VALUE] =>
)
[NAME_LONG] => Array
(
[ID] => 45
[TIMESTAMP_X] => 2023-04-13 00:55:00
[IBLOCK_ID] => 2
[NAME] => Название (для очень длинных заголовков)
[ACTIVE] => Y
[SORT] => 500
[CODE] => NAME_LONG
[DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
[PROPERTY_TYPE] => S
[ROW_COUNT] => 1
[COL_COUNT] => 30
[LIST_TYPE] => L
[MULTIPLE] => N
[XML_ID] => 45
[FILE_TYPE] =>
[MULTIPLE_CNT] => 5
[TMP_ID] =>
[LINK_IBLOCK_ID] => 0
[WITH_DESCRIPTION] => N
[SEARCHABLE] => N
[FILTRABLE] => N
[IS_REQUIRED] => N
[VERSION] => 1
[USER_TYPE] => HTML
[USER_TYPE_SETTINGS] => Array
(
[height] => 80
)
[HINT] =>
[PROPERTY_VALUE_ID] =>
[VALUE] =>
[DESCRIPTION] =>
[VALUE_ENUM] =>
[VALUE_XML_ID] =>
[VALUE_SORT] =>
[~VALUE] =>
[~DESCRIPTION] =>
[~NAME] => Название (для очень длинных заголовков)
[~DEFAULT_VALUE] => Array
(
[TYPE] => HTML
[TEXT] =>
)
)
)
Ildar M. Barkhatov, Nikolay Yu Tsvetkov, Daniil E. Ershov, Marina Y. Lavrukhina, Alena I. Shakirova, Alisa Ya. Potter,
Anna G. Smirnova, Ludmila S. Zubarovskaya, Boris V. Afanasyev
Transfusiology and Transplantation, The First St. Petersburg State I. Pavlov Medical University, St. Petersburg, Russian
Federation
Dr. Ildar M.Barkhatov, PhD, Laboratory Head, R. M. Gorbacheva
Memorial Research Institute of Children Oncology,
Hematology and Transplantation, The First St. Petersburg
State I. Pavlov Medical University, L.Tolstoy St. 6/8, 197022,
St. Petersburg, Russian Federation
Phone: +7(911) 778 2785
E-mail: i.barkhatov@gmail.com