Graft enrichment with host-derived mature dendritic cells does not allow long-term engraftment in canine DLA-identical hematopoietic stem cell transplantation recipients after conditioning with 1 Gy TBI
Sandra Lange1, Simone Altmann1, Heike Vogel2, Volker Weirich3, Mathias Freund1, Christian Junghanss1
1Department of Internal Medicine, Medical Clinic III – Hematoloy, Oncology, Palliative Medicine; 2Department of Radiation Oncology; 3Institute of Legal Medicine, Medical Faculty, University of Rostock, Rostock, Germany
Accepted 28 February 2012
Published 30 April 2012
Summary
The canine nonmyeloablative hematopoietic stem cell transplantation (HSCT) model allows the establishment of mixed hematopoietic chimerism following 2 Gy total body irradiation (TBI) conditioning, but in general fails to do so after 1 Gy TBI. Several studies have used this model to investigate cellular-based therapies as well as pharmaceutical-based approaches in their ability to support engraftment. In the present study we investigated the impact of an intravenous administration of host derived dendritic cells (DC) during (day 0) and/or 1 week post transplantation on long-term engraftment in a canine HSCT model. Dogs were transplanted after conditioning with 1 Gy TBI and received immunosuppression consisting of 15 mg/kg cyclosporin A BID PO (days -1 to +35) in combination with 20 mg/kg mycophenolate mofetil BID PO (days 0 to +27). All dogs transiently engrafted but eventually rejected the graft by a median 56–84 days. Therefore, additional DC administration failed to significantly improve graft survival compared to historical controls. However, administration of DC twice seemed to be superior to a single administration when looking at their ability to support initial engraftment. Future studies might therefore aim at more frequent DC administrations to support successful HSCT in this setting.
Keywords
allogeneic hematopoietic stem cell transplantation, nonmyeloablative, vaccination, dendritic cells, dog, chimerism, engraftment
Introduction
In the canine hematopoietic stem cell transplantation (HSCT) model, durable engraftment of allogeneic dog leukocyte antigen (DLA)-identical littermate bone marrow cells can be achieved following nonmyeloablative conditioning with 2 Gy of total body irradiation (TBI) in combination with pre- and post-transplant immunosuppression consisting of cyclosporin A (CSA) and mycophenolate mofetil (MMF) [1]. A reduction of the radiation dose to 1 Gy TBI resulted in only transient engraftment, and led eventually to graft rejection in this model [1]. Therefore, combinations of 1 Gy HSCT with different forms of immunotherapy were investigated. Post-graft vaccination with recipient hematopoietic cell lysates as well as graft augmentation with donor-derived monocyte-derived dendritic cells (DC) did not support durable engraftment after 1 Gy TBI [2]. Pre-transplant immunosuppression using CTLA4Ig or Pentostatin or graft modification with donor peripheral blood mononuclear cells (PBMC) was shown to improve engraftment to some extent, but long-term engraftment was only seen in 22–63% of the HSCT [5]. Recently, Mielcarek et al. studied 9 different immunosuppressive and/or tolerance-inducing regimens using a TBI dose of 0.5 Gy. None of these strategies was sufficient to ensure sustained engraftment after HSCT [6].
The role of DC as key cells of the immune system is well known. Mature DC especially are highly immunogenic and efficiently induce T-cell proliferation [7]. Furthermore it has been shown that recipient antigen-presenting cells are required to trigger efficiently alloreactive donor T-cells early after total body irradiation [8]. Currently, monocyte-derived DC are the most widely used, since generation of bone-marrow-derived DC is much more invasive. However, when using bone marrow higher numbers of DC were obtained, with a higher capacity to stimulate allogeneic T-cell responses compared to monocyte-derived DC [9].
Therefore, this study investigated whether the addition of monocyte-derived as well as bone marrow-derived mature DC of host origin to the graft and/or administration of DC one week post-transplantation facilitates stable marrow engraftment after 1 Gy conditioning in the canine HSCT model.
Materials and Methods
Experiments were approved by the review board of the state Mecklenburg-Vorpommern (Landesamt für Landwirtschaft, Lebensmittelsicherheit und Fischerei M-V, Germany).
Animals
Dogs were purchased from commercial kennels licensed by the German Department of Agriculture. All animals were de-wormed and immunized against rabies, parainfluenca, leptospirosis, distemper, hepatitis, and parvovirus. DLA-identical donor/recipient sibling pairs were selected on the basis of matching for highly polymorphic DLA class I and class II microsatellite markers [1].
Hematopoietic stem cell transplantation
HSCT was performed as described previously [1]. Briefly, 7 recipient dogs were conditioned with nonmyeloablative TBI at a single dose of 1 Gy using a high-energy linear accelerator (Siemens Primus; 10 MV X-ray) with a dose rate of 0.25 Gy/min. Bone marrow from DLA-identical siblings was collected under general anesthesia from the humeri, femora, and iliac crest and infused intravenously into the recipients within 24 h after TBI. Immunosuppression consisted of CSA at 15 mg/kg orally 2 times daily on days -1 to +35 in combination with MMF at 20 mg/kg orally 2 times daily on days 0 to +27. In addition to the graft, the dogs were given i.v. infusions of (I) monocyte-derived mature DC of host origin (MoDC) once on day +5 (n=2, group I) and (II) MoDC or CD34+-derived mature DC (CD34+-DC) of host origin twice: once on day 0 together with the graft and once on median day +7 (range +5 to +7) (n=5, group II). The clinical status of recipients was checked twice daily.
Cell isolation and generation of DC
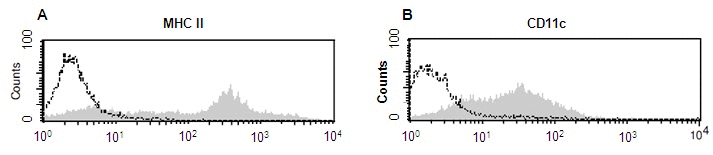
Monocytes were isolated from 300 ml peripheral blood of the recipient at day -2 (group I, one-time DC administration) and at days 7 and -2, respectively (n=2, group II, double DC administration) before transplantation by using an AutoMACS device (Miltenyi Biotec, Bergisch-Gladbach, Germany) [2]. Cells were cultured in 24-well plates for 7 days in RPMI-medium supplemented with 10% dog serum, 1% penicillin/streptomycin, 500 U/ml canine IL-4 and 100 ng/ml human GM-CSF. At day 5 of culture 10 ng/ml TNF-alpha was added to the medium for DC maturation. For generation of CD34+-cell derived DC (n=3; group II, double DC administration) 100 ml of bone marrow were aspirated on days -14 and -7 before transplantation and CD34+ cells were isolated using the AutoMACS device. CD34+ cells were cultured as described previously [10]. For in vitro characterization of cultured DC expression of the DC-specific cell surface marker MHC II and CD11c was analyzed by flow cytometry (Fig. 1).
Figure 1. Analysis of surface marker expression of MHC II (A) and CD11c (B) by flow cytometry. Each histogram represents an overlay of the specific monoclonal antibodies (filled histograms) and isotype-control monoclonal antibodies (solid lines). The histograms show one representative example of bone marrow-derived DC after 14 days of cultivation
Hematopoietic chimerism analysis
The recipients' peripheral blood was collected weekly. Granulocytes and PBMC were separated by Ficoll-Hypaque density gradient centrifugation (density 1.074 g/ml) and genomic DNA of the cell fractions was isolated (Nucleobond CB 100; Macherey-Nagel, Düren, Germany). Polymorphic tetranucleotide repeats that differed between donors and recipients were amplified and quantified as described previously [11]. Engraftment was defined as detection of >5% of donor-derived DNA. Graft rejection was defined as detection of no donor-derived DNA in the peripheral blood and the bone marrow.
Statistics
The distribution of data was described using medians and ranges. Differences between treatment groups were estimated according to the Mann-Whitney U-Test. Possible association between TNC numbers and their impact on graft rejection was assessed using the Spearman correlation analysis. Probability of p<0.05 was considered significant.
Results
HSCT recipients received DLA-identical marrow grafts containing a median of 4.9 (range 2.6–7.4) x 108 total nucleated cells (TNC)/kg intravenously within 24 h after TBI. The number of DC applied were a median 10.3 (range 6.0–14.5) x 105 DC/kg and 4.7 (range 1.2–7.2) x 105 DC/kg per dose in groups I and II, respectively.
The results of chimerism analyses are depicted in Figure 2. All dogs showed an initial engraftment in the granulocytes and PBMC compartments. The maximum donor chimerisms of the granulocytes accounted for a median 19% (range 14–23) and 33% (range 12–59) in groups I and II, respectively. The maximum PBMC donor chimerisms reached median values of 12% (range 11–12) and 17% (range 14–26) for both groups. Median times to maximum granulocytes chimerisms were 32 days (range 14–49) and 42 days (range 28–56). PBMC chimerisms peaked at days 21 (range 14–28) and 28 (range 28–35) in groups I and II, respectively.
Figure 2. Engraftment kinetics of donor peripheral blood granulocytes (A) and PBMC (B) after 1 Gy HSCT. Dogs received either MoDC vaccination on day +5 (group I, dotted lines) or two-time administration of MoDC and CD34+-DC on day 0 together with the graft and on median day +7 (range +5 to +7) as i.v. vaccination, respectively (group II, solid lines)
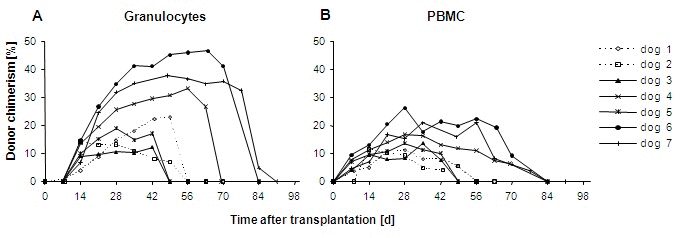
Induction of long-term engraftment could not be achieved in any dog. Rejection of grafts occurred after a median 56 days (range 56–56) and 84 days (range 49–91), respectively. Spearman correlation analysis revealed a significant association between body weight-adjusted numbers of TNC in the graft and time of graft rejection (r=0.871, p=0.01). The maximum chimerism in the granulocyte compartment tended to correlate to the number of TNC/kg as well (r=0.739, p=0.058).
Of interest, if taking into consideration the influence of TNC counts on graft rejection and chimerism, is the comparison of dog 1 (4.9 TNC/kg, 1x DC) with dog 4 (4.9 TNC/kg, 2x DC) as well as dog 2 (5.4 TNC/kg, 1x DC) with dog 6 (5.5 TNC/kg, 2x DC). These indicate a trend toward lower maximum granulocytes (23% vs 33% and 14% vs 59%) and PBMC donor chimerisms (11% vs 17% and 12% vs 26%) and shortened allograft survival (56 d vs 84 d, both) after a single DC administration.
Discussion
The canine nonmyeloablative HSCT model can be used to investigate novel approaches aiming at improved engraftment as well as long-term graft survival [6]. In the present study we investigated whether the intravenous application of host-derived DC at the time of HSCT and/or one week after HSCT improves engraftment.
All dogs in our study engrafted. In comparison to our own historical 2 Gy conditioned control animals the engraftment was delayed after 1 Gy conditioning plus DC administration, which lead to significant differences in engraftment kinetics [2]. The maximum levels of donor PBMC and granulocytes attained after 1 Gy TBI in combination with DC application were clearly lower than the maximum chimerisms of 75 and 41% in the granulocytes and PBMC compartments of 2 Gy control animals as well [2]. Times to maximum chimerism were not significantly different compared to the 2 Gy historical control (35 and 29 days, respectively).
No long-term engraftment could be achieved in any of the dogs in this study. This contrasts to the 2 Gy conditioning, which allows sustained engraftment in the majority of recipients [1, 2]. We hypothesize that the failure of long term engraftment occurred due to the delayed engraftment kinetics and the overall reduced percentage of donor-derived hematopoietic cells. Storb et al. have investigated pharmaceutical approaches combined with 1 Gy conditioning in regards to engraftment in the canine HSCT model. Rejection occurred in their studies 3–12 weeks after HSCT [1]. This corresponds to the time of graft rejection in the present study.
Our hypothesis that additional host DC administrations can sufficiently support engraftment and prolong graft survival after 1 Gy TBI could not be substantiated by our study. Instead, the number of TNC transplanted seemed to have an impact on graft rejection. This result supports previous data that showed a trend for decreased risk of graft rejection in dogs when transplanted with higher numbers of TNC [12].
A comparison of results regarding host DC application once versus twice after HSCT was performed after adjustment for the TNC/kg infused. Although numbers are low, our data indicate that two applications of host-derived DC i.e., concomitant to and one week after HSCT tend to enhance allo-immune response in the graft-versus-host direction leading to longer graft survival and higher peak chimerisms. An influence of the origin of DC (MoDC versus CD34+-DC) on engraftment could not be suggested from this study; additionally, numbers were small. This does not reflect previous data that shows a higher capacity to stimulate allogeneic T-lymphocyte reactivity for bone-marrow derived DC compared to MoDC [9]. Differences in the applied methods, i.e., the use of unseparated bone marrow mononuclear cells, the human origin of cells, and the in vitro analyses may explain the different immunological response in the previous study.
The role of DC in the graft is not yet completely understood. In contrast to our results, addition of plasmacytoid precursor DC to the graft significantly enhanced HSC engraftment in a mouse model [13]. However, in this study a myeloablative conditioning regimen was applied. In a clinical trial by Reddy et al. high DC numbers during engraftment were associated with better survival and decreased incidence of relapse [14]. However, the authors suggested that only the number of DC reconstituted in the recipient was pivotal. In contrast, another study showed that higher DC counts in donor bone marrow caused an increase in relapse rates following HSCT [15]. Reasons for the variable immunologic responses may be the heterogeneity of the DC population as well as different states of differentiation, maturation, or activation of the DC in these studies [16, 17].
In conclusion, our data indicates that application of host DC concomitant to the HSCT and/or one week after HSCT is not sufficient to support stable engraftment in a canine nonmyeloablative 1 Gy TBI HSCT model. Since the double administration seemed to be superior compared to a single boost further studies might address whether more frequent DC applications support engraftment more efficiently.
Conflict of Interest
The authors of this paper declare no financial or personal conflict of interest.
Acknowledgements
The authors thank all the staff of the animal care facility. We also thank Anett Sekora and Gudrun Knuebel (both from Department of Internal Medicine, Medical Clinic III – Hematoloy, Oncology, Palliative Medicine, University of Rostock, Germany) for technical assistance. This work was supported by the German Research Council (Deutsche Forschungsgemeinschaft) grants JU 417/2-1, 2 2.
References
4. Panse JP, Storb R, Storer B, Santos EB, Wentzel C, Sandmaier BM. Prolonged allogeneic marrow engraftment following nonmyeloablative conditioning using 100 cGy total body irradiation and pentostatin before and pharmacological immunosuppression after transplantation. Transplantation. 2005;80:1518-1521.
5. Zaucha JM, Zellmer E, Georges G, et al. G-CSF-mobilized peripheral blood mononuclear cells added to marrow facilitates engraftment in nonmyeloablated canine recipients: CD3 cells are required. Biol Blood Marrow Transplant. 2001;7:613-619. doi: 10.1053/bbmt.2001.v7.pm11760149.
6. Mielcarek M, Torok-Storb B, Storb R. Pharmacological Immunosuppression Reduces But Does Not Eliminate the Need for Total-Body Irradiation in Nonmyeloablative Conditioning Regimens for Hematopoietic Cell Transplantation. Biol Blood Marrow Transplant. 2011 Jan 8. [Epub ahead of print]. doi: 10.1016/j.bbmt.2011.01.003.
9. Bai L, Feuerer M, Beckhove P, et al. Generation of dendritic cells from human bone marrow mononuclear cells: advantages for clinical application in comparison to peripheral blood monocyte derived cells. Int J Oncol. 2002;20:247-253.
10. Hägglund HG, McSweeney PA, Mathioudakis G, et al. Ex vivo expansion of canine dendritic cells from CD34+ bone marrow progenitor cells. Transplantation. 2000;70:1437-1442.
11. Hilgendorf I, Weirich V, Zeng L, et al. Canine haematopoietic chimerism analyses by semiquantitative fluorescence detection of variable number of tandem repeat polymorphism. Vet Res Commun. 2005;29:103-110. doi: 10.1023/B:VERC.0000047486.01458.c5.
12. Baron F, Sandmaier BM, Zellmer E, Sorror M, Storer B, Storb R. Failure of donor lymphocyte infusion to prevent graft rejection in dogs given DLA-identical marrow after 1 Gy of total body irradiation. Biol Blood Marrow Transplant. 2006;12:813-817. doi: 10.1016/j.bbmt.2006.05.001.


